30 July 2020: Clinical Research
Identification and Expression of Several Circular RNAs and Knockdown of hsa_circ_0005556 Exerts Oncogenic Functions by miR-767-5p in Gastric Cancer
Hanke Geng1BCEF, Kaixuan Li1BCDF, Qi Pan2BCEF, Shaohui Tao1BCEF, Chunjuan Li1BDF, Haiwei Zhao1BF, Xiaojun Zhao1ACEFG*DOI: 10.12659/MSM.921163
Med Sci Monit 2020; 26:e921163
Abstract
BACKGROUND: Gastric cancer (GC) remains one of the most fatal digestive cancers in the world; nevertheless, its etiology remains vague. With the development of bioinformatics analysis, numerous circular RNAs (circRNAs) have been found to be dysregulated in GC. However, the functions of a large portion of dysregulated circRNAs in GC need further validation. In this study, we aimed to validate the biological functions of circ_0005556, which was previously identified to be dysregulated in GC.
MATERIAL AND METHODS: Levels of circRNAs and miRNAs in GC tissues and cells were estimated by qRT-PCR. The target miRNAs of circ_0005556 were predicted by bioinformatics methods. The interplay between circ_0005556 and miR-767-5p was validated by dual-luciferase reporter and circRNA immunoprecipitation assays. The effects of circ_0005556 and miR-767-5p on GC cell viability, apoptosis, migration, and invasion were assessed by MTT, flow cytometry, wound-healing and in vitro transwell experiments, respectively.
RESULTS: The upregulation of circ_0005556 was validated by qRT-PCR in GC tissues and cells, and a higher circ_0005556 level indicated a poorer prognosis. miR-767-5p was demonstrated to target circ_0005556 in GC cells, and a negative correlation was found between their expression levels in GC tissues. Knockdown of circ_0005556 promoted miR-767-5p expression in GC cells. Knockdown of circ_0005556 was revealed to repress GC cell viability, invasion, and migration and to promote GC cell apoptosis. Moreover, overexpression of miR-767-5p could significantly augment the repressive impacts of circ_0005556 knockdown on GC cell progression in vitro.
CONCLUSIONS: The in vitro knockdown of circ_0005556 remarkably repressed GC cell progression by increasing the expression of miR-767-5p.
Keywords: Apoptosis Regulatory Proteins, Base Pairing, Base Sequence, carcinogenesis, Genes, Reporter, Luciferases, RNA, Circular, RNA, Small Interfering
Background
Gastric cancer (GC) remains one of the most fatal digestive tumors worldwide. Despite tremendous progress in preventive, diagnostic, and therapeutic options for GC in the past decades, the incidence, mortality, and prognosis of GC are still disappointing [1]. It was estimated by the American Cancer Society that approximately 27 510 new GC cases, including 17 230 males and 10 280 females, will be diagnosed as GC in the United States in 2019 [2]. Among these, 11 140 deaths will occur, and the fatality rate will reach 40% [2]. Although the combined therapy of surgical removal and chemotherapy has been proven to be a relatively effective therapeutic strategy for GC, the 5-year overall survival rate of GC patients remains below 30% in most areas of the world [3]. A great deal of effort was made during the past decades to develop effective plasma-based biomarkers for diagnosing GC [4–6]. Nevertheless, due to their low sensitivity and specificity, the most commonly used GC screening technologies, including endoscopy- or X-ray-based examinations, can hardly be replaced by the current GC-related plasma markers, such as CA19-9, carcinoembryonic antigen, and carbohydrate antigen 72-4 [7]. The complicated molecular mechanisms of GC may be the predominate reason for the difficulty of GC diagnosis and therapy. Therefore, it is extremely important to understand the details of the molecular characteristics of GC.
Circular RNAs (circRNAs) are novel noncoding RNAs that have been identified in recent years, with very wide expression levels in the eukaryotic transcriptome [8]. CircRNAs are primarily located in the cytoplasm and can also be sorted into exosomes [9]. Unlike linear RNAs, circRNAs have no 5′ cap or 3′poly(A) tail [9]. Its unique circular structure makes it possess the ability to resist digestion of endogenous nucleotide enzymes; thus, it can maintain a relatively stable expression level in cells [10]. In view of its high stability, circRNAs are believed to be promising biomarkers [11]. Interestingly, numerous studies have shown critical roles for circRNAs in modulating tumor initiation and progression [12]. With the rapid development of molecular and biological techniques and bioinformatics analyses, a growing number of circRNAs have been found to be dysregulated in GC, suggesting that circRNAs play key roles during GC pathogenesis [13]. Recently, Tianwen Li et al. explored the expression pattern of circRNAs in plasma samples from GC patients and healthy controls using a circRNA microarray [14]. A total of 343 circRNAs were demonstrated to be dysregulated in GC patients’ plasma compared to their healthy counterparts; moreover, they found 17 dysregulated circRNAs in plasma samples of GC patients compared to their tissue samples [14]. However, they did not examine the biological functions of these dysregulated circRNAs during GC tumorigenesis.
In this study, we selected circ_0005556, one of the dysregulated circRNAs, for a functional study. We aimed to investigate whether circ_0005556 plays a role in GC cell viability, apoptosis, migration, and invasion and attempted to understand its molecular mechanism.
Material and Methods
GC TUMOR COLLECTION AND:
GC and matched normal tissue specimens were gathered from patients who were diagnosed with GC at the Second Affiliated Hospital of Xi’an Medical University and had not received chemotherapy. Samples were kept at −80°C until the study began. GC patients involved in this study provided written informed consent.
CELL CULTURE:
Five human GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823) and 1 human normal gastric mucosa cell line (GES-1) were provided by the Chinese Academy of Science Cell Bank (Shanghai, China). Cells were cultured in RPMI-1640 medium (Invitrogen, USA) containing 10% fetal bovine serum (Gibco, USA). Both mycoplasma contamination and STR tests were conducted on all of these cells.
QUANTITATIVE REAL-TIME PCR (QRT-PCR) ASSAY:
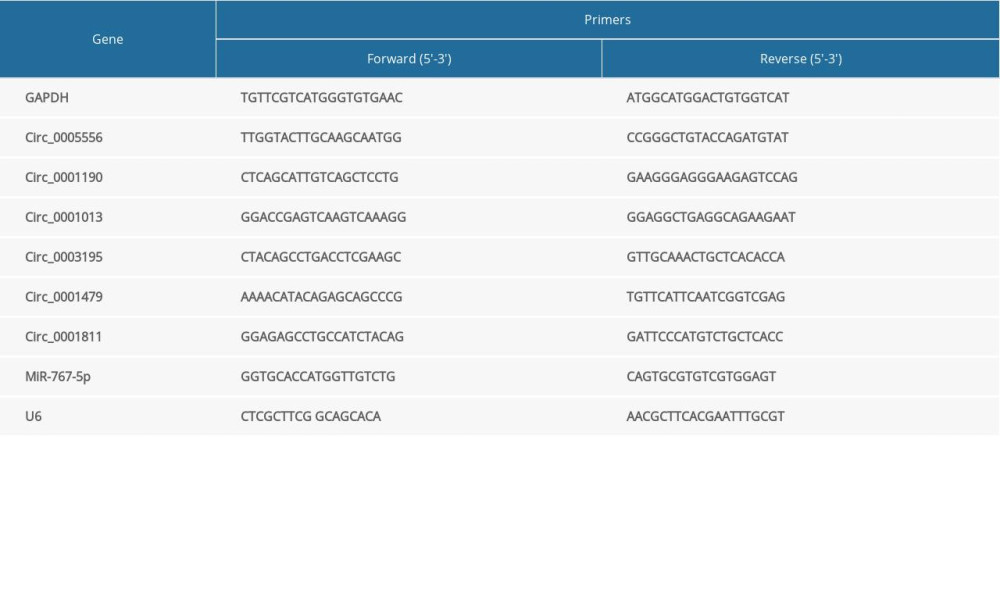
RNA from GC tissues and cells was extracted by the application of TRIzol reagent (Invitrogen, USA) following the instructions of the manufacturer. RNA quality was estimated by a NanoDrop 2000c (Thermo Scientific, USA). Next, 4 μg of examined RNAs were used as templates to synthesize cDNAs using PrimeScript RT reagent (Takara, Japan). Relative expression levels of circRNAs and miRNAs were assessed through SYBR Premix Taq (Takara) following the manufacturer’s protocol. The PCR process was finished on an ABI 7300 Real-Time System (Ambion, USA). GAPDH was used as the internal control for circRNAs, and U6 was used as the internal control for miRNAs. The relative expression levels of genes were calculated using 2−ΔΔCt method [15]. Primers are shown in Table 1.
RNA TRANSFECTION:
The si-circ_0005556, miR-767-5p and their scrambled controls were from GenePharma (Shanghai, China). For cell transfections, GC cells were plated into 96-well plates and cultured at 37°C for 16 h. Cells were transfected with the indicated RNAs using Lipofectamine 3000 (Invitrogen).
LUCIFERASE REPORTER ASSAY:
A luciferase reporter experiment was performed to validate the interplay between circ_0005556 and miR-767-5p. Circ_0005556-wild-type (circ_0005556-WT) and circ_0005556-mutant (circ_0005556-Mut) plasmids were established by inserting WT and Mut miR-767-5p binding sequences of circ_0005556 into a pCMV6 vector (Origene, Rockville, MD), respectively. Circ_0005556-WT or circ_0005556-Mut was cotransfected into GC cells with miR-767-5p mimics or a scrambled control, followed by detection of luciferase intensity using the Dual-Luciferase Assay System Kit (Promega).
CIRCRNA IMMUNOPRECIPITATION (CIRCRIP):
A specific biotin-labeled circ_0005556 probe was obtained from Sangon Biotech (Shanghai, China). BGC-823 cells were fixed in 4% formaldehyde for 15 min and then centrifuged at 10 000×g/min at 4°C. Next, 100 μl of the supernatants were incubated overnight with specific biotin-labeled circ_0005556 a streptavidin-coated probe mixture (Dynabeads, Invitrogen) at 30°C. Subsequently, the mixture was analyzed by RT-PCR for targeted miRNA detection.
Cell viability assessment The impact of circ_0005556 and miR-767-5p on GC cell viability were estimated through 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Sigma, USA). Briefly, treated GC cells were plated into 96-well plates at a concentration of 2×105 cells/well, and MTT solution was then added into each well and incubated overnight. The optical density was examined at 570 nm with a microtiter plate reader.
CELL APOPTOSIS ANALYSIS:
PI and Annexin V-FITC double staining followed by flow cytometry detection was employed to estimate the number of apoptotic cells in treated GC cells. After treatment with the indicated RNAs, GC cells were fixed in 75% ethanol and stained with PI and Annexin V-FITC for 30 min. Finally, cells were subjected to flow cytometry analysis.
CELL MIGRATION AND INVASION EVALUATION:
Migratory or invasive ability was measured by wound-healing and transwell assays, respectively. For the wound-healing assay, treated GC cells (5×105) were seeded into 6-well plates and cultured at 37°C until approximately 90% confluence [16]. A straight wound was created using a sterile pipet tip on the surface of cells, and then cells were cultured at 37°C for another 48 h. The wounds were photographed at 0 h and 48 h after wound creation. For the transwell experiment, 500 μl of culture medium containing treated GC cells was added into a Matrigel-coated upper chamber, and FBS-containing culture medium (10%) was added into the bottom chamber. After incubation for 48 h, the bottom of the chamber was incubated with methanol and DAPI for 10 min. The number of cells that invaded through the membrane was counted using an inverted microscope.
STATISTICAL ANALYSIS:
Data are shown as the mean±SEM, and one-way ANOVA was performed on GraphPad Prism (Version 7.0, USA) to compare the differences between groups.
Results
VALIDATION OF 6 DYSREGULATED CIRCRNAS IN GC:
A previous study analyzed the expression pattern of circRNAs in tissue and plasma specimens collected from GC patients through an intersection analysis [14]. Their results indicated that 14 circRNAs were downregulated and 3 circRNAs were upregulated in GC patients’ plasma (Figure 1). To further validate this result, we selected 6 circRNAs (circ_0005556, circ_0001190, circ_0001013, circ_0003195, circ_0001479, and circ_0001181) and examined their levels in 20 pairs of GC and adjacent normal tissues. As shown in Figure 2, circ_0005556 and circ_0001013 were all remarkably increased, while circ_0001190, circ_0003195 and circ_0001811 were found to be decreased in GC tissues compared to normal tissues. There was no change in the expression levels of circ_0001479 between GC and normal tissues. Therefore, the trends of circ_0005556, circ_0001190, circ_0001013, circ_0003195, and circ_0001181 levels were consistent with the results from a previous study [14].
THE CIRC_0005556 LEVEL WAS HIGHER IN GC AND WAS CORRELATED WITH A WORSE PROGNOSIS:
To investigate whether these dysregulated circRNAs play a functional role during GC tumorigenesis, circ_0005556 was selected for further study in this research. We first examined its level in 118 pairs of GC and matched normal adjacent tissue specimens via qRT-PCR, and circ_0005556 was revealed to be dramatically elevated in GC tissues compared to normal tissues (Figure 3A, *** P<0.001). Subsequently, we analyzed the diagnostic value of circ_0005556 using these 118 paired tissue specimens by receiver operating characteristic curve (ROC) analysis. The results showed that the area under the ROC (AUC) of circ_0005556 in GC tissues was 0.635, and the sensitivity and specificity reached 56.48% and 72.52%, respectively (Figure 3B). Moreover, these 118 patients were divided into a high-circ_0005556 level group and a low-circ_0005556 group according to the cutoff levels detected by the ROC analysis. The results from the Kaplan-Meier survival curve showed that GC patients with a high circ_0005556 level were markedly correlated with poor prognosis compared to those patients with a low circ_0005556 level (Figure 3C, ** P<0.01). In addition, we tested the level of circ_0005556 in the plasma samples of these 118 GC patients and matched healthy controls. The results showed that the circ_0005556 level was significantly higher in the plasma of GC patients than that in health controls (Figure 3D, *** P<0.001). Moreover, the results of ROC also revealed that the AUC of circ_0005556 in the plasma samples of GC patients was 0.635, and the sensitivity and specificity were 72.0% and 60.2%, respectively (Figure 3E). Correlation analysis showed that there is a positive correlation between the GC tissue plasma circ_0005556 levels (Figure 3F, P=0.0089, R2=0.0576). In addition, the downregulation of circ_0005556 was still observed in postoperative GC patients’ plasma compared to preoperative plasma (Figure 3G, P<0.05). These findings further confirmed the upregulation of circ_0005556 in GC tissue and plasma samples, and elevated circ_0005556 was correlated with a poor overall survival rate.
CIRC_0005556 WAS BOUND BY MIR-767-5P:
The expression levels of circ_0005556 in GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823) were also estimated by qRT-PCR. Compared to the human normal gastric mucosa cell line (GES-1), circ_0005556 was highly expressed in the SGC-7901, AGS, HGC-27, and BGC-823 cell lines; however, no significant alteration was observed in the MGC-803 cell line (Figure 4A, * P<0.05, ** P<0.01, *** P<0.001). To explore the potential mechanism of circ_0005556 in GC tumorigenesis, a bioinformatics method was employed to screen the putative miRNA targets. The results indicated that circ_0005556 possessed multiple putative miRNA targets, including miR-548c-3p, miR-548p, miR-587, miR-659, and miR-767-5p (Figure 4B, upper panel). Moreover, we found that miR-548c-3p and miR-767-5p were drastically decreased in the BGC-823 cell line, while miR-548p, miR-587, and miR-659 exhibited no significant changes (Figure 4B, lower panel). Due to miR-767-5p showing a higher fold change compared to miR-548c-3p in BGC-823 cells, we selected miR-767-5p for further study. A dual-luciferase reporter assay was employed to validate the interplay between miR-767-5p and circ_0005556 in BGC-823 cells. Cotransfection of miR-767-5p and circ_0005556-WT plasmid markedly attenuated the luciferase activity of BGC-823 cells compared to that in the circ_0005556 and miR-NC cotransfected groups; the luciferase intensity of BGC-823 cells driven by circ_0005556-Mut was not affected by either miR-767-5p or miR-NC (Figure 4C, * P<0.05). Furthermore, a circ_0005556-specific probe was used to conduct RNA precipitation to confirm the interaction between circ_0005556 and miR-767-5p in BGC-823 cells. The circ_0005556-pulled-down RNAs were purified and detected by RT-PCR. As expected, a specific enrichment of circ_0005556 and miR-767-5p was observed in the circ_0005556-specific probe group compared to the control group (Figure 4D, *** P<0.001). The level of miR-767-5p was also examined in 118 pairs of GC and normal tissues by qRT-PCR. The results indicated that miR-767-5p was remarkably decreased in GC tissues compared to normal tissues (Figure 4E, *** P<0.001). The correlation analysis showed that in GC tissues there was a negative correlation between the circ_0005556 and miR-767-5p levels (Figure 4F, P=0.0104, R2=0.0553). These findings suggested that circ_0005556 was targeted by miR-767-5p in GC.
MIR-767-5P TRANSFECTION ENHANCED THE INHIBITORY IMPACTS OF CIRC_0005556 KNOCKDOWN ON GC CELLS:
To investigate the functional correlation between circ_0005556 and miR-767-5p during GC tumorigenesis, we assessed the cell proliferation, apoptosis, migration, and invasion in si-circ_0005556- or miR-767-5p-transfected BGC-823 cells. qRT-PCR analysis of miR-767-5p indicated that miR-767-5p was significantly increased in si-circ_0005556-transfected BGC-823 cells compared to the scrambled group (Figure 5A, ** P<0.01). Cotransfection of si-circ_0005556 and miR-767-5p augmented the upregulation of miR-767-5p induced by the si-circ_0005556 transfection alone (Figure 5A, ## P<0.01). MTT analysis of the GC cell viability indicated that the circ_0005556 knockdown caused a significant repression of cell viability, which was augmented by cotransfection of circ_0005556 and miR-767-5p (Figure 5B, ** P<0.01, ## P<0.01). In the flow cytometry analysis, we found that GC cell apoptosis was significantly increased in the circ_0005556 blocked group compared to the scrambled group (Figure 5C, ** P<0.01). Moreover, cotransfection of circ_0005556 and miR-767-5p resulted in a higher apoptosis rate than that from the si-circ_0005556 transfection alone (Figure 5C, ** P<0.01). In the wound-healing assay, the scratch gap was significantly wider in circ_0005556-silenced GC cells compared with scrambled GC cells, and cotransfection of circ_0005556 and miR-767-5p led to a wider scratch gap than that in the si-circ_0005556-transfected group, suggesting that the inhibitory impact of the circ_0005556 knockdown on GC cells could be further enhanced by miR-767-5p (Figure 5D, ** P<0.01, ## P<0.01). In addition, in the transwell assay, we found that the si-circ_0005556 transfection resulted in a remarkable repression of cell invasive ability, and cotransfection of circ_0005556 and miR-767-5p augmented the inhibitory effect of si-circ_0005556 on GC cell invasion (Figure 5E, ** P<0.01, ## P<0.01). These findings suggested that circ_0005556 knockdown repressed GC cell growth by increasing the level of miR-767-5p.
Discussion
CircRNAs are predominately generated by back-splicing of multiple exons or introns and can be categorized into exonic, intronic, and exon-intron circRNAs based on their exact components [17]. CircRNAs have been studied for several decades since their first identification, but have received little attention until recent years [18]. A growing number of studies have shown the involvement of circRNAs in the processes of various human diseases, especially in human cancer initiation and development [17,19]. With the huge development of bioinformatics analysis, circRNA microarray analysis, and next-generation sequencing technology, circRNA expression patterns in GC patients have been investigated in an increasing number of studies, and numerous dysregulated circRNAs have been identified [20,21]. A study found 440 dysregulated circRNAs (176 increased and 264 decreased) between normal and tumor tissues collected from 6 GC patients [22]. Moreover, a study revealed 343 differentially expressed circRNAs between the plasma samples of GC patients and healthy controls. When compared to GC tissue samples, a total of 17 dysregulated circRNAs were found [14]. Most of these screened circRNAs have not been further identified in GC.
In our study, we further screened and identified 6 underlying circRNAs with high differential expression levels in GC. Our results further verified that circ_0005556 and circ_0001013 were remarkably upregulated, while circ_0001190, circ_0003195 and circ_0001811 were dramatically downregulated in GC. However, the exact functional roles of these dysregulated circRNAs in GC remain undetermined. Circ_0005556 was previously found to be increased in GC by 2 studies: Tianwen Li et al. [14] and Shao Y et al. [13]. Therefore, in the present study, we aimed to investigate the functional impacts of circ_0005556 in GC. Consistent with their findings, we validated its upregulation in GC tissues and cells. In addition, we demonstrated that the high expression of circ_0005556 was related to poor prognosis and short 5-year survival. In the functional analysis, we also proved for the first time that the knockdown of circ_0005556 could repress GC cell viability, invasion, and migration and promote GC cell apoptosis. Therefore, we suggest that circ_0005556 might act as an oncogene of GC and that circ_0005556 might be a promising diagnostic and therapeutic RNA molecule of GC.
Many studies have confirmed that miRNAs can be involved in the biological processes of various cancers [23,24]. These miRNAs can affect cancer cell proliferation, apoptosis, metastasis, and invasion by targeting certain oncogenic or tumor suppressor genes [25,26]. In GC, some miRNAs have also been proven to be dysregulated and are closely related to the occurrence and development of GC [27,28]. miR-767-5p has also been proven to be involved in cancer progression [29]. However, the expression and functions of miR-767-5p in GC have not been reported thus far. In our study, our results revealed that miR-767-5p was expressed at low levels in GC, and its expression showed a negative correlation with circ_0005556 expression in GC. In addition, miR-767-5p could suppress cell proliferation, migration, and invasion and facilitate apoptosis of GC cells mediated by circ_0005556 knockdown, suggesting that miR-767-5p can enhance the suppressive effects of circ_0005556 knockdown on GC cells.
Mounting evidence has shown that circRNAs participate in regulating gene expression by serving as mRNA sponges that abolish the repressive effects of miRNA on its target mRNAs, splicing of target mRNA, or modulating RNA-binding proteins [30–32]. In our study, we found that circ_0005556 could be bound by multiple miRNAs; therefore, we selected miR-767-5p for further study. Recently, miR-767-5p was also reported to mediate the repressive effects of circ_SMARCA5 and circ_0000190 in myeloma [33,34]. For the molecular mechanism, we also confirmed that circ_0005556 could restrain the expression of miR-767-5p by targeting its binding. Therefore, we proved that miR-767-5p not only targeted circ_0005556, but also augmented the suppressive impacts of circ_0005556 in GC. Taken together, we provided evidence that the knockdown of circ_0005556 repressed GC cell progression through upregulating miR-767-5p.
Conclusions
In summary, for the first time, we demonstrated the upregulation of circ_0005556 and circ_0001013 and the downregulation of circ_0001190, circ_0003195 and circ_0001811 in 20 pairs of GC samples. Our findings suggested that the circ_0005556/miR-767-5p axis is involved in the tumorigenesis of GC, and this improves the knowledge of circRNAs in GC and will contribute to the development of novel diagnostic and therapeutic options against GC. However, the present study was relatively limited, and more work should be conducted in the future. First, we should further explore the effects of circ_0001013, circ_0001190, circ_0003195 and circ_0001811 on the functions of GC. Second, we should further certify more biological functions, including cell cycle, autophagy and tumor growth
Figures
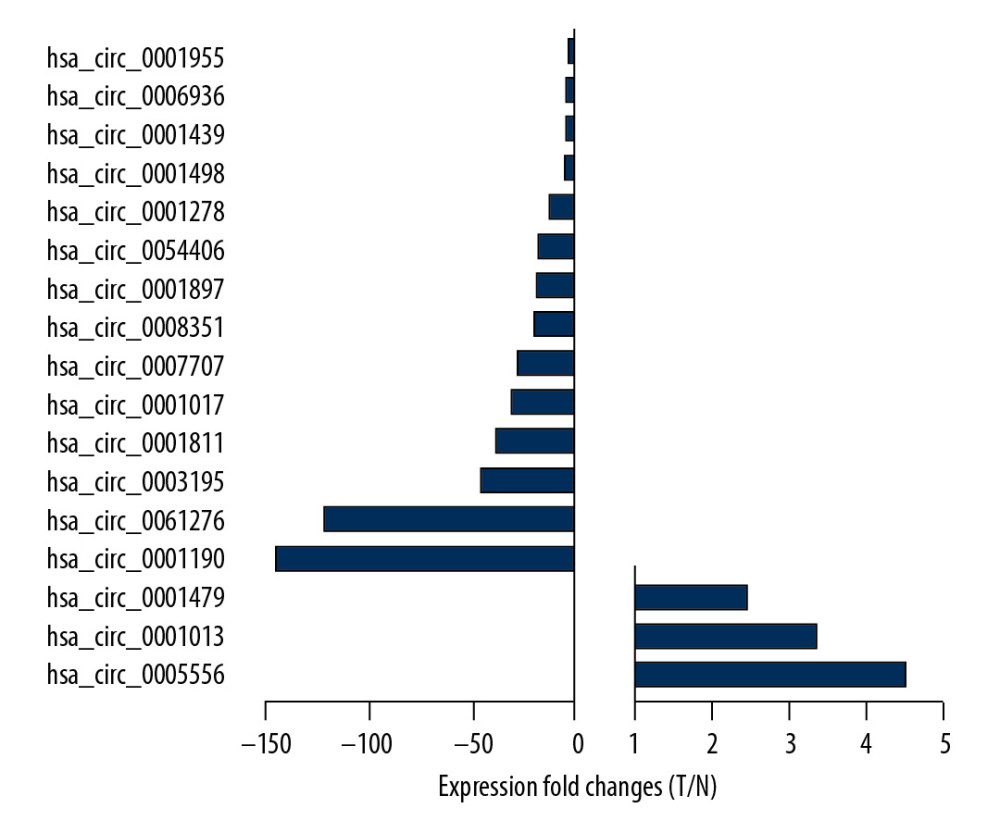 Figure 1. Expression pattern of circRNAs in GC patients’ plasma. Intersection analysis was conducted on GC tissue (GSE89143) and blood (GSE93541) datasets to detect differentially expressed circRNAs.
Figure 1. Expression pattern of circRNAs in GC patients’ plasma. Intersection analysis was conducted on GC tissue (GSE89143) and blood (GSE93541) datasets to detect differentially expressed circRNAs. 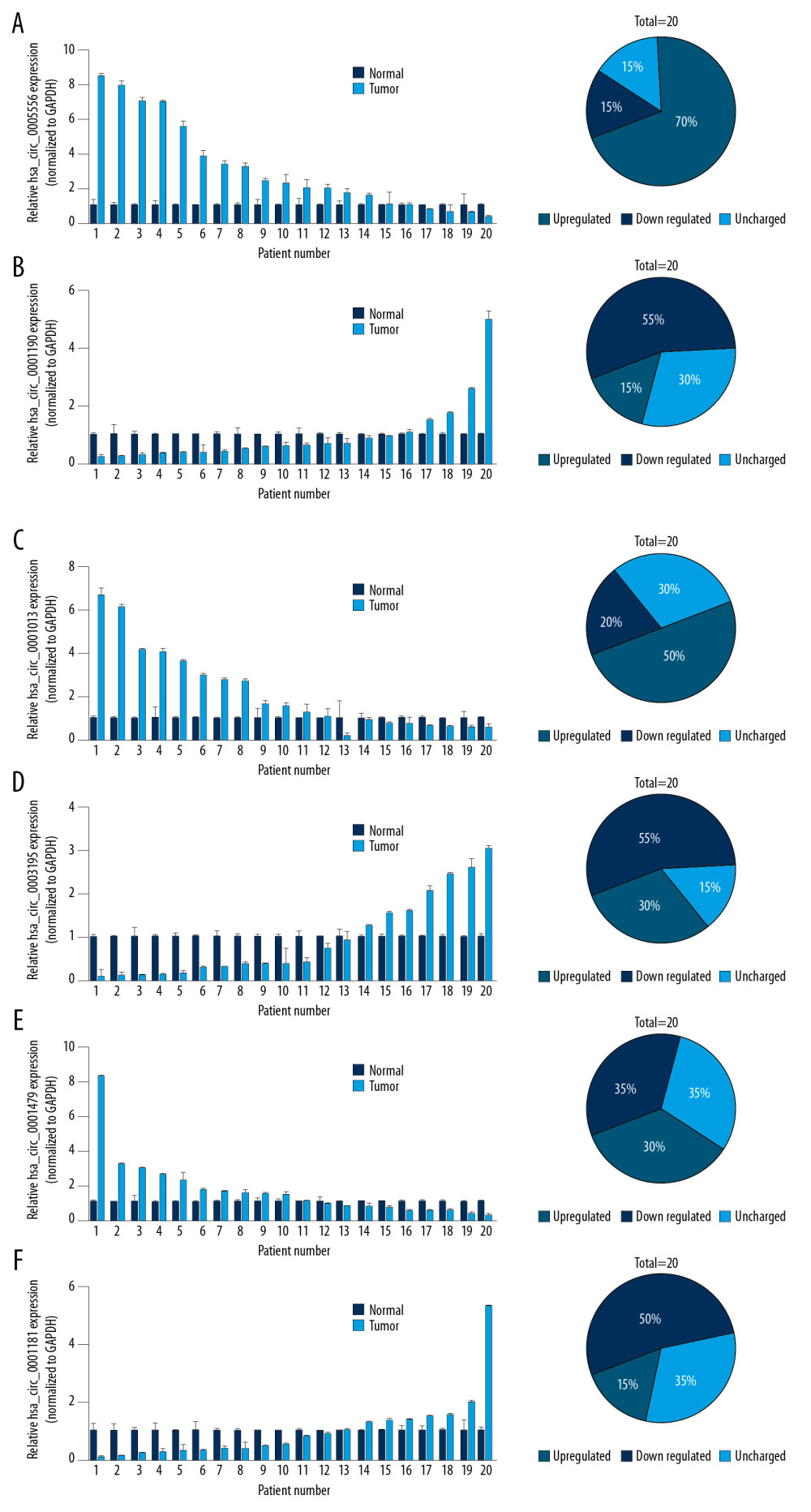 Figure 2. Validation of 6 dysregulated circRNAs in GC. qRT-PCR was used to confirm the dysregulation of (A) circ_0005556, (B) circ_0001190, (C) circ_0001013, (D) circ_0003195, (E) circ_0001479, and (F) circ_0001181 in 20 pairs GC and normal tissue samples.
Figure 2. Validation of 6 dysregulated circRNAs in GC. qRT-PCR was used to confirm the dysregulation of (A) circ_0005556, (B) circ_0001190, (C) circ_0001013, (D) circ_0003195, (E) circ_0001479, and (F) circ_0001181 in 20 pairs GC and normal tissue samples. 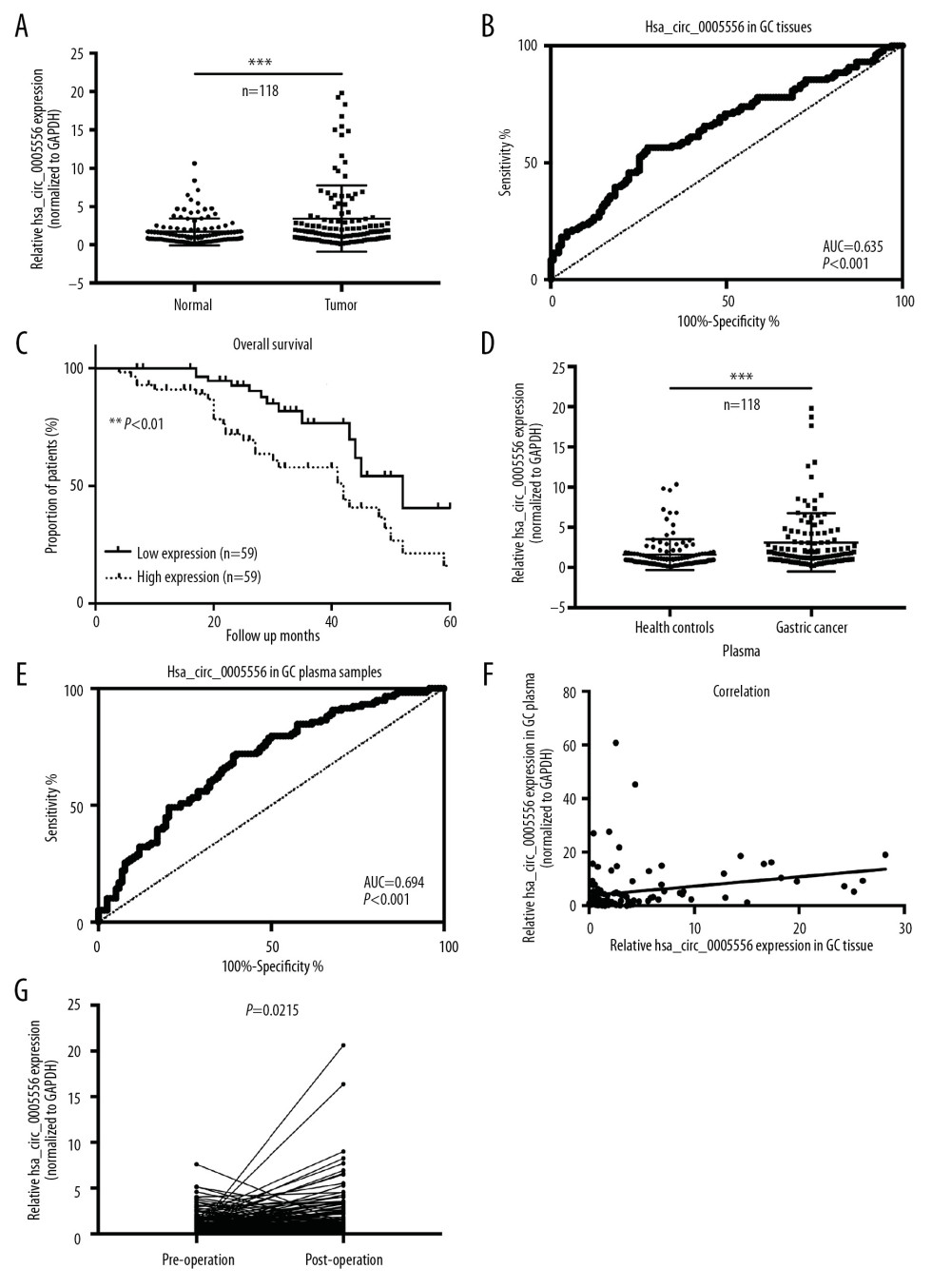 Figure 3. The circ_0005556 level was higher in GC and was correlated with a worse prognosis. (A) Circ_0005556 expression was tested by qRT-PCR in GC tissue and adjacent normal tissue specimens (*** P<0.001). (B) ROC analysis was carried out by using GC and normal tissues to explore the diagnostic value of circ_0005556. (C) Kaplan-Meier survival curve analysis showing the correlation between circ_0005556 level and prognosis in GC patients (** P<0.01). (D) Level of circ_0005556 was detected in the plasma samples from GC patients and healthy controls (*** P<0.001). (E) ROC analysis of circ_0005556 in the plasma samples of GC patients. (F) Correlation analysis between the GC tissue and plasma circ_0005556 levels (R2=0.0576, P=0.0089). (G) Circ_0005556 was examined in preoperative and postoperative GC patients (P=0.0215).
Figure 3. The circ_0005556 level was higher in GC and was correlated with a worse prognosis. (A) Circ_0005556 expression was tested by qRT-PCR in GC tissue and adjacent normal tissue specimens (*** P<0.001). (B) ROC analysis was carried out by using GC and normal tissues to explore the diagnostic value of circ_0005556. (C) Kaplan-Meier survival curve analysis showing the correlation between circ_0005556 level and prognosis in GC patients (** P<0.01). (D) Level of circ_0005556 was detected in the plasma samples from GC patients and healthy controls (*** P<0.001). (E) ROC analysis of circ_0005556 in the plasma samples of GC patients. (F) Correlation analysis between the GC tissue and plasma circ_0005556 levels (R2=0.0576, P=0.0089). (G) Circ_0005556 was examined in preoperative and postoperative GC patients (P=0.0215). 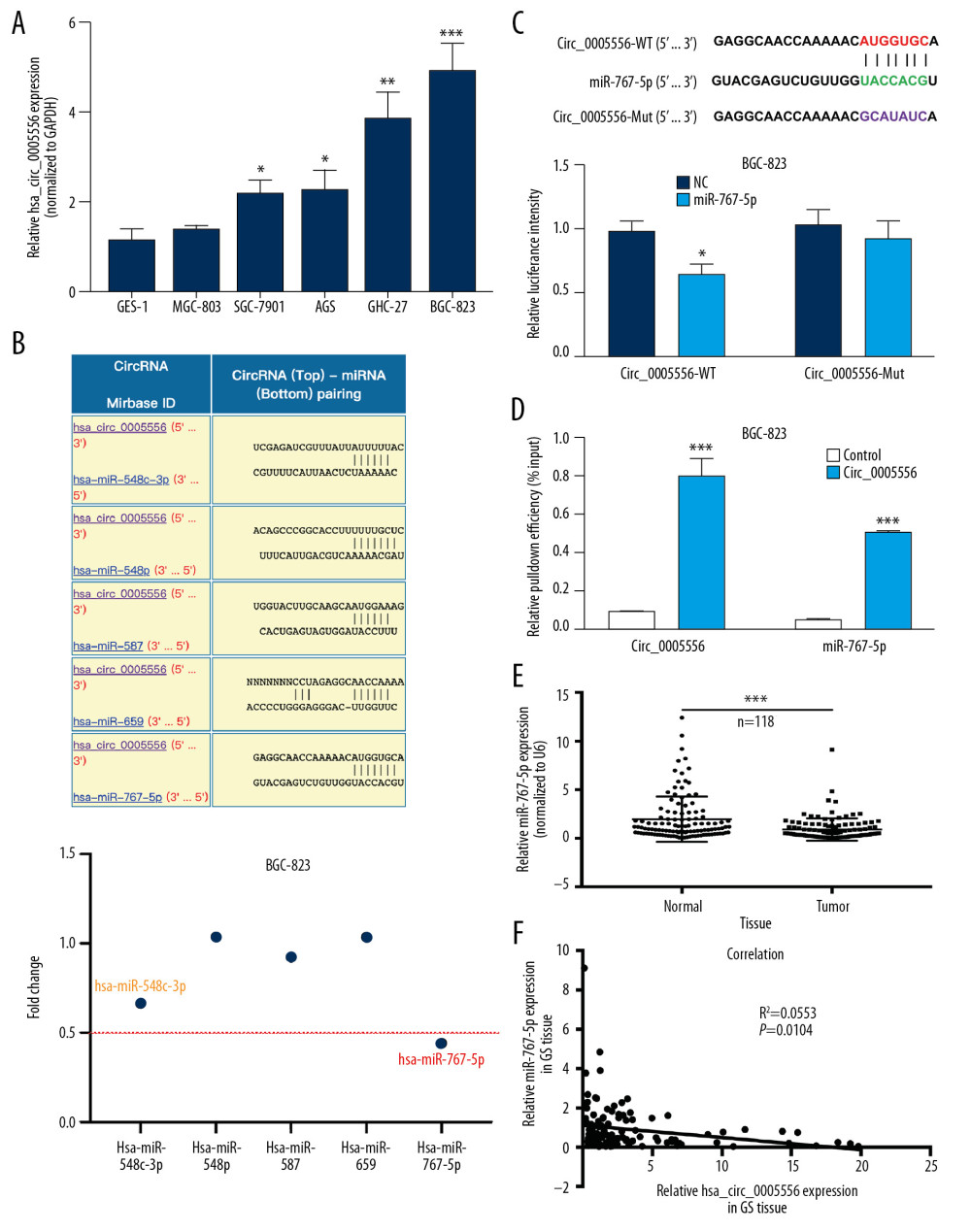 Figure 4. Circ_0005556 was bound by miR-767-5p. (A) Circ_0005556 levels were measured in GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823), (* P<0.05, ** P<0.01, *** P<0.001 vs. GES-1 group). (B) Target miRNAs of circ_0005556 were predicted by bioinformatics method, and the relative fold change of these target miRNAs in BGC-823 was detected. The physical interplay between circ_0005556 and miR-767-5p was validated by (C) a dual-luciferase reporter experiment and (D) a circRNA pulldown assay in BGC-823 cells (* P<0.05, *** P<0.001). (E) The relative expression level of miR-767-5p was investigated by qRT-PCR in 118 pairs of GC and normal tissue samples (*** P<0.001). (F) Correlation analysis between circ_0005556 level and miR-767-5p level in GC tissue specimens (P=0.0104).
Figure 4. Circ_0005556 was bound by miR-767-5p. (A) Circ_0005556 levels were measured in GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823), (* P<0.05, ** P<0.01, *** P<0.001 vs. GES-1 group). (B) Target miRNAs of circ_0005556 were predicted by bioinformatics method, and the relative fold change of these target miRNAs in BGC-823 was detected. The physical interplay between circ_0005556 and miR-767-5p was validated by (C) a dual-luciferase reporter experiment and (D) a circRNA pulldown assay in BGC-823 cells (* P<0.05, *** P<0.001). (E) The relative expression level of miR-767-5p was investigated by qRT-PCR in 118 pairs of GC and normal tissue samples (*** P<0.001). (F) Correlation analysis between circ_0005556 level and miR-767-5p level in GC tissue specimens (P=0.0104). 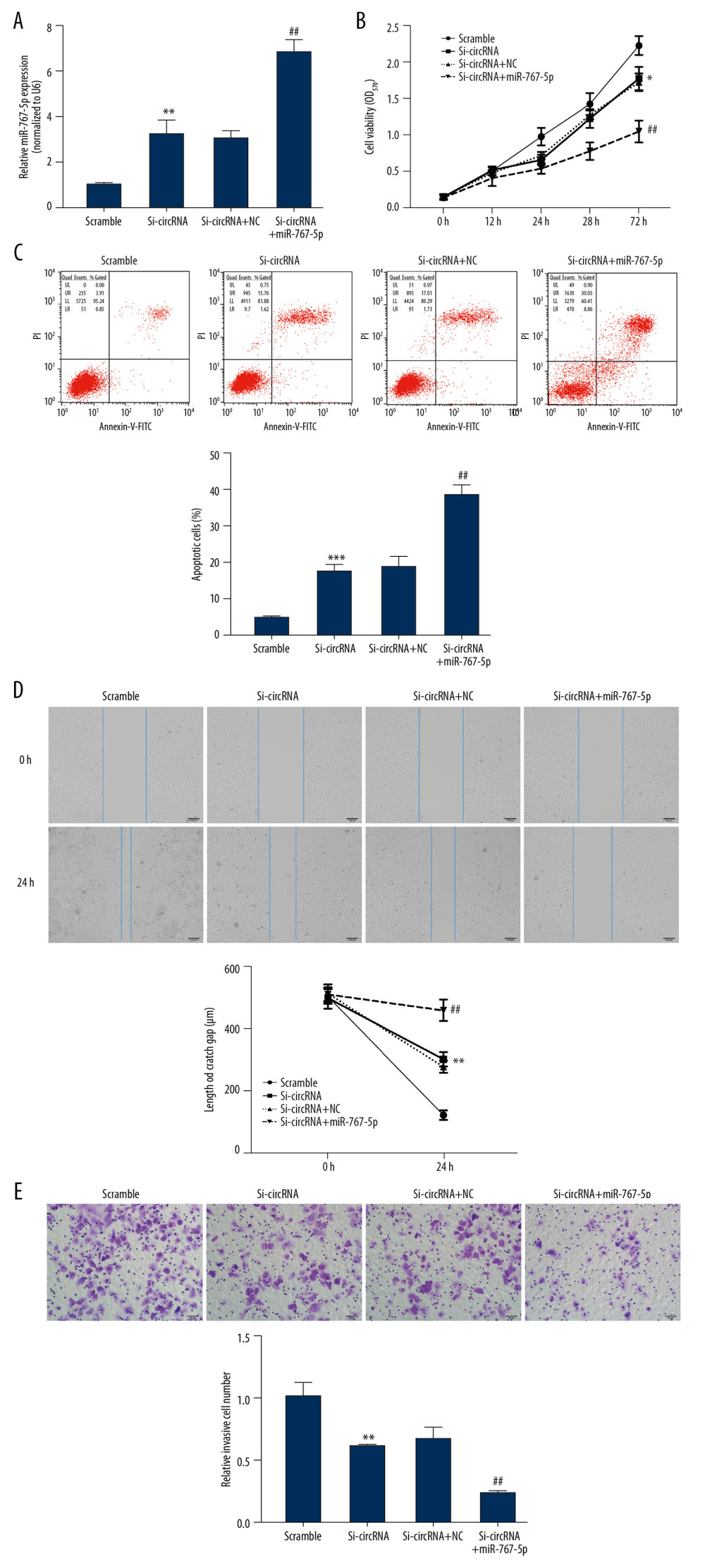 Figure 5. miR-767-5p transfection enhanced the inhibitory impacts of circ_0005556 knockdown on GC cells. (A) Expression of miR-767-5p was analyzed by qRT-PCR in GC cells transfected with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). (B) GC cell viability, (C) apoptosis, (D) migration, and (E) invasion were estimated by MTT, flow cytometry, wound-healing, and transwell assays, respectively, after transfection with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group).
Figure 5. miR-767-5p transfection enhanced the inhibitory impacts of circ_0005556 knockdown on GC cells. (A) Expression of miR-767-5p was analyzed by qRT-PCR in GC cells transfected with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). (B) GC cell viability, (C) apoptosis, (D) migration, and (E) invasion were estimated by MTT, flow cytometry, wound-healing, and transwell assays, respectively, after transfection with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). References
1. Karimi P, Islami F, Anandasabapathy S, Gastric cancer: Descriptive epidemiology, risk factors, screening, and prevention: Cancer Epidemiol Biomarkers Prev, 2014; 23(5); 700-13
2. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2019: Cancer J Clin, 2019; 69(1); 7-34
3. Allemani C, Weir HK, Carreira H, Global surveillance of cancer survival 1995–2009: Analysis of individual data for 25,676,887 patients from 279 population-based registries in 67 countries (CONCORD-2): Lancet, 2015; 385(9972); 977-1010
4. Li Q, Shao Y, Zhang X, Plasma long noncoding RNA protected by exosomes as a potential stable biomarker for gastric cancer: Tumour Biol, 2015; 36(3); 2007-12
5. Cui L, Lou Y, Zhang X, Detection of circulating tumor cells in peripheral blood from patients with gastric cancer using piRNAs as markers: Clin Biochem, 2011; 44(13); 1050-57
6. Bar-Zeev M, Nativ L, Assaraf YG, Livney YD, Re-assembled casein micelles for oral delivery of chemotherapeutic combinations to overcome multidrug resistance in gastric cancer: Journal of Molecular and Clinical Medicine, 2018; 1(1); 55-65
7. Schneider J, Schulze G, Comparison of tumor M2-pyruvate kinase (tumor M2-PK), carcinoembryonic antigen (CEA), carbohydrate antigens CA 19-9 and CA 72-4 in the diagnosis of gastrointestinal cancer: Anticancer Res, 2003; 23(6D); 5089-93
8. Belousova EA, Filipenko ML, Kushlinskii NE, Circular RNA: New regulatory molecules: Bull Exp Biol Med, 2018; 164(6); 803-15
9. Eger N, Schoppe L, Schuster S, Circular RNA splicing: Adv Exp Med Biol, 2018; 1087; 41-52
10. Hsiao KY, Sun HS, Tsai SJ, Circular RNA – New member of noncoding RNA with novel functions: Exp Biol Med (Maywood), 2017; 242(11); 1136-41
11. Lei B, Tian Z, Fan W, Ni B, Circular RNA: A novel biomarker and therapeutic target for human cancers: Int J Med Sci, 2019; 16(2); 292-301
12. Chen B, Huang S, Circular RNA: An emerging non-coding RNA as a regulator and biomarker in cancer: Cancer Lett, 2018; 418; 41-50
13. Shao Y, Li J, Lu R, Global circular RNA expression profile of human gastric cancer and its clinical significance: Cancer Med, 2017; 6(6); 1173-80
14. Li T, Shao Y, Fu L, Plasma circular RNA profiling of patients with gastric cancer and their droplet digital RT-PCR detection: J Mol Med (Berl), 2018; 96(1); 85-96
15. Livak KJ, Schmittgen TD, Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method: Methods, 2001; 25(4); 402-8
16. Shan D, Shang Y, Hu T, Long noncoding RNA BLACAT1 promotes cell proliferation and invasion in human cervical cancer: Oncol Lett, 2018; 15(3); 3490-95
17. Yin Y, Long J, He Q, Emerging roles of circRNA in formation and progression of cancer: J Cancer, 2019; 10(21); 5015-21
18. Mei M, Wang Y, Li Z, Zhang M, Role of circular RNA in hematological malignancies: Oncol Lett, 2019; 18(5); 4385-92
19. Yu T, Wang Y, Fan Y, CircRNAs in cancer metabolism: A review: J Hematol Oncol, 2019; 12(1); 90
20. Shan C, Zhang Y, Hao X, Biogenesis, functions and clinical significance of circRNAs in gastric cancer: Mol Cancer, 2019; 18(1); 136
21. Zhang M, Du X, Noncoding RNAs in gastric cancer: Research progress and prospects: World J Gastroenterol, 2016; 22(29); 6610-18
22. Gu W, Sun Y, Zheng X, Identification of gastric cancer-related circular RNA through microarray analysis and bioinformatics analysis: Biomed Res Int, 2018; 2018 2381680
23. Qadir MI, Faheem A, miRNA: A diagnostic and therapeutic tool for pancreatic cancer: Crit Rev Eukaryot Gene Expr, 2017; 27(3); 197-204
24. Yonemori K, Kurahara H, Maemura K, Natsugoe S, MicroRNA in pancreatic cancer: J Hum Genet, 2017; 62(1); 33-40
25. Mishra S, Yadav T, Rani V, Exploring miRNA-based approaches in cancer diagnostics and therapeutics: Crit Rev Oncol Hematol, 2016; 98; 12-23
26. Rupaimoole R, Slack FJ, MicroRNA therapeutics: towards a new era for the management of cancer and other diseases: Nat Rev Drug Discov, 2017; 16(3); 203-22
27. Alessandrini L, Manchi M, De Re V, Proposed molecular and miRNA classification of gastric cancer: Int J Mol Sci, 2018; 19(6); 1683
28. Mao Y, Liu R, Zhou H, Transcriptome analysis of miRNA-lncRNA-mRNA interactions in the malignant transformation process of gastric cancer initiation: Cancer Gene Ther, 2017; 24(6); 267-75
29. Zhang J, Xu S, Xu J, miR7675p inhibits glioma proliferation and metastasis by targeting SUZ12: Oncol Rep, 2019; 42(1); 55-66
30. Hansen TB, Jensen TI, Clausen BH, Natural RNA circles function as efficient microRNA sponges: Nature, 2013; 495(7441); 384-88
31. Ashwal-Fluss R, Meyer M, Pamudurti NR, circRNA biogenesis competes with pre-mRNA splicing: Mol Cell, 2014; 56(1); 55-66
32. Zang J, Lu D, Xu A, The interaction of circRNAs and RNA binding proteins: An important part of circRNA maintenance and function: J Neurosci Res, 2020; 98(1); 87-97
33. Liu H, Wu Y, Wang S, Circ-SMARCA5 suppresses progression of multiple myeloma by targeting miR-767-5p: BMC Cancer, 2019; 19(1); 937
34. Feng Y, Zhang L, Wu J, CircRNA circ_0000190 inhibits the progression of multiple myeloma through modulating miR-767-5p/MAPK4 pathway: J Exp Clin Cancer Res, 2019; 38(1); 54
Figures
 Figure 1. Expression pattern of circRNAs in GC patients’ plasma. Intersection analysis was conducted on GC tissue (GSE89143) and blood (GSE93541) datasets to detect differentially expressed circRNAs.
Figure 1. Expression pattern of circRNAs in GC patients’ plasma. Intersection analysis was conducted on GC tissue (GSE89143) and blood (GSE93541) datasets to detect differentially expressed circRNAs. Figure 2. Validation of 6 dysregulated circRNAs in GC. qRT-PCR was used to confirm the dysregulation of (A) circ_0005556, (B) circ_0001190, (C) circ_0001013, (D) circ_0003195, (E) circ_0001479, and (F) circ_0001181 in 20 pairs GC and normal tissue samples.
Figure 2. Validation of 6 dysregulated circRNAs in GC. qRT-PCR was used to confirm the dysregulation of (A) circ_0005556, (B) circ_0001190, (C) circ_0001013, (D) circ_0003195, (E) circ_0001479, and (F) circ_0001181 in 20 pairs GC and normal tissue samples. Figure 3. The circ_0005556 level was higher in GC and was correlated with a worse prognosis. (A) Circ_0005556 expression was tested by qRT-PCR in GC tissue and adjacent normal tissue specimens (*** P<0.001). (B) ROC analysis was carried out by using GC and normal tissues to explore the diagnostic value of circ_0005556. (C) Kaplan-Meier survival curve analysis showing the correlation between circ_0005556 level and prognosis in GC patients (** P<0.01). (D) Level of circ_0005556 was detected in the plasma samples from GC patients and healthy controls (*** P<0.001). (E) ROC analysis of circ_0005556 in the plasma samples of GC patients. (F) Correlation analysis between the GC tissue and plasma circ_0005556 levels (R2=0.0576, P=0.0089). (G) Circ_0005556 was examined in preoperative and postoperative GC patients (P=0.0215).
Figure 3. The circ_0005556 level was higher in GC and was correlated with a worse prognosis. (A) Circ_0005556 expression was tested by qRT-PCR in GC tissue and adjacent normal tissue specimens (*** P<0.001). (B) ROC analysis was carried out by using GC and normal tissues to explore the diagnostic value of circ_0005556. (C) Kaplan-Meier survival curve analysis showing the correlation between circ_0005556 level and prognosis in GC patients (** P<0.01). (D) Level of circ_0005556 was detected in the plasma samples from GC patients and healthy controls (*** P<0.001). (E) ROC analysis of circ_0005556 in the plasma samples of GC patients. (F) Correlation analysis between the GC tissue and plasma circ_0005556 levels (R2=0.0576, P=0.0089). (G) Circ_0005556 was examined in preoperative and postoperative GC patients (P=0.0215). Figure 4. Circ_0005556 was bound by miR-767-5p. (A) Circ_0005556 levels were measured in GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823), (* P<0.05, ** P<0.01, *** P<0.001 vs. GES-1 group). (B) Target miRNAs of circ_0005556 were predicted by bioinformatics method, and the relative fold change of these target miRNAs in BGC-823 was detected. The physical interplay between circ_0005556 and miR-767-5p was validated by (C) a dual-luciferase reporter experiment and (D) a circRNA pulldown assay in BGC-823 cells (* P<0.05, *** P<0.001). (E) The relative expression level of miR-767-5p was investigated by qRT-PCR in 118 pairs of GC and normal tissue samples (*** P<0.001). (F) Correlation analysis between circ_0005556 level and miR-767-5p level in GC tissue specimens (P=0.0104).
Figure 4. Circ_0005556 was bound by miR-767-5p. (A) Circ_0005556 levels were measured in GC cell lines (MGC-803, SGC-7901, AGS, HGC-27, and BGC-823), (* P<0.05, ** P<0.01, *** P<0.001 vs. GES-1 group). (B) Target miRNAs of circ_0005556 were predicted by bioinformatics method, and the relative fold change of these target miRNAs in BGC-823 was detected. The physical interplay between circ_0005556 and miR-767-5p was validated by (C) a dual-luciferase reporter experiment and (D) a circRNA pulldown assay in BGC-823 cells (* P<0.05, *** P<0.001). (E) The relative expression level of miR-767-5p was investigated by qRT-PCR in 118 pairs of GC and normal tissue samples (*** P<0.001). (F) Correlation analysis between circ_0005556 level and miR-767-5p level in GC tissue specimens (P=0.0104). Figure 5. miR-767-5p transfection enhanced the inhibitory impacts of circ_0005556 knockdown on GC cells. (A) Expression of miR-767-5p was analyzed by qRT-PCR in GC cells transfected with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). (B) GC cell viability, (C) apoptosis, (D) migration, and (E) invasion were estimated by MTT, flow cytometry, wound-healing, and transwell assays, respectively, after transfection with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group).
Figure 5. miR-767-5p transfection enhanced the inhibitory impacts of circ_0005556 knockdown on GC cells. (A) Expression of miR-767-5p was analyzed by qRT-PCR in GC cells transfected with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). (B) GC cell viability, (C) apoptosis, (D) migration, and (E) invasion were estimated by MTT, flow cytometry, wound-healing, and transwell assays, respectively, after transfection with si-circ_0005556 or miR-767-5p (** P<0.01 vs. scrambled group, ## P<0.01 vs. si-circRNA+NC group). In Press
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952