03 August 2020: Review Articles
Galectins for Diagnosis and Prognostic Assessment of Human Diseases: An Overview of Meta-Analyses
Yiting Liu12ABCDEF, Hao Meng13ABCDEF, Shixue Xu1ABCDEF, Xingshun Qi1ABCDEF*DOI: 10.12659/MSM.923901
Med Sci Monit 2020; 26:e923901
Abstract
ABSTRACT: An increasing number of studies have explored the activities and functions of galectins. However, translation of these researches into clinical practice seems to be lacking. As compared to scattered individual studies, meta-analyses can provide a more comprehensive review of current evidence and reach a more unbiased and powered conclusion by synthesizing data from diverse studies. In this paper, findings from meta-analyses were reviewed to establish the role of galectins in diagnosis and prognostic assessment of various human diseases. First, in patients with cancer, galectin-1 expression is often associated with poorer survival, but galectin-9 expression is associated with better survival. Galectin-3 is a diagnostic biomarker for thyroid cancer and a predictor of worse survival in patients with colorectal cancer and improved survival in patients with gastric cancer. Second, galectin-3 is useful for diagnosis and prognostic assessment of heart failure and prediction of atrial fibrillation and its recurrence. Third, in chronic kidney disease, galectin-3 is valuable for predicting poor survival. Fourth, during pregnancy, galectin-13 is potentially helpful for identifying patients who do not have preeclampsia.
Keywords: Clinical Alarms, Early Detection of Cancer, Evidence-Based Practice, Galectins, Atrial Fibrillation, Evidence-Based Medicine, Neoplasms, Odds Ratio, Pre-Eclampsia, Pregnancy, Renal Insufficiency, Chronic
Background
In 1994, galectins were recognized as a distinct family of beta-galactoside-binding lectins [1]. Today, at least 15 members of the galectin family have been identified, all of which contain carbohydrate-recognition domains (CRDs) in charge of carbohydrate binding [2]. Galectins are classified into three main groups according to their structural features: 1) prototype galectins, which have a single CRD, and include galectins −1, 2, 5, 7, 10, 11, 13, and 14; 2) tandem repeat galectins, which have two distinct CRDs, and include galectins 4, 6, 8, 9, and 12; and 3) chimeric galectins, which have tandem repeats of proline- and glycine-rich short stretches fused onto the CRD, and include galectin-3.
Galectin family members have their respective biological behaviors and functions. Experimental studies have shown that galectins function both intracellularly and extracellularly, and are involved in modulation of cell activation, proliferation, apoptosis, adhesion, migration, and inflammation [3–5]. In addition, clinical evidence has demonstrated the importance of galectins in diagnosis and prognostic assessment of cancer [6], cardiovascular disease (CVD) [7], nephropathies [8], skin diseases [9], and diet-induced steatohepatitis [10].
Meta-analysis, through which data are analyzed after systematic collection from relevant studies, can avert potential reporting bias from a single small sample study, and therefore, may reach a more reliable conclusion [11]. Herein, we have summarized current findings of meta-analyses on clinical use of galectins in various human diseases. This work is potentially worthwhile for researchers to clarify evidence that has been widely published about galectins which could potentially be translated into clinical practice and to identify issues about galectins that have been insufficiently and rarely explored.
Material and Methods
First, we systematically searched the PubMed database on April, 2020 on the terms “(galectin) AND (meta-analysis)”. All eligible papers reported results of meta-analyses regarding the role of galectins in clinical diagnosis and assessment of any human disease. Narrative reviews, experiment studies, and single original studies were excluded. Initially, 41 papers were identified, and 25 meta-analyses were finally included. The role of galectins has been studied in meta-analyses only in cancer, CVD, kidney disease, and pregnancy (Figure 1).
Second, we reviewed all eligible papers and recorded which member(s) of the galectin family was/were explored, which type(s) of diseases was/were evaluated, and which outcome(s) of interest was/were analyzed. Overall survival (OS), cancer-specific survival (CSS), relapse-free survival/progression-free survival (RFS/PFS), and disease-free survival/recurrence-free survival (DFS/RFS) are the main parameters for assessing cancer prognosis and tumor size, stage, and metastasis are the main parameters for assessing the clinicopathological characteristics of cancer.
We also reviewed the effect sizes reported in all included studies. Hazard ratio (HR), odds ratio (OR), risk ratio (RR), and mean difference (MD) were employed to evaluate the association of galectins with survival and clinicopathological data of diseases; and area under the curve (AUC), diagnostic odds ratio (DOR), sensitivity, and specificity were employed to evaluate the accuracy of galectins in diagnosis and prognostic assessment of diseases.
Finally, we reviewed the main findings and conclusions. If two or more meta-analyses focused on the same scientific question, but had different findings, we compared the difference in the number of included papers and quality of the included studies, and also reevaluated the accuracy of statistical methods employed by each meta-analysis, if necessary.
Cancer
VARIOUS CANCERS:
Two different study groups conducted similar meta-analyses to explore the relationship between galectin-1 expression and prognosis in patients with various cancers. Wu R et al. systematically identified 18 studies with 2674 patients, and found that high galectin-1 expression should predict an increased risk of mortality [12]. More recently, Huang M et al. also performed a meta-analysis of 29 studies with 3543 patients, and similarly showed a statistically significant association of high galectin-1 expression with poorer OS (HR=2.12), DFS (HR=1.60), CSS (HR=1.82), and PFS (HR=1.93) [13]. These findings should be cautiously interpreted, because they were produced for unclassified cancer, but not specific to a particular type of cancer.
SOLID TUMORS:
Two different study groups conducted similar meta-analyses in 2018 to explore the association of galectin-9 expression with prognosis in patients with solid tumors [14,15]. However, their conclusions were a bit different. Wang K et al. included 14 studies with 2408 patients up to June 2017, and demonstrated no significant association of high galectin-9 expression with OS (HR=0.80, P=0.311) or RFS/PFS (HR=0.58, P=0.097) in spite of its significant association with better CSS (HR=0.48, P<0.001) [14]. By contrast, Zhou X et al. included 14 studies with 2326 patients up to October 2017, and reported a statistically significant association of high galectin-9 expression with better OS (HR=0.70, P=0.006), rather than DFS/RFS (HR=0.85, P=0.527) [15]. This difference between the two meta-analyses should be discussed. Although the meta-analysis by Wang K et al. included fewer studies than that by Zhou X et al. to explore the association of galectin-9 expression with OS (7 versus 12) [14,15], a further review of studies included in the two meta-analyses suggested that the meta-analysis by Wang K et al., rather than that by Zhou X et al., clearly distinguished CSS from OS. Considering a potential discrepancy between the two outcomes of interests, a conservative conclusion drawn by Wang K et al. should be more reasonable.
Zhou X et al. also reported a significant association of high galectin-9 expression with better clinicopathological characteristics of solid tumors, including less invasion and absence of lymph node or distal tumor metastasis [15].
Wang Y et al. also evaluated the association of galectin-3 with outcomes of solid tumors, and found a statistically significant association of high galectin-3 expression with poorer OS (HR=1.79) and DFS/PFS/RFS (HR=1.57) [16].
THYROID CANCER:
Four different study groups performed similar meta-analyses to evaluate the role of galectin-3 in diagnosis of thyroid cancer [17–20]. According to the effect sizes, meta-analyses by de Matos L et al. [17], Xin Y et al. [18], and Trimboli P et al. [19] calculated the sensitivity and specificity to evaluate the diagnostic performance of galectin-3, but the meta-analysis by Tang W et al. calculated the OR to compare the difference in positive galectin-3 expression rate between patients with and without thyroid cancer [20]. Regardless, all of them supported the diagnostic value of galectin-3. Notably, Xin Y et al. also explored the role of Hector Battifora mesothelial antigen-1 in the diagnosis of thyroid cancer and suggested that the AUC of Hector Battifora mesothelial antigen-1 should be larger than that of galectin-3.
Tang W et al. reported a higher rate of positive galectin-3 expression in patients with thyroid cancer who had lymph node metastasis than those without lymph node metastasis [20].
PANCREATIC CANCER:
A meta-analysis by Sun Q et al. included 11 studies to explore the role of galectins in diagnosis and prognostic assessment of pancreatic cancer [21]. They found that galectin-3 had diagnostic value for pancreatic cancer. In addition, high galectin-1 expression and low galectin-4 and galectin-9 expression were significantly associated with poorer OS in pancreatic cancer, but galectin-3 expression was not significantly associated with OS or clinicopathological characteristics.
GASTRIC CANCER:
Long et al. systematically identified eight studies involving 2093 patients with gastric cancer [22]. Among them, two studies explored the association between galectin-1 expression and OS, and a meta-analysis further suggested a statistically significant relationship between high galectin-1 expression and poorer OS (HR=1.85, P<0.001). By comparison, four, one, and one studies explored the association of galectin-3, -8, and -9 expressions with OS, respectively; and there were statistically significant associations of high galectin-3 (HR=0.49, P<0.001), galectin-8 (HR=0.35, P<0.001), and galectin-9 (HR=0.78, P=0.003) expressions with better OS.
The meta-analysis also found a significant correlation of high galectin-1 expression with worse clinicopathological characteristics, including larger tumor size, higher TNM stage, presence of lymphatic vessel invasion, and poorer tumor differentiation.
COLORECTAL CANCER:
A meta-analysis by Wang C et al. included 15 studies with 1661 patients to explore the association of galectin-3 with OS and clinicopathological features of colorectal cancer [23]. The meta-analysis found statistically significant associations of galectin-3 expression with poorer OS (HR=1.77, 95%CI=1.36–2.31, P<0.0001) and worse clinicopathological features, including higher tumor stage and venous invasion.
Cardiovascular Disease
HEART FAILURE:
In terms of diagnosis, Huang Z et al. identified eight studies and conducted a meta-analysis on diagnostic value of galectin-3 for heart failure [24]. The pooled AUC, DOR, sensitivity, and specificity of galectin-3 for diagnosis of heart failure were 0.89, 18.29, 81%, and 63%, respectively.
In terms of prognostic assessment, three different study groups performed similar meta-analyses to evaluate the role of galectin-3 for assessing outcomes of heart failure [25–27]. But they employed different statistical methods and calculated different effect sizes. In 2015, Chen A et al. included 11 studies and found a positive association of galectin-3 with all-cause mortality by using combining categorical (HR=1.30) and continuous (HR=1.28) data [25]. They also found a statistically significant association of galectin-3 with cardiovascular mortality (HR=1.59). In 2016, Chen Y et al. conducted meta-analyses in chronic heart failure (9 studies) and acute heart failure (4 studies) [26]. The pooled AUC, DOR, sensitivity, and specificity of galectin-3 for predicting all-cause death were 0.64, 2.36, 60%, and 61% in chronic heart failure and 0.64, 2.30, 64%, and 57% in acute heart failure, respectively. In 2017, Imran T et al. found a statistically significant association of elevated plasma galectin-3 level with higher risk of all-cause death (HR=1.09) in nine studies and CVD (HR=1.44) in five studies [27]. Taken together, all three meta-analyses indicated that galectin-3 negatively influenced outcomes of heart failure, but its ability might be relatively weak.
ATRIAL FIBRILLATION:
A meta-analysis by Gong M et al. explored the relationship of galectin-3 with risk of atrial fibrillation [28]. They found a significantly higher galectin-3 concentration in patients with atrial fibrillation than those without (MD=−0.268ng/mL) and a significantly higher galectin-3 concentration in patients with persistent atrial fibrillation than those with paroxysmal atrial fibrillation (MD=−0.94ng/mL).
Three different study groups performed similar meta-analyses to evaluate the relationship of galectin-3 with recurrence of atrial fibrillation [28–30]. Two of them found that patients with recurrence of atrial fibrillation had a significantly higher galectin-3 concentration than those without [28,29], but another did not find such a statistically significant difference in galectin-3 concentration between patients with and without recurrence of atrial fibrillation [30]. By comparison, all of them showed that a higher galectin-3 concentration was significantly related to recurrence of atrial fibrillation.
CORONARY ARTERY DISEASE:
A meta-analysis by Li W et al. identified seven studies involving 10 552 cases and 10 545 controls to evaluate the association of galectin-2-encoding gene (LGALS2) with risk of coronary artery disease [31]. But there was no significant association between them.
ISCHEMIC STROKE:
Misra S et al. conducted a meta-analysis of two case-control studies to evaluate the association of LGALS2 with risk of ischemic stroke [32]. But there was no significant association between them.
Chronic Kidney Disease
A meta-analysis by Zhang T et al. included five studies with 5 226 patients and evaluated the association of galectin-3 with outcomes of chronic kidney diseases (CKD) [33]. They found that galectin-3 level positively influenced risks of all-cause death (HR=1.379) and cardiovascular events (HR=1.054).
Pregnancy
Three meta-analyses evaluated the diagnostic value of galectins alone or in combination with pulsatility index for preeclampsia (Table 3) [34–36].
Two different study groups published similar meta-analyses in 2015 to explore the diagnostic accuracy of galectin-13 alone for preeclampsia [34,35]. Wu P et al. identified nine studies exploring the role of galectin-13, also known as placental protein 13, for diagnosis of preeclampsia [34]. The AUC was 0.882, sensitivity 37%, and specificity 88%. In another meta-analysis by Zhong Y et al., the sensitivity and specificity of galectin-13 were 47% and 89% for early preeclampsia, 60% and 78% for early preeclampsia, 36% and 90% for small for gestational age, and 51% and 88% for preterm delivery, respectively [35]. Both meta-analyses suggested a high specificity (i.e., true negative rate) for galectin-13, but low sensitivity (i.e., true positive rate).
Zhu X et al. evaluated diagnostic performance of galectin-13 in combination with uterine artery pulsatility index for all, early, and late preeclampsia [36]. The sensitivities were 69%, 77%, and 54%, respectively. These findings suggested that galectin-13 in combination with uterine artery pulsatility index had a relatively good diagnostic ability for early preeclampsia, but not for late pre-eclampsia.
Conclusions
The current work, which reviews well-established evidence from meta-analyses, is valuable for investigators to avoid duplicate studies, guide clinical translation of research, and explore untouched issues associated with galectins in future. Several major findings regarding the clinical usefulness of galectins in various human diseases can be summarized as follows.
Notably, the clinical importance of other members of the galectin family in these diseases has not been fully studied by meta-analyses. In addition, clinical use of galectins in other human diseases needs to be further explored.
References
1. Barondes SH, Castronovo V, Galectins: aA family of animal beta-galactoside-binding lectins: Cell, 1994; 76; 597-98
2. Liu FT, Rabinovich GA, Galectins as modulators of tumour progression: Nat Rev Cancer, 2005; 5; 29-41
3. Gilson RC, Gunasinghe SD, Johannes L, Gaus K, Galectin-3 modulation of T-cell activation: mechanisms of membrane remodelling: Prog Lipid Res, 2019; 76; 101010
4. Seyrek K, Richter M, Lavrik IN, Decoding the sweet regulation of apoptosis: The role of glycosylation and galectins in apoptotic signaling pathways: Cell Death Differ, 2019; 26; 981-93
5. Hughes RC, Galectins as modulators of cell adhesion: Biochimie, 2001; 83; 667-76
6. Thijssen VL, Heusschen R, Caers J, Griffioen AW, Galectin expression in cancer diagnosis and prognosis: A systematic review: Biochim Biophys Acta, 2015; 1855; 235-47
7. Gehlken C, Suthahar N, Meijers WC, de Boer RA, Galectin-3 in heart failure: An update of the last 3 years: Heart Fail Clin, 2018; 14; 75-92
8. Saccon F, Gatto M, Ghirardello A, Role of galectin-3 in autoimmune and non–autoimmune nephropathies: Autoimmun Rev, 2017; 16; 34-47
9. Wu NL, Liu FT, The expression and function of galectins in skin physiology and pathology: Exp Dermatol, 2018; 27; 217-26
10. Pejnovic N, Jeftic I, Jovicic N, Galectin-3 and IL-33/ST2 axis roles and interplay in diet-induced steatohepatitis: World J Gastroenterol, 2016; 22; 9706-17
11. Egger M, Smith GD, Meta-analysis. Potentials and promise: BMJ, 1997; 315; 1371-74
12. Wu R, Wu T, Wang K, Prognostic significance of galectin-1 expression in patients with cancer: A meta-analysis: Cancer Cell Int, 2018; 18; 108
13. Huang MY, He JP, Zhang WQ, Liu JL, Pooling analysis reveals that galectin-1 is a reliable prognostic biomarker in various cancers: J Cell Physiol, 2019; 234; 13788-98
14. Wang K, Chen Z, Wu R, Prognostic role of high gal-9 expression in solid tumours: A meta-analysis: Cell Physiol Biochem, 2018; 45; 993-1002
15. Zhou X, Sun L, Jing D, Galectin-9 expression predicts favorable clinical outcome in solid tumors: A systematic review and meta-analysis: Front Physiol, 2018; 9; 452
16. Wang Y, Liu S, Tian Y, Prognostic role of galectin-3 expression in patients with solid tumors: A meta-analysis of 36 eligible studies: Cancer Cell Int, 2018; 18; 172
17. de Matos LL, Del Giglio AB, Matsubayashi CO, Expression of CK-19, galectin-3 and HBME-1 in the differentiation of thyroid lesions: Systematic review and diagnostic meta-analysis: Diagn Pathol, 2012; 7; 97
18. Xin Y, Guan D, Meng K, Diagnostic accuracy of CK-19, Galectin-3 and HBME-1 on papillary thyroid carcinoma: A meta-analysis: Int J Clin Exp Pathol, 2017; 10; 8130-40
19. Trimboli P, Virili C, Romanelli F, Galectin-3 performance in histologic a cytologic assessment of thyroid nodules: A systematic review and meta-analysis: Int J Mol Sci, 2017; 18; 1756
20. Tang W, Huang C, Tang C, Galectin-3 may serve as a potential marker for diagnosis and prognosis in papillary thyroid carcinoma: A meta-analysis: Onco Targets Ther, 2016; 9; 455-60
21. Sun Q, Zhang Y, Liu M, Prognostic and diagnostic significance of galectins in pancreatic cancer: A systematic review and meta-analysis: Cancer Cell Int, 2019; 19; 309
22. Long B, Yu Z, Zhou H, Clinical characteristics and prognostic significance of galectins for patients with gastric cancer: A meta-analysis: Int J Surg, 2018; 56; 242-49
23. Wang C, Zhou X, Ma L, Galectin-3 may serve as a marker for poor prognosis in colorectal cancer: A meta-analysis: Pathol Res Pract, 2019; 215; 152612
24. Huang Z, Zhong J, Ling Y, Diagnostic value of novel biomarkers for heart failure: A meta-analysis: Herz, 2018; 45; 65-78
25. Chen A, Hou W, Zhang Y, Prognostic value of serum galectin-3 in patients with heart failure: A meta-analysis: Int J Cardiol, 2015; 182; 168-70
26. Chen YS, Gi WT, Liao TY, Using the galectin-3 test to predict mortality in heart failure patients: A systematic review and meta-analysis: Biomark Med, 2016; 10; 329-42
27. Imran TF, Shin HJ, Mathenge N, Meta-analysis of the usefulness of plasma galectin-3 to predict the risk of mortality in patients with heart failure and in the general population: Am J Cardiol, 2017; 119; 57-64
28. Gong M, Cheung A, Wang QS, A systematic review and meta-analysis: J Clin Lab Anal, 2020; 34; e23104
29. Zhang G, Wu Y, Circulating Galectin-3 and atrial fibrillation recurrence after catheter ablation: A meta-analysis: Cardiovasc Ther, 2019; 2019 4148129
30. Pranata R, Yonas E, Chintya V, Serum Galectin-3 level and recurrence of atrial fibrillation post-ablation – systematic review and meta-analysis: Indian Pacing Electrophysiol J, 2020; 20; 64-69
31. Li W, Xu J, Wang X, Lack of association between lymphotoxin-alpha, galectin-2 polymorphisms and coronary artery disease: A meta-analysis: Atherosclerosis, 2010; 208; 433-36
32. Misra S, Kumar P, Kumar A, Genetic association between inflammatory genes (IL-1alpha, CD14, LGALS2, PSMA6) and risk of ischemic stroke: A meta-analysis: Meta Gene, 2016; 8; 21-29
33. Zhang T, Cao S, Yang H, Li J, Prognostic impact of galectin-3 in chronic kidney disease patients: A systematic review and meta-analysis: Int Urol Nephrol, 2019; 51; 1005-11
34. Wu P, van den Berg C, Alfirevic Z, Early pregnancy biomarkers in pre-eclampsia: A systematic review and meta-analysis: Int J Mol Sci, 2015; 16; 23035-56
35. Zhong Y, Zhu F, Ding Y, Serum screening in first trimester to predict pre-eclampsia, small for gestational age and preterm delivery: Systematic review and meta-analysis: BMC Pregnancy Childbirth, 2015; 15; 191
36. Zhu XL, Wang J, Jiang RZ, Teng YC, Pulsatility index in combination with biomarkers or mean arterial pressure for the prediction of pre-eclampsia: Systematic literature review and meta-analysis: Ann Med, 2015; 47; 414-22
Tables
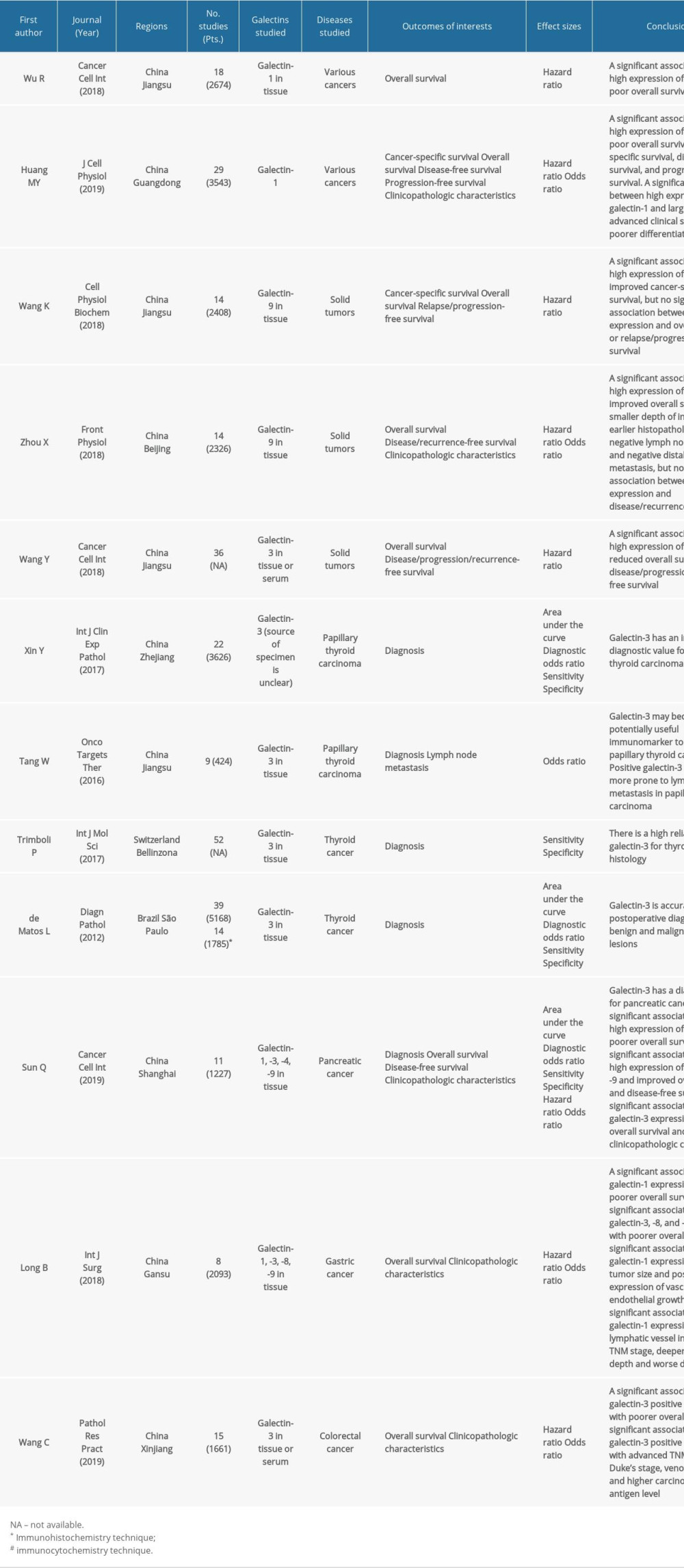 Table 1. Evidence from meta-analyses regarding clinical use of galectins in cancer.
Table 1. Evidence from meta-analyses regarding clinical use of galectins in cancer.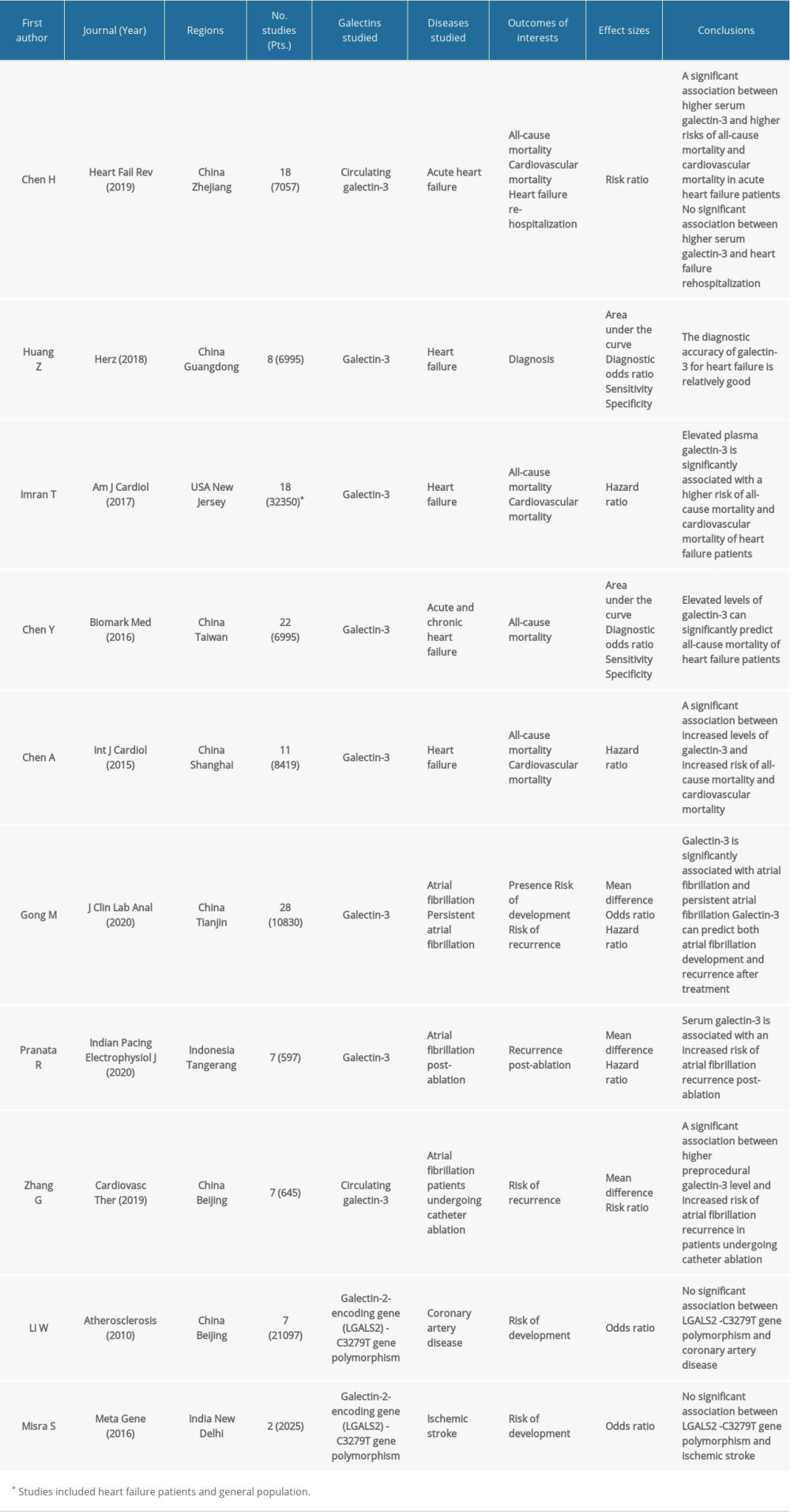 Table 2. Evidence from meta-analyses regarding clinical use of galectins in cardiovascular diseases.
Table 2. Evidence from meta-analyses regarding clinical use of galectins in cardiovascular diseases.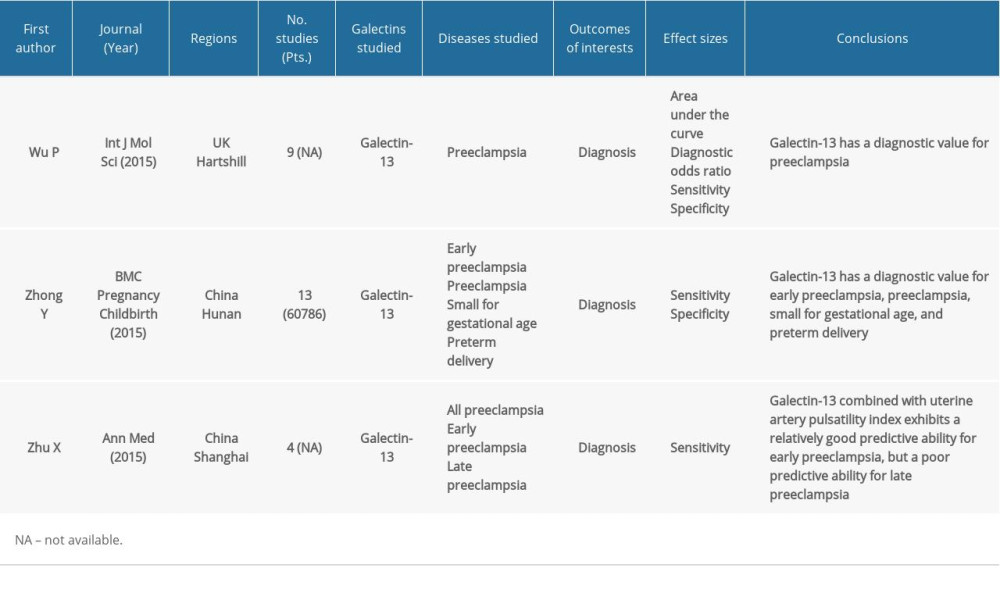 Table 3. Evidence from meta-analyses regarding clinical use of galectins in pregnancy.
Table 3. Evidence from meta-analyses regarding clinical use of galectins in pregnancy. Table 1. Evidence from meta-analyses regarding clinical use of galectins in cancer.
Table 1. Evidence from meta-analyses regarding clinical use of galectins in cancer. Table 2. Evidence from meta-analyses regarding clinical use of galectins in cardiovascular diseases.
Table 2. Evidence from meta-analyses regarding clinical use of galectins in cardiovascular diseases. Table 3. Evidence from meta-analyses regarding clinical use of galectins in pregnancy.
Table 3. Evidence from meta-analyses regarding clinical use of galectins in pregnancy. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952