02 August 2020: Review Articles
The Potential Protective Role of Aspirin Against Migraine in Pregnant Women
Xijing Liu12AEF, Yunhui Gong12AG*DOI: 10.12659/MSM.923959
Med Sci Monit 2020; 26:e923959
Abstract
ABSTRACT: Females are highly predisposed to the occurrence of migraine, a recurrent neurovascular headache disorder. Although migraine improves or disappears during pregnancy, a significant association between migraine and hypertension (i.e., pre-eclampsia) or vascular complications (i.e., stroke) during gestation has been determined. Low-dose aspirin exerts an antithrombotic effect and can improve vascular resistance by regulating endothelial function, which are implicated in the pathogenesis of migraine, pre-eclampsia, and other vascular complications during pregnancy. Low-dose aspirin is widely used prophylactically in the general population who are at higher risk of developing stroke or in pregnant women at higher risk of pre-eclampsia. In this paper we discuss the recent trends in research on the relationship between migraine and pre-eclampsia, an issue of paramount importance in obstetric care, and the potential relationship between migraine and vascular complications in pregnant women. In addition, the potential validity of low-dose aspirin prophylaxis in pregnant women with migraine is explored.
Keywords: Aspirin, Migraine Disorders, Pre-Eclampsia, Pregnancy, Stroke, Drug Administration Schedule, Drug Dosage Calculations, Endothelium, Vascular, Platelet Aggregation Inhibitors, Pregnancy Complications, Cardiovascular
Background
Migraine disorder is a common neurologic disorder in the general population, characterized by headache with severe pulsating one-sided attacks, often accompanied by photophobia and nausea, which can last up to 72 h [1,2]. It has a predilection for females. The lifetime incidence of migraine is 3-fold higher among women than men (prevalence 18%
The pathogenesis of migraine involves an incompletely clarified mechanism originating from neural activation in the brainstem, and then release of neuropeptides associated with vasodilation, vascular dysfunction, inflammation, and pain [1,4,5]. It is assumed ovarian hormonal fluctuation triggers common migraine. Estrogens can modulate neuronal excitability and interact with the vascular endothelium of the brain. An association have been found between estrogen and neurotransmitters such as serotonin, which is involved in modulating the pain threshold [6]. In a rodent model, the mRNA levels of tryptophan hydrolase in the trigeminal ganglia were 3-fold higher when associated with elevated estrogen levels compared to phases of the menstrual cycle in which estrogen levels are low [6]. The drop in estrogen levels increases susceptibility to the effects of prostaglandins (PG), the levels of which are 3-fold higher in the central nervous system and endometrium during the last days of the luteal phase [7,8]. Therefore, migraine benefits from the stable ovarian hormonal environment during pregnancy.
Most epidemiological studies have shown that migraine improves or disappears during pregnancy [9]. A systematic review summarized by Negro indicates about one-half to three-fourths of women with migraine experience a marked improvement during pregnancy, with a significant reduction in frequency and intensity of attacks, if not a complete resolution [10]. Despite these observations, recent studies into the medical complications of pregnancy in women with migraine have cast doubts on this assumption. Indeed, several studies have revealed a significant association between migraine and hypertension (i.e., pre-eclampsia), and vascular complications (i.e., stroke) during gestation.
Migraine and Pre-Eclampsia
Pre-eclampsia (PE), a pregnancy-specific syndrome affecting 3–5% of pregnancies, is one of the leading causes of maternal, fetal, and neonatal morbidity and mortality worldwide [11]. PE is a pregnancy-specific disease characterized by
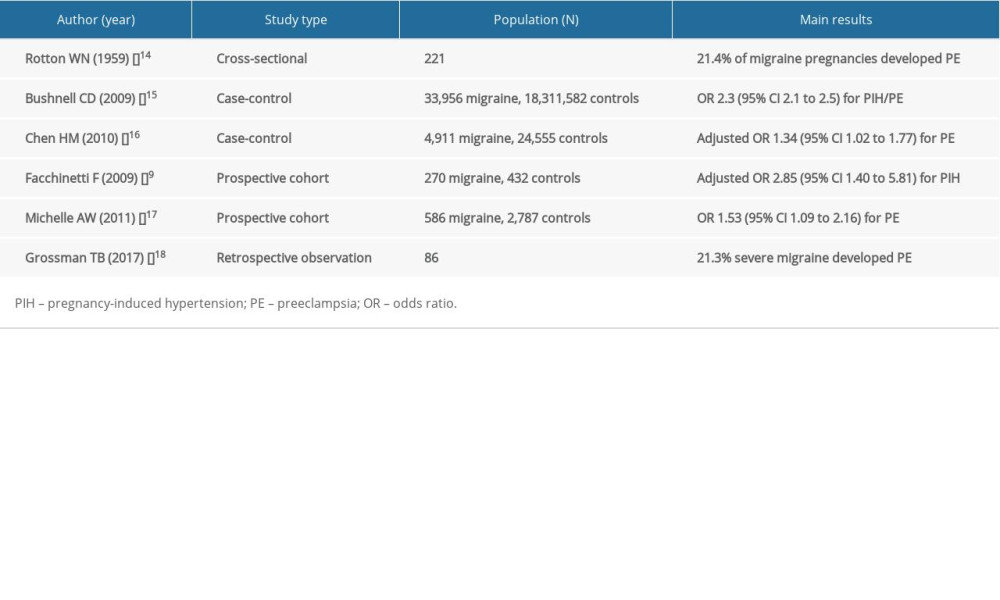
Although the primary mechanisms of both migraine and pre-eclampsia are poorly understood, both diseases are characterized by altered vasoreactivity and endothelial dysfunction [11–13]. In pregnancy, women with migraine may have poor vascular compensatory mechanisms to deal with pre-eclampsia, which implicates endothelial dysfunction. The hypothesis that women with migraine have a higher incidence of pre-eclampsia has been verified by some epidemiological studies (Table 1).
A correlation between migraine and pre-eclampsia was hypothesized as early as 1959 [14]. A cross-sectional study reported 21.4% of migraine pregnancies (n=221) developed some forms of pre-eclampsia [14]. However, the findings must be interpreted with caution as the diagnosis of both migraine and pre-eclampsia (as toxemia used to be called) was inconsistent with contemporary standards.
A US population-based case-control study analyzing pregnancy-related discharge diagnoses by distinguishing ICD-9 codes from 2000 to 2003 demonstrated 33 956 migraine codes were identified (total 18 345 538 pregnancy-related discharges) [15]. Pre-eclampsia/gestational hypertension codes were associated with migraine codes (OR 2.3, 95% CI 2.1 to 2.5) [15]. Although the study used a population-based sample, the study has obvious limitations related to analyzing ICD-9 codes at discharge from hospital; for instance, some diagnoses of peripartum migraine could have been miscoded or inadvertently listed separately in the setting of pre-eclampsia in which headache is a symptom [15].
In a large case-control study designed to examine the risk of adverse pregnancy outcomes in East Asian women with migraine, Chen et al. identified a total of 4911 women with migraine, together with 24 555 matched women as a comparison cohort [16]. After adjusting for infant sex, parity, maternal age, highest maternal education level, parental age difference, mother’s marital status, and family monthly income, as well as hypertension, diabetes, hyperlipidemia, and coronary heart disease, the odds ratio (OR) for pre-eclampsia was 1.34 (95% CI 1.02 to 1.77) [16]. Although, the sample size was relatively large, the sample may not accurately represent the population with migraine due to women diagnosed with migraine by ICD-9-CM code from the National Health Insurance Research Dataset. Also, the diagnostic criteria of PE were not detailed.
Facchinetti et al. conducted a prospective cohort study in 270 cases of migraine (based on ICHD-II) and 432 controls. The OR adjusted for age, family history of hypertension, and smoking habits was 2.85 (95% CI 1.40 to 5.81) for hypertensive disorders in pregnancy (as defined by ACOG 2012 criteria) [9]. The study further clarified the relationship between the course of migraine in pregnancy and onset of hypertensive disorders. Women whose symptoms did not change (OR 1.92, 95% CI 0.71 to 5.19) or worsened (OR 13.65, 95% CI 4.13 to 45.08) showed a higher risk of developing symptoms that either disappeared or improved [9]. However, the study did not perform separate analyses for the risk of PE.
In addition, a prospective cohort study of 3373 pregnant women indicated that migraineurs had 1.53-fold increased odds of having pre-eclampsia (95% CI 1.09 to 2.16) [17]. Additionally, migraineurs who were overweight or obese had 6.10-fold increased odds of pre-eclampsia (95% CI 3.83 to 9.75) as compared with lean non-migraineurs [17].
In a recent retrospectively study, Grossman et al. examined the data of 86 severe migraineurs manifesting in pregnancy requiring emergent treatment who gave birth between 2009 and 2014; patients experiencing severe migraine during pregnancy had high rates of PE 21.3% (95% CI 7.7–24.3). Patients at or above the age of 35 years had a greater chance of having PE (7/16 [43.8%]) compared to those who were younger (15/63 [23.8%]). Although this is the only study focusing on pregnant women with the most severe migraines, this was an observational study with a small sample size [18].
These preliminary results reinforced the idea that PE and migraine share similar pathogenetic mechanisms. Overall, evidence suggests that migraine is an independent risk factor in PE and the risk of PE may sharply rise when combined with other risk factor (e.g., obesity and age).
Migraine and Cardiovascular Disease During Pregnancy
Several case-control and cohort studies, as well as pooled data analyses, indicate that migraine is a risk factor for stroke and other vascular events [19–21]. Stroke is a relatively rare but devastating complication during pregnancy. James et al. conducted a study based on the United States Nationwide Inpatient Sample from the Healthcare Cost and Utilization Project of the Agency for Healthcare Research and Quality during 2000–2001, showing diagnosis of stroke at a rate of 34.2 per 100 000 deliveries in a total of 2850 pregnancy-related discharges [22]. However, the incidence was roughly 3 times that seen in the general population of young adults [23].
When ischemic stroke was the outcome of interest, migraine was strongly associated with stroke (OR 16.9, CI 9.7–29.5) [22]. A second analysis, performed by Ashley Wabnitz and Cheryl Bushnell, reported an association between increased risk of ischemic stroke and migraine in pregnancy (OR range 7.9–30.7) [24], in which the risk for stroke was 15-fold higher, with odds ratios of 30.7 for ischemic and 9.1 for hemorrhagic stroke.
In addition, evidence demonstrates migraine increases the risk of cardiovascular disease (CVD) (HR 1.5, 95% CI 1.33–1.69) and cardiovascular mortality (HR 1.37, 95% CI 1.02–1.83) [20]. Despite the higher relative risk, the absolute risk of CVD in young women is low except for pregnant women. For instance, pregnant women are 4 times more likely to develop venous thromboembolism (DVT) than non-pregnant women, with a standardized incidence ratio of 4.29 (95% CI 3.49–5.22) [25]. Nevertheless, few studies have specifically focused on the risk of pregnancy-related CVD in women with migraine. Bushnell conducted a US population-based case-control study based on the Nationwide Inpatient Sample from the Healthcare Cost and Utilization Project of the Agency for Healthcare Research and Quality during 2000–2003. The results showed that migraines were associated with nearly a 5-fold increased risk of acute myocardial infraction and heart disease during pregnancy (OR 4.9, 95% CI 1.7–14.2), and an increased risk of thromboembolism such as pulmonary embolism (OR 3.1, 95% CI 1.7–5.6), deep vein thrombosis (OR 2.4, 95% CI 1.3–4.2), and thrombophilia (OR 3.6, 95% CI 2.1–6.1) during pregnancy [15].
Pregnancy is widely thought of as a high-risk factor for stroke or CVD due to hypercoagulability and hemodynamic changes [1]. Researchers hypothesize that the vascular vulnerability and hypercoagulation state of pregnancy may be involved [5]. Migraine can be regarded as an expression of this underlying condition which, when combined with other modifiers of vascular health, can lead to a synergistic increase in CVD risk [1,5]. This hypothesis may explain the multiplicative risk seen in young females with migraine who take oral contraceptives [1,26]. In addition, increased inflammation, thrombogenicity, and altered vasodilatory reaction have been verified in migraine sufferers, and can be seen as markers of dysfunction of the endothelium, which is the inner lining of the blood vessel that normally has vasoprotective properties [1,13].
Aspirin in Pregnancy
It is widely accepted that aspirin plays a positive role in preventing cardiovascular events. As a nonsteroidal anti-inflammatory drug, aspirin prevents cyclooxygenases binding to arachidonic acid, which inhibits the production of prostanoids such as prostacyclins and thromboxane A2 [27]. It can also increase the production of nitric oxide (NO), an endothelial-derived vasodilator, by acetylating endothelial NO synthase [28]. In a recent animal study, aspirin was shown to have a direct vasodilatory effect on rat uterine artery, which is primarily mediated by endothelial cells [29].
There has been limited progress in developing remedies for pre-eclampsia, as the pathogenesis of the disease is not completely understood. The current strategy for management of established pre-eclampsia focuses on balancing of risks to the mother while minimizing the risk of iatrogenic prematurity [11]. To date, pregnancy termination is the only way to eradicate the disease. Therefore, emphasis has been placed on the prevention of pre-eclampsia. For years, low-dose aspirin has been commonly used to prevent or delay the onset of pre-eclampsia [30,31].
It is important to identify women who will benefit from aspirin treatment. Current guidelines recommend the use of clinical history in risk stratification of women [31,32]. The American College of Obstetricians and Gynecologists (ACOG) committee suggests low-dose aspirin (81 mg/day) prophylaxis is recommended and should be initiated at 12–28 weeks of gestation (optimally before 16 weeks) and continued daily until delivery [31]. Low-dose aspirin prophylaxis should be considered for women at high risk of pre-eclampsia or those who have more than 1 of several moderate risk factors for pre-eclampsia [31]. Women at risk of pre-eclampsia are defined based on the presence of 1 or more high-risk factors (history of pre-eclampsia, multifetal gestation, renal disease, autoimmune disease, type 1 or type 2 diabetes, and chronic hypertension) or more than 1 of several moderate risk factors (first pregnancy, maternal age >35 years, a body mass index >30, family history of pre-eclampsia, sociodemographic characteristics, and personal history factors) [31]. Unfortunately, with regard to pregnancy, doctors often ignore that women with migraines have an increasing risk of pre-eclampsia.
Aspirin therapy for stroke prevention has been extensively investigated and shown to reduce the risk of future vascular events; however, its use in pregnancy has been less well researched [33]. In terms of primary prevention of stroke in patients with migraine, aspirin (100 mg/day) reduced the risk of ischemic stroke (RR 0.76; 95% CI, 0.63–0.93), but not other CVDs [34]. However, the findings in the study should be interpreted with caution due to the small number of outcome events in subgroups [34]. Although evidence is insufficient to make clear evidence-based recommendations about stroke prevention in pregnant women, Mayte et al. summarized a practical guide to manage ischemic stroke during pregnancy, and suggested that aspirin (50–150 mg/day), which is well-tolerated during pregnancy, could be considered for women with higher risk of stroke, including cardioembolism, migraine, and coagulation disorders [35].
Although national guidelines recommend that women at risk of developing pre-eclampsia should be advised to take aspirin daily, the optimal timing and dose of the initiation of aspirin for pre-eclampsia prevention remain controversial [31,36]. Recent studies show that prophylaxis with 100–150 mg of aspirin given before 14–16 weeks can significantly reduce pre-eclampsia rates and may improve pregnancy outcome [30,37]. A meta-analysis conducted by Bujold et al. demonstrated that commencing low-dose aspirin before 16 weeks of gestation significantly decreased the rate of pre-eclampsia (RR 0.47, 95% CI 0.34–0.65) [38]. Although most centers use aspirin 75–100 mg daily, there has been a shift towards using a dose of greater than 100 mg daily based on more recent studies. A large multicenter randomized trial showed that aspirin (150 mg per day) from 11–14 weeks to 36 weeks of gestation reduced the risks of preterm pre-eclampsia [30]. Of particular note, a recent meta-analysis reported aspirin at a daily dose greater than or equal to 100 mg for the prevention of pre-eclampsia that was initiated at ≤16 weeks can reduce the risk of placental abruption or antepartum hemorrhage [37]. However, there is a need for more stronger evidence to support the benefits and safe use of aspirin over 100 mg daily before 16 weeks of gestation in the prevention of pre-eclampsia [30,39].
In general, aspirin is typically discontinued after terminating pregnancy. The Hale lactation rating of aspirin is L3 [40]. It is reported aspirin is associated with Reye’s syndrome in infants and potential adverse effects on infant’s platelet function [40]. Whether continued aspirin use after pregnancy is beneficial in women with a history of pre-eclampsia remains controversial and is rarely discussed. A large cohort in the US population raises the possibility that primary preventive treatment with aspirin after a pregnancy complicated by hypertensive disorders of pregnancy might reduce future stroke risk in this population [41]. Starting low-dose aspirin therapy in post-partum women with migraine also warrants comprehensive investigation.
Conclusions
Although pregnancy is a time of relative well-being for women with migraine, as headaches improve, migraine sufferers may be exposed to additional clinical risks, such as pre-eclampsia, stroke or thromboembolic events. Low-dose aspirin prophylaxis appears to have a protective role in decreasing pregnancy complications. It is of great importance to verify the validity of low-dose aspirin prophylaxis in pregnant women with migraine as vascular homeostasis and to elucidate the antithrombotic effects of aspirin use. In practice, randomized controlled trials could be conducted to evaluate the benefits of the starting low-dose aspirin in pregnant women with migraine. An animal model of migraine could be used to reveal the underling pathogenesis of both migraine and pre-eclampsia, as well as the mechanism of action of aspirin. Simple, inexpensive primary preventive interventions could have major consequences in this high-risk group.
References
1. Linstra KM, Ibrahimi K, Terwindt GM, Migraine and cardiovascular disease in women: Maturitas, 2017; 97; 28-31
2. Stovner L, Hagen K, Jensen R, The global burden of headache: A documentation of headache prevalence and disability worldwide: Cephalalgia, 2007; 27(3); 193-210
3. Bigal ME, Lipton RB, The epidemiology, burden, and comorbidities of migraine: Neurol Clin, 2009; 27(2); 321-34
4. Allais G, Chiarle G, Sinigaglia S, Estrogen, migraine, and vascular risk: Neurol Sci, 2018; 39(Suppl 1); 11-20
5. Bigal ME, Kurth T, Hu H, Migraine and cardiovascular disease: Possible mechanisms of interaction: Neurology, 2009; 72(21); 1864-71
6. Berman NE, Puri V, Chandrala S, Serotonin in trigeminal ganglia of female rodents: relevance to menstrual migraine: Headache, 2006; 46(8); 1230-45
7. Nattero G, Allais G, De Lorenzo C, Relevance of prostaglandins in true menstrual migraine: Headache, 1989; 29(4); 233-38
8. Durham PL, Vause CV, Derosier F, Changes in salivary prostaglandin levels during menstrual migraine with associated dysmenorrhea: Headache, 2010; 50(5); 844-51
9. Facchinetti F, Allais G, Nappi RE, Migraine is a risk factor for hypertensive disorders in pregnancy: A prospective cohort study: Cephalalgia, 2009; 29(3); 286-92
10. Negro A, Delaruelle Z, Ivanova TA, Headache and pregnancy: A systematic review: J Headache Pain, 2017; 18(1); 106
11. Mol BWJ, Roberts CT, Thangaratinam S, Pre-eclampsia: Lancet, 2016; 387(10022); 999-1011
12. Steegers EA, von Dadelszen P, Duvekot JJ, Pijnenborg R, Pre-eclampsia: Lancet, 2010; 376(9741); 631-44
13. Tietjen GE, Migraine as a systemic vasculopathy: Cephalalgia, 2009; 29(9); 987-96
14. Rotton WN, Sachtleben MR, Friedman EA, Migraine and eclampsia: Obstet Gynecol, 1959; 14; 322-30
15. Bushnell CD, Jamison M, James AH, Migraines during pregnancy linked to stroke and vascular diseases: US population based case-control study: BMJ, 2009; 338; b664
16. Chen HM, Chen SF, Chen YH, Lin HC, Increased risk of adverse pregnancy outcomes for women with migraines: A nationwide population-based study: Cephalalgia, 2010; 30(4); 433-38
17. Williams MA, Peterlin BL, Gelaye B, Trimester-specific blood pressure levels and hypertensive disorders among pregnant: Headache, 2011; 51(10); 1468-82
18. Grossman TB, Robbins MS, Govindappagari S, Dayal AK, Delivery outcomes of patients with acute migraine in pregnancy: A retrospective: headache, 2017; 57(4); 605-11
19. Etminan M, Takkouche B, Fau-Isorna FC, Risk of ischaemic stroke in people with migraine: Systematic review and meta-analysis of observational studies: BMJ, 2005; 330(7482); 63
20. Kurth T, Winter AC, Eliassen AH, Migraine and risk of cardiovascular disease in women: Prospective cohort study: BMJ, 2016; 31(353); i2610
21. Schurks M, Rist PM, Bigal ME, Migraine and cardiovascular disease: Systematic review and meta-analysis: BMJ, 2009; 27(339); b3914
22. James AH, Bushnell CD, Jamison MG, Myers ER, Incidence and risk factors for stroke in pregnancy and the puerperium: Obstet Gynecol, 2005; 106(3); 509-16
23. Singhal AB, Biller J, Elkind MS, Recognition and management of stroke in young adults and adolescents: Neurology, 2013; 81(12); 1089-97
24. Wabnitz A, Bushnell C, Migraine, cardiovascular disease, and stroke during pregnancy: Systematic review of the literature: Cephalalgia, 2015; 35(2); 132-39
25. Heit JA, Kobbervig CE, James AH, Trends in the incidence of venous thromboembolism during pregnancy or postpartum: A 30-year population-based study: Ann Intern Med, 2005; 143(10); 697-706
26. Sacco S, Merki-Feld GS, Ægidius KL, Hormonal contraceptives and risk of ischemic stroke in women with migraine: A consensus statement from the European Headache Federation (EHF) and the European Society of Contraception and Reproductive Health (ESC): J Headache Pain, 2017; 18(1); 108
27. Roth GJ, Calverley DC, Aspirin, platelets, and thrombosis: Theory and practice: Blood, 1994; 83(4); 885-98
28. Taubert D, Berkels R, Grosser N, Aspirin induces nitric oxide release from vascular endothelium: A novel mechanism of action: Br J Pharmacol, 2004; 143(1); 159-65
29. Helgadottir H, Tropea T, Gizurarson S, Aspirin causes endothelium-dependent vasodilation of resistance arteries from non-gravid and gravid rats: Pregnancy Hypertens, 2019(15); 141-45
30. Rolnik DL, Wright D, Poon LC, Aspirin versus placebo in pregnancies at high risk for preterm preeclampsia: N Engl J Med, 2017; 377(7); 613-22
31. , ACOG Committee Opinion No. 743: Low-dose aspirin use during pregnancy: Obstet Gynecol, 2018; 132(1); e44-52
32. Lowe SA, Bowyer L, Lust K, The SOMANZ guidelines for the management of hypertensive disorders of pregnancy 2014: Aust NZ J Obstet Gynaecol, 2015; 55(1); 11-16
33. Kernan WN, Ovbiagele B, Black HR, Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the American Heart Association/American Stroke Association: Stroke, 2014; 45(7); 2160-236
34. Kurth T, Diener HC, Buring JE, Migraine and cardiovascular disease in women and the role of aspirin: Subgroup analyses in the Women’s Health Study: Cephalalgia, 2011; 31(10); 1106-15
35. van Alebeek ME, de Heus R, Tuladhar AM, de Leeuw FE, Pregnancy and ischemic stroke: A practical guide to management: Curr Opin Neurol, 2018; 31(1); 44-51
36. Lowe SA, Bowyer L, Lust K, SOMANZ guidelines for the management of hypertensive disorders of pregnancy 2014: Aust NZ J Obstet Gynaecol, 2015; 55(5); e1-29
37. Roberge S, Bujold E, Nicolaides KH, Meta-analysis on the effect of aspirin use for prevention of preeclampsia on placental abruption and antepartum hemorrhage: Am J Obstet Gynecol, 2018; 218(5); 483-89
38. Bujold E, Roberge S, Lacasse Y, Prevention of preeclampsia and intrauterine growth restriction with aspirin started in early pregnancy: A meta-analysis: Obstet Gynecol, 2010; 116(2 Pt 1); 402-14
39. Park F, Russo K, Williams P, Prediction and prevention of early-onset pre-eclampsia: Impact of aspirin after first-trimester screening: Ultrasound Obstet Gynecol, 2015; 46(4); 419-23
40. Hutchinson S, Marmura MJ, Calhoun A, Use of common migraine treatments in breast-feeding women: A summary of recommendations: Headache, 2013; 53(4); 614-27
41. Miller EC, Boehme AK, Chung NT, Aspirin reduces long-term stroke risk in women with prior hypertensive disorders of pregnancy: Neurology, 2019; 92(4); e305-16
In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952