14 September 2020: Database Analysis
A Systematic Pharmacology and Study to Identify the Role of the Active Compounds of in the Treatment of Osteoarthritis
Zhenyuan Chen12ABCDE, Guangwen Wu13AG*, Ruoxi Zheng1EDOI: 10.12659/MSM.925545
Med Sci Monit 2020; 26:e925545
Abstract
BACKGROUND: Achyranthes bidentata is a Chinese traditional herbal medicine widely used to treat osteoarthritis (OA). This study aimed to identify active compounds from Achyranthes bidentata through systematic pharmacology and in vitro experiments to find the targets of Achyranthes bidentata in the treatment of OA.
MATERIAL AND METHODS: We screened the active compounds of Achyranthes bidentata from the Traditional Chinese Medicine Systems Pharmacology (TCMSP) database. Then, we used STITCH and Open Targets Platform databases to screen the active components and predict the potential targets of Achyranthes bidentata in the treatment of OA. Subsequently, we studied the compound-target network and protein interaction network and analyzed the enrichment of potential target proteins. Finally, we used Western blot analysis to verify the therapeutic effect of Achyranthes bidentata extract on the expression of OA-related target proteins.
RESULTS: There were 7 active components in Achyranthes bidentata, which were strongly related to the 74 targets of OA. Quercetin, baicalein, and berberine are the critical active compounds of Achyranthes bidentata in the treatment of OA. Protein interaction analysis and in vitro experiments suggested that TNF, IL-6, and TP53 are the critical targets of Achyranthes bidentata in the treatment of OA. Functional enrichment analysis showed that Achyranthes bidentata plays a pharmacological role in OA through apoptosis, inflammation, and immune regulation.
CONCLUSIONS: Quercetin, baicalein, and berberine are the critical active compounds of Achyranthes bidentata in the treatment of OA. TNF, IL-6, and TP53 may be potential targets for the treatment of OA.
Keywords: Molecular Mechanisms of Pharmacological Action, Osteoarthritis, Computer Communication Networks, Achyranthes, Chondrocytes, Drug Evaluation, Preclinical, Drugs, Chinese Herbal, In Vitro Techniques
Background
Osteoarthritis is a chronic degenerative and disabling arthropathy of complex etiology. Its main clinical manifestations are joint pain and deformity and movement limitation [1]. Most scholars believe that it is the result of an imbalance between degradation and synthesis among articular cartilage cells, extracellular matrix, and sub-cartilage bone [2]. At present, the leading role of drugs in the treatment of osteoarthritis is to relieve symptoms, but they cannot effectively prevent the development of osteoarthritis [3]. Therefore, it is of considerable significance to find a safe and effective drug for treating OA.
Systematic pharmacology applies the knowledge of system biology to pharmacology. The purpose of this discipline is to clarify how drugs act on the body through biological systems. Unlike evaluating the effect of a drug by a particular protein–drug reaction, systematic pharmacology suggests that drugs act by forming a network of interactions [8]. Interaction systems may include drug–protein and protein–protein interactions, as well as genetic, signaling, and physiological interactions. Systematic pharmacology uses bioinformatics and statistics to integrate and explain the network of drug interactions, which provides an effective method to study the biological mechanisms of TCM in treating diseases [9,10].
A recent systematic pharmacological study of
Material and Methods
SCREENING FOR ACTIVE COMPOUNDS:
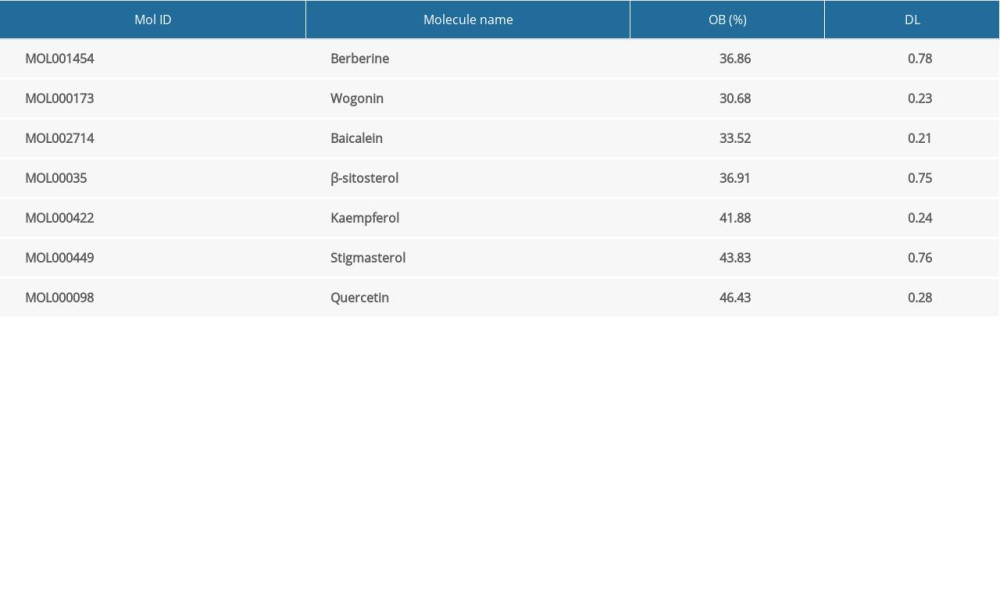
The Traditional Chinese Medicine Systems Pharmacology database (TCMSP; http://tcmspw.com/tcmsp.php) is a systematic pharmacological database for evaluating the pharmacokinetic properties of traditional Chinese medicine or related compounds [12]. It provides data on the absorption, distribution, metabolism, and excretion (ADME) of traditional Chinese herbs in vivo, including oral bioavailability (OB) and drug-like (DL). OB is the most critical index used to evaluate the effectiveness of oral drugs [13]. DL refers to the similarity between components and known drugs. In drug development, DL evaluation helps to identify qualified compounds and improve the success rate of candidate drugs [14]. Based on the published literature and preliminary information in the TCMSP database [15], we entered the drug name “Achyranthes bidentata” into the search box in the TCMSP database and screened the ingredients with OB ≥30% and DL ≥18% for further study.
IDENTIFICATION OF DRUG TARGETS:
The STITCH (http://stitch.embl.de/) database was used to search the potential target proteins of active components in Achyranthes bidentate [16]. STITCH is a database used to explore and predict the interaction between compounds and proteins. Through experiments, databases, and evidence in the literature, chemicals are shown to be associated with other chemicals and proteins. In the present study, we input active components into the STITCH database to screen Achyranthes bidentata target proteins with confidence >0.4 in the human species for further analysis.
IDENTIFICATION OF DISEASE TARGETS:
OA targets were collected from the Open Targets Platform (https://www.targetvalidation.org) [17], which is a comprehensive and authoritative database for screening and visualizing potential drug targets related to diseases. It brings together a variety of data types designed to help users identify and prioritize targets for further research [18]. We used the keyword “osteoarthritis” in the Open Targets Platform. Then, the proteins overlapping the Achyranthes bidentata targets were selected for further study.
CONSTRUCTION OF A COMPONENT–TARGET NETWORK:
A ‘component–target network’ diagram was constructed using Cytoscape 3.7.1 (https://cytoscape.org/) software to reveal the main active components and regulatory mechanisms of Achyranthes bidentata in the treatment of OA [19].
CONSTRUCTION OF A TARGET–TARGET INTERACTION NETWORK:
A target–target interaction network based on STRING11.0 (http://string-db.org/) was constructed in Cytoscape3.7.1 to identify the critical proteins among the target proteins listed above [20].
ANALYSIS OF GENE FUNCTION AND SIGNAL PATHWAY ENRICHMENT:
ClusterProfiler was used to analyze the enrichment of biological processes and signaling pathways. ClusterProfiler is an R package for gene cluster enrichment analysis, which can be used to thoroughly understand the function of genes and the enrichment of pathways [21]. Given the overlapping target gene list, clusterProfiler will use the Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) databases to analyze the biological process and signal pathway enrichment of the given gene list [22–24].
ANIMALS: We purchased a total of 6 Sprague Dawley (SD) rats (4 weeks old, weight 95–110 g, both sexes) from Shanghai SLAC Laboratory Animal, Inc., China [Laboratory Animal Use Certificate no. SCXK(SH)2017-0005] and raised them in a sterile environment. Experiments involving animals complied with the 2006 edition of the guidelines for the Care and Use of Experimental Animals by the Ministry of Science and Technology, China [2]5.
PREPARATION OF ACHYRANTHES BIDENTATA EXTRACT:
CHONDROCYTES CULTURE AND IDENTIFICATION: Briefly, the rats were anesthetized by intraperitoneal injection of 2% pentobarbital and then sacrificed via rapid decapitation. Then, cartilage tissue of rats was taken under aseptic conditions. The cartilage tissue was cut into pieces and digested with 0.5% type II collagenase (Sigma, Darmstadt, Germany) at 37°C for 5 hours. We filtered the cells with a 70-μm cell filter and washed them 3 times with aseptic phosphate-buffered saline (PBS; HyClone, Logan, UT, USA). After that, the collected cells were inoculated into a cell culture flask containing Dulbecco’s modified Eagle’s medium (DMEM; HyClone, Logan, UT, USA) with 10% fetal bovine serum (Gibco, New York, USA) and cultured at 37°C and 5% CO2. We changed the culture medium every 48 hours. We identified primary chondrocytes by type II collagen immunofluorescence staining using a previously described method [26,27]. Only primary or second-generation chondrocytes were used for the experiment.
ESTABLISHMENT OF A DEGENERATIVE CHONDROCYTE MODEL: We used IL-1β to establish a model of degenerative chondrocytes using methods described elsewhere [28,29]. Briefly, we treated secondary chondrocytes with 10 ng/ml IL-1β for 24 hours.
EXPERIMENTAL GROUPING: We divided the cells into 2 groups: i) the degenerative chondrocyte group and ii) the degenerative chondrocyte with Achyranthes bidentata extract group. The intervention time for each group was 24 hours. We treated the chondrocytes with the Achyranthes bidentata extract, as described previously [30].
WESTERN BLOT ANALYSIS: The expression level of the target protein in chondrocytes of each group was calculated by Western blot and semi-quantitative analysis, using methods we described previously [31]. The following antibodies were used: TNF-α antibody (1: 1000; Abcam, Cambridge, MA, USA), IL-6 antibody (1: 1000; Abcam, Cambridge, MA, USA), p53 antibody (1: 1000; Cell Signaling Technology, Inc., Danvers, MA, USA), β-actin antibody (1: 2000; Abcam, Cambridge, MA, USA), and secondary antibodies (1: 20 000; Merck Millipore, Darmstadt, Germany).
Results
:
As described in the Methods section above, we searched the TCMSP database with the keyword “
We searched the STITCH database for 20 compounds screened from the TCMSP database, of which 7 compounds had 131 target proteins. Among them, baicalin has 37 targets, β-sitosterol has 11 targets, kaempferol has 18 targets, quercetin has 46 targets, stigmasterol has 3 targets, baicalin has 11 targets, and berberine has 49 targets. Table 1 shows the ADME properties of these 7 compounds.
IDENTIFICATION OF OSTEOARTHRITIS-RELATED PROTEINS:
By searching the Open Targets Platform database, we identified 74 targets closely related to OA as potential targets for the treatment of OA (Figure 2A). Then, we established a network of compounds–targets (Figure 2B). Among these 7 compounds, quercetin (Degree=28), berberine (Degree=25), and baicalein (Degree=26) connect more targets than the other 4 compounds, which suggests that they play an essential role in OA. All active ingredients of Achyranthes bidentata are linked to more than 2 targets, which indicates that Achyranthes bidentata showed multi-targeting in the treatment of OA.
TARGET–TARGET INTERACTION ANALYSIS:
By using Cytoscape 3.7.1, we constructed a diagram of the “target–target network” based on the results from the STRING database (Figure 3A). We constructed a bar plot according to the degrees of connection between the targets (Figure 3B). The results show that IL-6, TNF, and TP53 are the central targets in the network and connect to the most targets.
GENE FUNCTION AND SIGNAL PATHWAY ENRICHMENT ANALYSIS:
GO and KEGG analyses were performed with clusterProfiler to analyze the functional characteristics of the targets, and the threshold was set as P≤0.01. The results of GO analysis showed that 317 GO terms were enriched in ‘Biological Process’ (BP). The first 20 items with the most significant P-values are shown in Figure 4A. As presented, response to lipopolysaccharide, response to oxidative stress, response to a steroid hormone, and regulation of inflammatory response are involved in supporting the role of Achyranthes bidentata in treating OA.
The results of KEGG pathway enrichment showed that there are 34 signal pathways. The first 20 signaling pathways with the most significant P-values are shown in Figure 4B. As presented, Apoptosis, TNF signal pathway, T cell receptor signal pathway, Toll-like receptor signal pathway, Osteoclast differentiation, NF-κB signal pathway, and MAPK signal pathway are involved in supporting the effect of Achyranthes bidentata in treating OA.
WESTERN BLOT ANALYSIS:
The protein expressions of TNF-α, IL-6, and p53 were calculated to verify the effect of Achyranthes bidentata on OA, as shown in Figure 5A–5D. Compared with the degenerative chondrocyte group, the Achyranthes bidentata group had lower expressions of TNF-α (P<0.05), IL-6 (P<0.01), and p53 (P<0.05) proteins.
Discussion
The systematic pharmacology method was developed for use with the molecular biological network analysis, which is used to discover new therapeutic drugs derived from natural products. Therefore, it provides a systematic means to expand the application of available drug compounds in TCM to treat a variety of complex diseases. OA is a common type of chronic arthritis with painful symptoms that affect patient quality of life. Although
In this study, we evaluated the active components and target proteins of
TNF-α and IL-6 are 2 essential targets of OA anti-inflammatory therapy and are central inflammatory cytokines involved in articular cartilage degradation. TNF-α has a variety of biological effects that can directly cause peroxidation of chondrocytes and promote degradation of the cartilage matrix. It can also induce the release of other inflammatory cytokines such as IL-1 and IL-6 by activating the NF-κB pathway and MAPK pathway [40]. IL-6 has a synergistic effect with TNF in the pathogenesis of OA and can induce osteoclast differentiation and accelerate the destruction of articular cartilage [41]. In this study, TNF and IL-6 were the found to be critical elements of the protein interaction network, and Western blot analysis confirmed that
The TP53-dependent apoptosis pathway is an essential mechanism of OA induced by chondrocyte apoptosis [42]. In osteoarthritis, TP53 can inhibit DNA replication, block cell cycle, lead to apoptosis, and accelerate cartilage degradation [43]. TP53 is also a vital element of the protein interaction network. In this study, Western blot analysis confirmed that
The results of GO and KEGG pathway analyses indicated that
There are some limitations to this study. Systematic pharmacology is still in the early stage of development. At present, the data on drugs, compounds, proteins, and genes in the TCM database are incomplete, and the availability of the data obtained is limited [44]. In addition, there are differences between rat chondrocytes and human chondrocytes, and the present results need to be further verified in human chondrocytes and by
Conclusions
In conclusion,
Figures
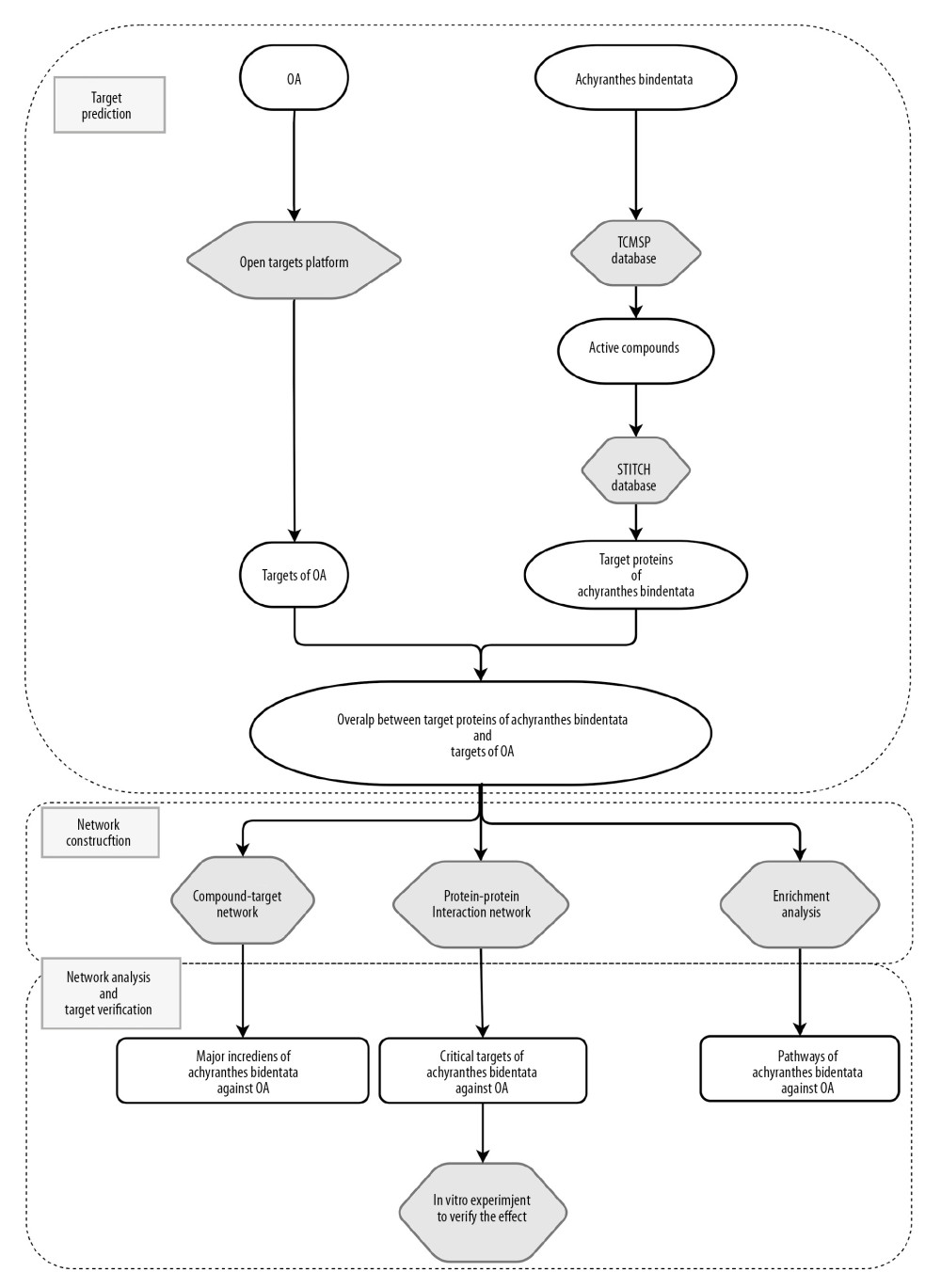 Figure 1. Study flow chart.
Figure 1. Study flow chart. 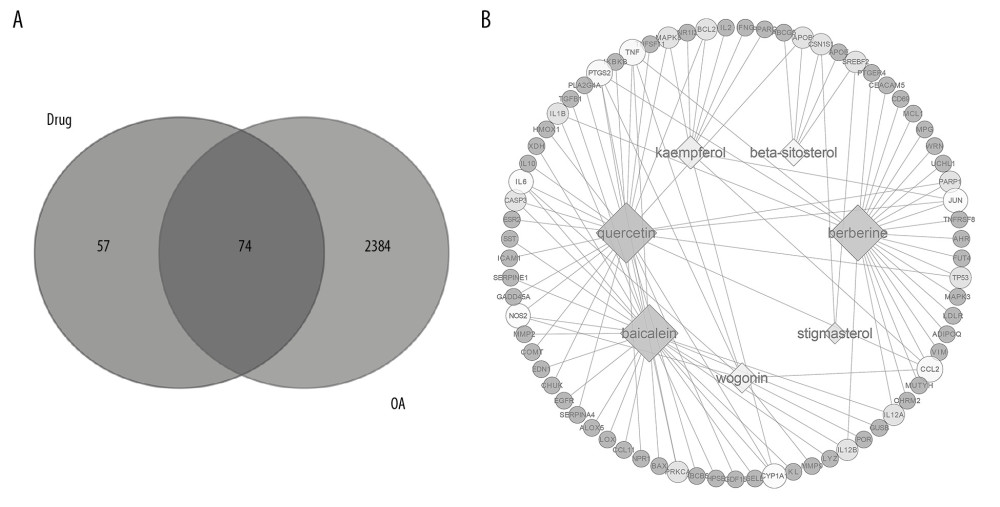 Figure 2. Venn analysis and network analysis of active compounds and potential targets of Achyranthes bidentata on OA. (A) The Venn diagram of target proteins of Achyranthes bidentata and OA-related targets based on the Open Targets Platform. (B) The compound-targets network diagram of Achyranthes bidentata to treat OA, the square nodes represent compounds. The circular nodes represent target proteins.
Figure 2. Venn analysis and network analysis of active compounds and potential targets of Achyranthes bidentata on OA. (A) The Venn diagram of target proteins of Achyranthes bidentata and OA-related targets based on the Open Targets Platform. (B) The compound-targets network diagram of Achyranthes bidentata to treat OA, the square nodes represent compounds. The circular nodes represent target proteins. 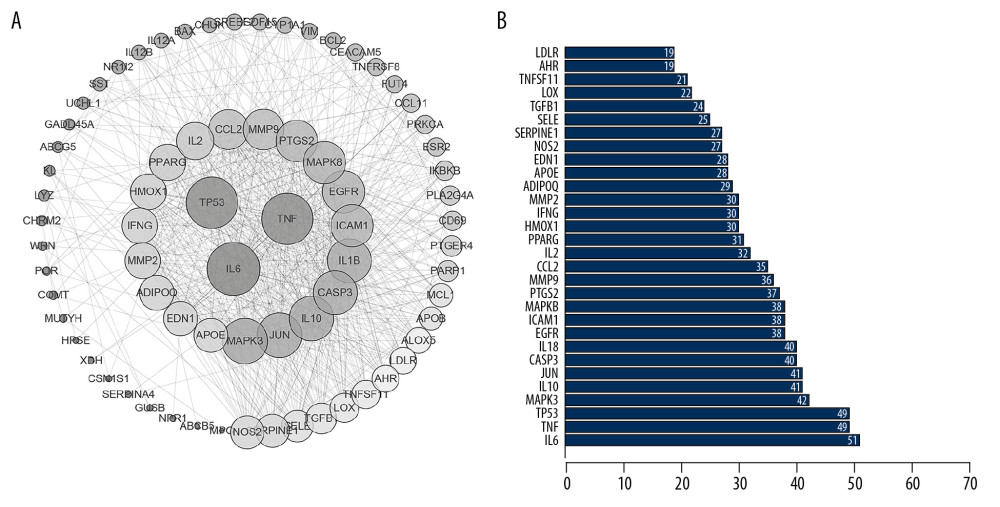 Figure 3. ‘Target–Target’ interaction network analysis. (A) The target–target interaction network is constructed by using Cytoscape 3.7.1. The size of the nodes is proportional to degrees. (B) The bar plot on degrees of connection among the targets.
Figure 3. ‘Target–Target’ interaction network analysis. (A) The target–target interaction network is constructed by using Cytoscape 3.7.1. The size of the nodes is proportional to degrees. (B) The bar plot on degrees of connection among the targets. 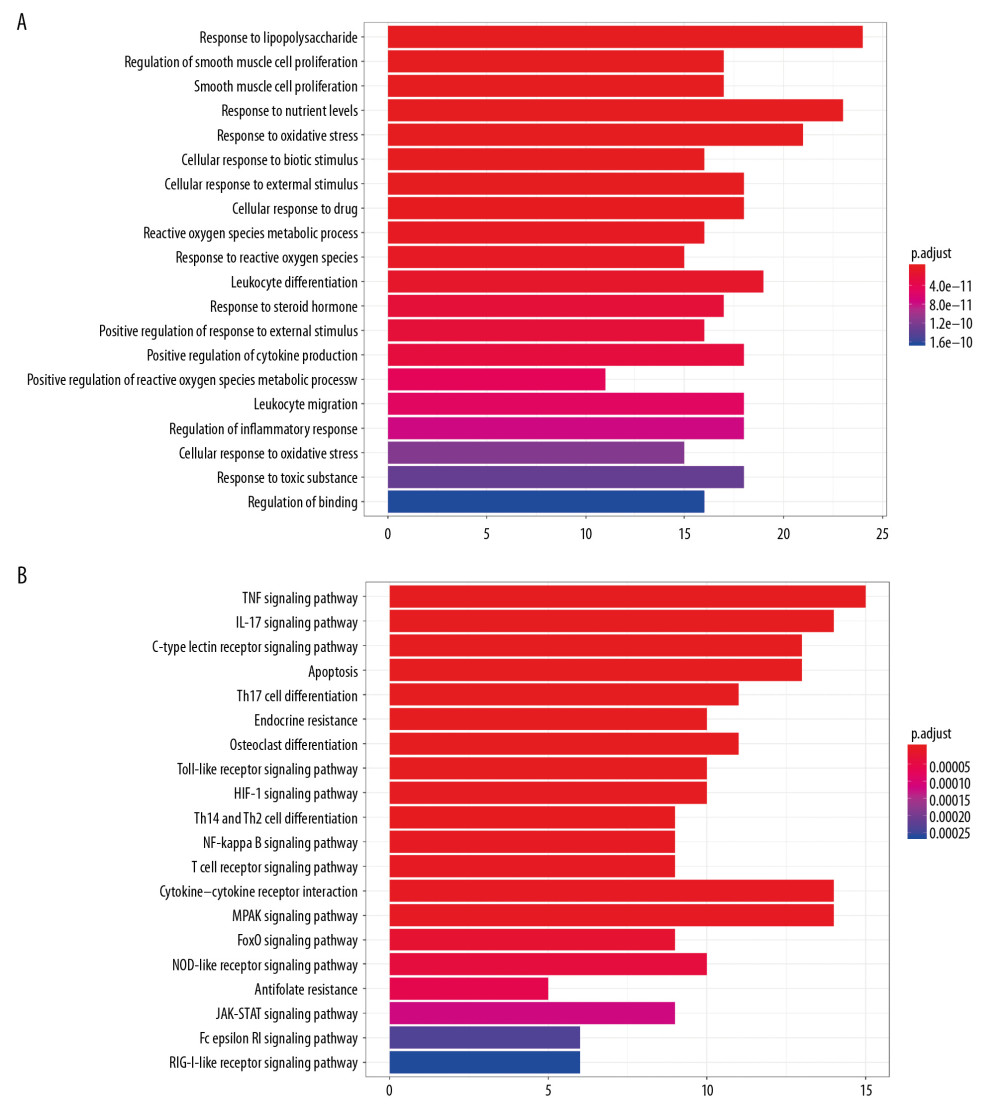 Figure 4. The bar plot of Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis. (A) The first 20 biological processes of GO analysis. (B) The first 20 signal pathways of KEGG enrichment analysis.
Figure 4. The bar plot of Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis. (A) The first 20 biological processes of GO analysis. (B) The first 20 signal pathways of KEGG enrichment analysis. 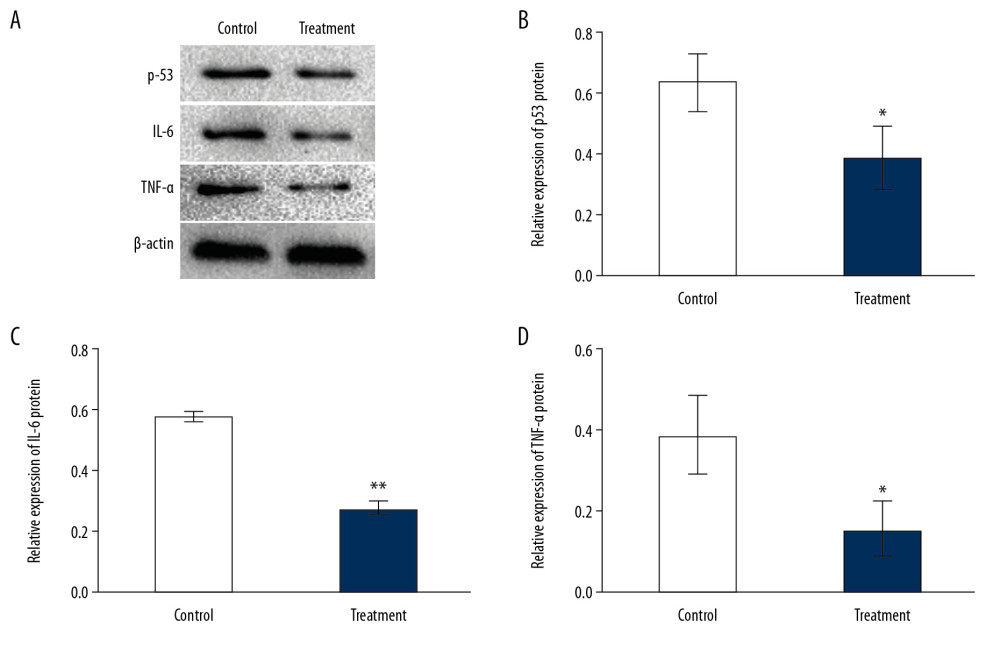 Figure 5. Expression of proteins in chondrocytes exposed to IL-1β and/or Achyranthes bidentata extract (A–D). * P<0.05 and ** P<0.01 vs. IL-1β-treated chondrocytes.
Figure 5. Expression of proteins in chondrocytes exposed to IL-1β and/or Achyranthes bidentata extract (A–D). * P<0.05 and ** P<0.01 vs. IL-1β-treated chondrocytes. References
1. Bijlsma JW, Berenbaum F, Lafeber FP, Osteoarthritis: An update with relevance for clinical practice: Lancet, 2011; 377(9783); 2115-26
2. Chahla J, Piuzzi NS, Mitchell JJ, Intra-articular cellular therapy for osteoarthritis and focal cartilage defects of the knee: A systematic review of the literature and study quality analysis: J Bone Joint Surg Am, 2016; 98(18); 1511-21
3. Taruc-Uy RL, Lynch SA, Diagnosis and treatment of osteoarthritis: Prim Care, 2013; 40(4); 821-36
4. He X, Wang X, Fang J, The genus Achyranthes: A review on traditional uses, phytochemistry, and pharmacological activities: J Ethnopharmacol, 2017; 203; 260-78
5. Li J, Wang C, Han X, Transcriptome analysis to identify the putative biosynthesis and transport genes associated with the medicinal components of achyranthes bidentata Bl: Front Plant Sci, 2016; 7; 1860
6. Ma D, Li Y, Xiao W, Achyranthes bidentata extract protects chondrocytes functions through suppressing glycolysis and apoptosis via MAPK/AKT signaling axis: Am J Transl Res, 2020; 12(1); 142-52
7. Weng X, Lin P, Liu F, Achyranthes bidentata polysaccharides activate the Wnt/beta-catenin signaling pathway to promote chondrocyte proliferation: Int J Mol Med, 2014; 34(4); 1045-50
8. Hopkins AL, Network pharmacology: The next paradigm in drug discovery: Nat Chem Biol, 2008; 4(11); 682-90
9. Yue SJ, Xin LT, Fan YC, Herb pair Danggui-Honghua: Mechanisms underlying blood stasis syndrome by system pharmacology approach: Sci Rep, 2017; 7; 40318
10. Xiong Y, Yang Y, Xiong W: J Cell Biochem, 2019; 120(8); 12461-72
11. Ma D, Yu T, Peng L: Evid Based Complement Alternat Med, 2019; 2019 1205942
12. Ru J, Li P, Wang J, TCMSP: A database of systems pharmacology for drug discovery from herbal medicines: J Cheminform, 2014; 6; 13
13. Cabrera-Pérez M, Pham-The H, Computational modeling of human oral bioavailability: What will be next?: Expert Opin Drug Discov, 2018; 13(6); 509-21
14. Tao W, Xu X, Wang X, Network pharmacology-based prediction of the active ingredients and potential targets of Chinese herbal Radix Curcumae formula for application to cardiovascular disease: J Ethnopharmacol, 2013; 145(1); 1-10
15. Yue SJ, Liu J, Feng WW, System pharmacology-based dissection of the synergistic mechanism of huangqi and huanglian for diabetes mellitus: Front Pharmacol, 2017; 8; 694
16. Kuhn M, Szklarczyk D, Franceschini A, STITCH 3: Zooming in on protein-chemical interactions: Nucleic Acids Res, 2012; 40(Database issue); D876-80
17. Carvalho-Silva D, Pierleoni A, Pignatelli M, Open Targets Platform: New developments and updates two years on: Nucleic Acids Res, 2019; 47(D1); D1056-65
18. Kafkas Ş, Dunham I, McEntyre J, Literature evidence in open targets – a target validation platform: J Biomed Semantics, 2017; 8(1); 20
19. Shannon P, Markiel A, Ozier O, Cytoscape: A software environment for integrated models of biomolecular interaction networks: Genome Res, 2003; 13(11); 2498-504
20. von Mering C, Jensen LJ, Snel B, STRING: known and predicted protein-protein associations, integrated and transferred across organisms: Nucleic Acids Res, 2005; 33(Database issue); D433-37
21. Yu G, Wang LG, Han Y, He QY, clusterProfiler: An R package for comparing biological themes among gene clusters: OMICS, 2012; 16(5); 284-87
22. Huntley RP, Harris MA, Alam-Faruque Y, A method for increasing expressivity of Gene Ontology annotations using a compositional approach: BMC Bioinformatics, 2014; 15; 155
23. Ashburner M, Ball CA, Blake JA, Gene ontology: Tool for the unification of biology. The Gene Ontology Consortium: Nat Genet, 2000; 25(1); 25-29
24. Kanehisa M, Furumichi M, Tanabe M, KEGG: New perspectives on genomes, pathways, diseases and drugs: Nucleic Acids Res, 2017; 45(D1); D353-61
25. Science Mo, China TotPsRo: Guidance suggestions for the care and use of laboratory animals, 2006, China, Ministry of Science and Technology of P.R
26. Li X, Du M, Liu X, Millimeter wave treatment inhibits NO-induced apoptosis of chondrocytes through the p38MAPK pathway: Int J Mol Med, 2010; 25(3); 393-99
27. Li XH, Wu MX, Ye HZ, Experimental study on the suppression of sodium nitroprussiate-induced chondrocyte apoptosis by Tougu Xiaotong Capsule – containing serum: Chin J Integr Med, 2011; 17(6); 436-43
28. Sanchez C, Mathy-Hartert M, Deberg MA, Effects of rhein on human articular chondrocytes in alginate beads: Biochem Pharmacol, 2003; 65(3); 377-88
29. Wu SQ, Otero M, Unger FM, Anti-inflammatory activity of an ethanolic Caesalpinia sappan extract in human chondrocytes and macrophages: J Ethnopharmacol, 2011; 138(2); 364-72
30. Jiang Y, Zhang Y, Chen W, Achyranthes bidentata extract exerts osteoprotective effects on steroid-induced osteonecrosis of the femoral head in rats by regulating RANKL/RANK/OPG signaling: J Transl Med, 2014; 12; 334
31. Lin J, Wu G, Chen J, Electroacupuncture inhibits sodium nitroprusside-mediated chondrocyte apoptosis through the mitochondrial pathway: Mol Med Rep, 2018; 18(6); 4922-30
32. Zheng YZ, Deng G, Liang Q, Antioxidant activity of quercetin and its glucosides from propolis: A theoretical study: Sci Rep, 2017; 7(1); 7543
33. Meng LQ, Yang FY, Wang MS, Quercetin protects against chronic prostatitis in rat model through NF-κB and MAPK signaling pathways: Prostate, 2018; 78(11); 790-800
34. Na JY, Song K, Kim S, Kwon J, Rutin protects rat articular chondrocytes against oxidative stress induced by hydrogen peroxide through SIRT1 activation: Biochem Biophys Res Commun, 2016; 473(4); 1301-8
35. Li L, Liu H, Shi W, Insights into the action mechanisms of Traditional Chinese Medicine in osteoarthritis: Evid Based Complement Alternat Med, 2017; 2017 5190986
36. Ha TK, Hansen AH, Kol S, Baicalein reduces oxidative stress in CHO cell cultures and improves recombinant antibody productivity: Biotechnol J, 2018; 13(3); e1700425
37. Xing D, Gao H, Liu Z, Baicalin inhibits inflammatory responses to Interleukin-1β stimulation in human chondrocytes: J Interferon Cytokine Res, 2017; 37(9); 398-405
38. Kumar A, Ekavali , Chopra K, Current knowledge and pharmacological profile of berberine: An update: Eur J Pharmacol, 2015; 761; 288-97
39. Li X, He P, Hou Y, Berberine inhibits the interleukin-1 beta-induced inflammatory response via MAPK downregulation in rat articular chondrocytes: Drug Dev Res, 2019; 80(5); 637-45
40. Hussein MR, Fathi NA, El-Din AM, Alterations of the CD4(+), CD8 (+) T cell subsets, interleukins-1beta, IL-10, IL-17, tumor necrosis factor-alpha and soluble intercellular adhesion molecule-1 in rheumatoid arthritis and osteoarthritis: Preliminary observations: Pathol Oncol Res, 2008; 14(3); 321-28
41. Bjornsson GL, Thorsteinsson L, Gudmundsson KO, Inflammatory cytokines in relation to adrenal response following total hip replacement: Scand J Immunol, 2007; 65(1); 99-105
42. Li J, Lu D, Dou H, Publisher correction: Desumoylase SENP6 maintains osteochondroprogenitor homeostasis by suppressing the p53 pathway: Nat Commun, 2018; 9(1); 646
43. Lin M, Lin Y, Li X, Warm sparse-dense wave inhibits cartilage degradation in papain-induced osteoarthritis through the mitogen-activated protein kinase signaling pathway: Exp Ther Med, 2017; 14(4); 3674-80
44. Zhou Z, Chen B, Chen S, Applications of network pharmacology in Traditional Chinese Medicine research: Evid Based Complement Alternat Med, 2020; 2020 1646905
Figures
 Figure 1. Study flow chart.
Figure 1. Study flow chart. Figure 2. Venn analysis and network analysis of active compounds and potential targets of Achyranthes bidentata on OA. (A) The Venn diagram of target proteins of Achyranthes bidentata and OA-related targets based on the Open Targets Platform. (B) The compound-targets network diagram of Achyranthes bidentata to treat OA, the square nodes represent compounds. The circular nodes represent target proteins.
Figure 2. Venn analysis and network analysis of active compounds and potential targets of Achyranthes bidentata on OA. (A) The Venn diagram of target proteins of Achyranthes bidentata and OA-related targets based on the Open Targets Platform. (B) The compound-targets network diagram of Achyranthes bidentata to treat OA, the square nodes represent compounds. The circular nodes represent target proteins. Figure 3. ‘Target–Target’ interaction network analysis. (A) The target–target interaction network is constructed by using Cytoscape 3.7.1. The size of the nodes is proportional to degrees. (B) The bar plot on degrees of connection among the targets.
Figure 3. ‘Target–Target’ interaction network analysis. (A) The target–target interaction network is constructed by using Cytoscape 3.7.1. The size of the nodes is proportional to degrees. (B) The bar plot on degrees of connection among the targets. Figure 4. The bar plot of Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis. (A) The first 20 biological processes of GO analysis. (B) The first 20 signal pathways of KEGG enrichment analysis.
Figure 4. The bar plot of Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis. (A) The first 20 biological processes of GO analysis. (B) The first 20 signal pathways of KEGG enrichment analysis. Figure 5. Expression of proteins in chondrocytes exposed to IL-1β and/or Achyranthes bidentata extract (A–D). * P<0.05 and ** P<0.01 vs. IL-1β-treated chondrocytes.
Figure 5. Expression of proteins in chondrocytes exposed to IL-1β and/or Achyranthes bidentata extract (A–D). * P<0.05 and ** P<0.01 vs. IL-1β-treated chondrocytes. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952