02 September 2020: Clinical Research
Value of Angioembolization in the Treatment of Iatrogenic Renal Vascular Injury Assisted by 3-Dimensional Digital Subtraction Angiography
Xin Liao1ABCDEFG, Hao Xu2ACDEF*, Fan Liu2BCF, Xuli Min2BCD, Yugen Li3BCDF, Lin Yang2DF, Yongjun Ren2CFDOI: 10.12659/MSM.927208
Med Sci Monit 2020; 26:e927208
Abstract
BACKGROUND: This study was conducted to investigate the reliability and efficacy of polyvinyl alcohol combined with coils in the embolization of iatrogenic renal vascular injury with the assistance of 3-dimensional digital subtraction angiography (3D-DSA).
MATERIAL AND METHODS: Twenty-six patients with minimally invasive renal bleeding who underwent transarterial embolization from January 2012 to January 2019 in our hospital were included in the study. We obtained demographic data from these patients, as well as information on clinical presentation, renal procedures used for treatment, and perioperative details. The changes in renal function tests, serum hemoglobin, serum hematocrit, and technetium Tc 99m dimercaptosuccinic acid (99mTc-DMSA) levels pre- and postembolization were compared. In addition, the embolic area and the technical and clinical success rates were analyzed. Finally, an angiographic manifestation of the renal artery, 3D-DSA, and the effect of embolization were analyzed retrospectively.
RESULTS: All patients achieved technical and clinical success after embolization (100%, 26/26). There were no significant differences between pre- and postoperative estimated glomerular filtration rate, serum parameters, and 99mTc-DMSA. The embolic area was 12%±10%. Patients did not exhibit severe complications during the follow-up period.
CONCLUSIONS: Proximal embolization technique assisted by 3D-DSA for renal iatrogenic hemorrhage and vascular lesions is both safe and efficacious, offering high rates for both clinical and technical success. It maximizes the protection of the kidney and reduces the rate of renal resection.
Keywords: Angiography, Digital Subtraction, Embolization, Therapeutic, Iatrogenic Disease, Nephrostomy, Percutaneous, Polyvinyl Alcohol, Reproducibility of Results, Vascular System Injuries
Background
Renal vascular injury can occur secondary to iatrogenic, blunt, or penetrating injuries. Most renal injuries are self-limiting, but massive bleeding or persistent hematuria requires active treatment [1]. Percutaneous nephrolithotomy (PCNL) and percutaneous nephrostomy (PN) are the routine treatments for urolithiasis, but they have a high incidence of causing iatrogenic injury [2,3]. Hematuria is a severe life-threatening complication of these procedures, requiring timely treatment. Temporary clipping of the nephrostomy tube, application of hemostatic drugs, and blood transfusion are necessary conservative treatment measures [4]. It is recommended that angiography and embolization be performed by conservative treatment as early as possible in patients with severe hemorrhage [5–7].
Renal artery embolization is frequently employed to embolize tumors, especially renal angiomyolipoma, that might lead to large areas of embolization due to embolic agent migration [8]. With the development of the newer materials, a subsegmental artery is catheterized with a microcatheter and occluded to reduce the area of embolization and protect renal function as much as possible [9]. In this study, we sought to assess the reliability and efficacy of polyvinyl alcohol (PVA) combined with coil embolization of iatrogenic renal vascular injury with the assistance of 3-dimensional digital subtraction angiography (3D-DSA).
Material and Methods
PATIENTS AND DATA COLLECTION:
A retrospective study was performed in our hospital. The Institutional Ethical Committee granted ethical clearance, and the procedures were consistent with ethical standards (File Number 2019 ER(A)03-012). Patients admitted with hemorrhage after minimally invasive renal surgery who underwent transcatheter arterial embolization between January 2012 and January 2019 were included in the study. Angiography and embolization included vascular injuries, intraoperative blood loss that could not be controlled, and imaging findings consistent with injuries to the vasculature, such as pseudoaneurysm formation, active contrast medium extravasation, and arteriovenous fistula (AVF) formation.
Medical records and imaging findings of 26 patients, including demographics (age and sex), type of minimally invasive renal procedures (PCNL or PN), clinical symptoms, angiographic features, technical success, clinical success, complications, renal function, serum hemoglobin (Hb), and serum hematocrit (Htc) were reviewed. Assessment of renal function included the estimated glomerular filtration rate (eGFR) and the serum urea and serum creatinine levels. Technetium Tc 99m dimercaptosuccinic acid (99mTc-DMSA) is a commonly used radiopharmaceutical and is equally reliable in calculating relative renal function. Patients gave informed consent before participation.
ANGIOGRAPHY AND EMBOLIZATION:
The Seldinger technique was used to puncture the femoral artery and insert a 5F catheter sheath percutaneously. Abdominal aortography was performed with a pigtail catheter to analyze the direction, diameter, and variation of the renal artery. Then, renal arteriography was performed by introducing a Yashiro, Simons 1, or Cobra catheter (Terumo or Cordis Company) according to the condition of the renal artery. Patients were properly positioned, and 3D-DSA and volume rendering (VR) were performed. The location of the artery suspected of bleeding, the extent of bleeding, pseudoaneurysm, and AVF were found under DSA and VR. A 2.5F microcatheter (Renegade™ STC18, Boston Science, USA) was performed on arteries with suspected bleeding to identify active hemorrhage (contrast medium overflow) and vascular lesions by angiography. The proximal embolization technique involves blocking the proximal end of vascular lesions with different embolic agents. The types of embolic materials are determined according to the size and location of the ruptured branches of the internal renal artery, the diameter of the pseudoaneurysmal parent artery, and the flow rate of the AVF. PVA particles (100, 300, and 500 μm; COOK Company, USA) were used as the embolic material. Tower-like or diamond-like embolic coils (Boston VortX™-18 platinum coil or VortX™-18 Diamond platinum coil) that were consistent with or slightly larger than the diameter of the diseased artery were selected for embolization. In the case of a simple active hemorrhage without AVF, a small amount of PVA was injected into the bleeding artery first, and then coil embolization was decided according to the repeat angiography. If hemorrhage and pseudoaneurysm were found by angiography, a few PVA particles were injected into the hemorrhagic artery and suitable size coils were placed into the parent artery. If AVF existed, coils were used to reduce the flow rate and then a few PVA particles were embolized.
POSTEMBOLIZATION OBSERVATION:
Technical and clinical success are the primary endpoints of embolization. The definitions and evaluation criteria of the efficacy and technical and clinical success of embolization were according to the Society of Interventional Radiology guidelines. Technical success was determined by the resolution of hemorrhage, the disappearance of pseudoaneurysm or AVF, or the occlusion of the parent artery. Clinical success was defined as the disappearance of hematuria, without recurrence; no requirement for blood transfusion; no further decrease in Hb; and no requirement for repeat angiography, embolization, or renal surgery [10]. Technical complications included nontarget vascular embolism, iatrogenic vascular injury, and puncture site bleeding. Clinical complications included posterior embolism syndrome (back pain and fever), perirenal or renal abscess, renal dysfunction, and renal artery damage [3,9,11]. Follow-up included physical examination, computed tomography (CT) or magnetic resonance imaging (MRI), and serum renal indexes. Follow-up (6 months) consisted mainly of serum renal function evaluation of indicators, including eGFR, serum urea, Hb, Htc, and renal nuclear scintigraphy.
STATISTICAL ANALYSIS:
Pre- and postoperative eGFR and serum parameters of renal function are presented as means±standard deviation. Levels of eGFR, serum urea, creatinine, and Hb were compared before and after operations, with paired
Results
A total of 26 patients aged 52.23±12.71 years, including 7 women and 19 men, were included in the study. There were 21 cases of PCNL and 5 cases of PN. PN was performed in 5 patients due to hydronephrosis (n=4) and vesical invasion of rectal cancer (n=1). The clinical manifestations of 26 patients were gross hematuria, back pain, and increased drainage fluid. Six patients had multiple sites of bleeding (Figure 1); 19 patients had pseudoaneurysms, as exemplified in a representative case with arteriography images provided in Figure 2; and 1 patient had pseudoaneurysms with renal arteriovenous fistula (Figure 3). All vessels involved were the subsegmental artery, including right posterior (n=12), left posterior (n=8), right inferior (n=4), and left inferior subsegmental artery (n=2). Three patients stopped bleeding after embolization with PVA, and the other 23 patients were embolized with PVA and coil.
With the kidney viewed as a cylinder, the embolic volume was calculated. The embolic area was 12%+10%. Postembolization syndrome, including back pain, fever, or both, were detected in 10 patients (38%, 10/26), and 4 had pain and fever (15%, 4/26). Although the patients had pain and other discomfort after embolization, they were relieved by antipyretic and analgesic drugs, and no serious complications occurred during the follow-up period. All patients achieved technical and clinical success after embolization (100%, 26/26). After discharge from hospital, 25 patients (1 was lost to follow-up) were followed for 6 or more months. No bleeding recurred, and no patients exhibited decreases in renal function or other complications (Table 1). Pre- and postoperative values of eGFR, serum urea, creatinine, Hb, Htc, and relative renal function values did not differ significantly (Table 2). The characteristics of the 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula are presented in Table 3.
Discussion
Iatrogenic renal vascular injury, which needs to be recognized and treated in a timely manner to reduce morbidity and mortality, is mainly caused by PCNL, PN, percutaneous renal biopsy, tumor surgery, and other surgeries [2]. The clinical and pathological manifestations are AVF, arteriocaliceal fistula, pseudoaneurysm, or perirenal hematomas. PCNL and PN are effective methods to improve the symptoms and treatment of renal and ureteral calculi, but hemorrhage is the most common clinical complication and its incidence is high [2,3]. In most patients, bleeding after the procedures is controlled by conservative treatment, but severe bleeding requires emergency management. The risk factors for severe bleeding are huge stones, lower puncture site, number and type of stones, operative history of ipsilateral renal stones, intraoperative perforation of the renal pelvis, and no tube drainage [4]. At present, there is no standard protocol for risk assessment of postoperative complications after PCNL and PN.
For patients with hematuria after surgery, CT or MRI examination can be performed to determine whether there is active blood extravasation and also to evaluate the vascular injury in the arterial phase [12–14]. However, negative results cannot wholly exclude vascular lesions [13]. Ultrasonography is not recommended as a routine examination because of its close relationship with the operator, but dilated veins of AVF and perirenal or subcapsular hematomas can be observed [15]. DSA is the gold standard for the diagnosis of vascular diseases. It not only shows the lesions but also provides endovascular treatment during the examination. In this study, patients were evaluated with DSA as the first imaging method because they needed emergency angiography and embolization. 3D-DSA, which has been widely used in clinical settings, especially in cerebral angiography and reconstruction technique (especially VR), can be used for multi-angle observation of the parent artery of the pseudoaneurysm, AVF, and other factors, to provide more anatomical information for the selection of embolic materials. It can be used as a supplementary inspection method for DSA and CT angiography. When DSA reveals the hemorrhagic artery to be complex, 3D-DSA is performed. Routine 3D-DSA is not recommended to undergo to avoid unnecessary radiation exposure.
Transarterial embolization is considered the treatment of choice for severe bleeding after low-grade renal trauma, including iatrogenic renal vascular injury [6,16]. However, there is no standard consensus on how and when interventional therapy should be performed. Conclusions from various studies [17–20] indicate that transcatheter arterial embolization could be used for the treatment of patients with gross hematuria when conservative treatment is ineffective; if CT angiography finds direct bleeding, pseudoaneurysm, or AVF; and when patients have a large hematoma (>25-mm depth).
A proximal embolization technique is used to exclude the lesion from the renal circulation without compromising the vascular supply to the rest of the kidney [21]. This technique is widely used in clinical practice with various embolic materials, such as particulate, solid, and liquid embolic materials. Material selection should be based on the location, diameter, and blood flow velocity of the artery. For patients with coagulopathy, coils might not embolize thoroughly, and multiple coils might be required to achieve occlusion. It might lead to increasing rate of repeat embolization, cost, and surgical time [22,23]. PVA is inexpensive and insert, has excellent biocompatibility and irregular particle characteristics, and was previously considered as a permanent embolic material; however, histologically, the particles are embedded in the vessel wall and most of the large vessels embolized by PVA are not completely occluded. PVA along with proximal coil embolization could make up for the insufficiency of embolization caused by a single embolic material and enhance the embolization effect because both the cilia of the coil and PVA particles make platelets aggregate quickly [23]. PVA embolization was performed in patients with multiple dot-like contrast media extravasation, and angiography was performed after 3 and 8 minutes. If the signs of bleeding disappeared, technical success was considered to have been achieved. The direct application of coil embolization is believed to achieve higher technical success and maximize the protection of the kidney [24]. Because of the high risk of rupture, pseudoaneurysm was treated by embolization as soon as possible, and a few PVA particles were injected into the proximal end of the parent artery. To enable direct embolization, percutaneous puncture of the pseudoaneurysm may represent an alternative approach for instances in which difficulties are encountered during endovascular management [11,22,25]. Some researchers have used different embolic materials, such as ethanol, Onyx, N-butyl cyanoacrylate (NBCA), Glubran-2, and other liquid embolic materials, and achieved high technical and clinical success rate, but there is a potential risk of regurgitant misembolization, and only an experienced interventional physician should perform the operation [22,23,26]. It is not recommended in emergency conditions [26]. Embolization of AVF and other lesions first avoids the risk of anterograde misembolization. In our cohort, the coil was applied to reduce the flow rate and then larger diameter PVA particles, which can reduce the risk of misembolization and make embolization more thorough, were introduced.
The injury after PCNL and PN, which may only show puncture injury, was significantly less than that of traumatic renal injury. Microcatheter catheterization is commonly used for the diseased artery. The embolic volume should be reduced as much as possible to ensure the curative effect. In this group of cases, the average embolic volume was about 12%, which had a nonsignificant effect on renal function and was lower than that reported by Kervancioglu et al. (12%
This study was limited by its retrospective nature and small sample size, as it was difficult to conduct a prospective randomized clinical trial with a large number of patients. Hence, more randomized controlled trials should be conducted in future enrolling large number of patients.
Conclusions
The proximal embolization technique assisted by 3D-DSA is safe and efficacious for treating iatrogenic hemorrhage and vascular lesions of the kidney. It has high rates of technical and clinical success, which can protect the kidney to the greatest extent and reduce the rate of nephrectomy.
Figures
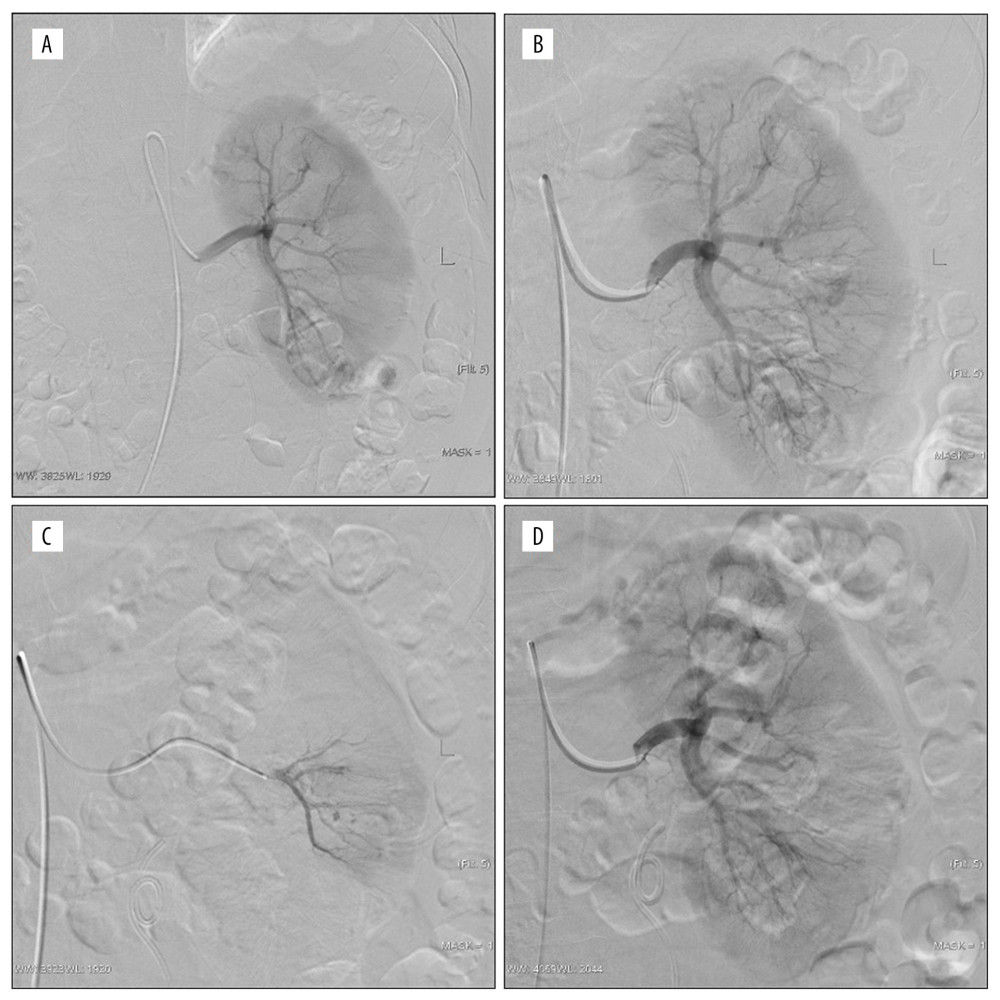 Figure 1. Active bleeding areas and persistent hematuria after the left percutaneous nephrolithotomy (PCNL). (A) No definite abnormality; (B, C) multiple spikes of contrast media in puncture tract were observed by magnification angiography and superselective anterior inferior segment angiography; (D) postembolized by 300 μm polyvinyl alcohol, the embolization volume was about 15%.
Figure 1. Active bleeding areas and persistent hematuria after the left percutaneous nephrolithotomy (PCNL). (A) No definite abnormality; (B, C) multiple spikes of contrast media in puncture tract were observed by magnification angiography and superselective anterior inferior segment angiography; (D) postembolized by 300 μm polyvinyl alcohol, the embolization volume was about 15%. 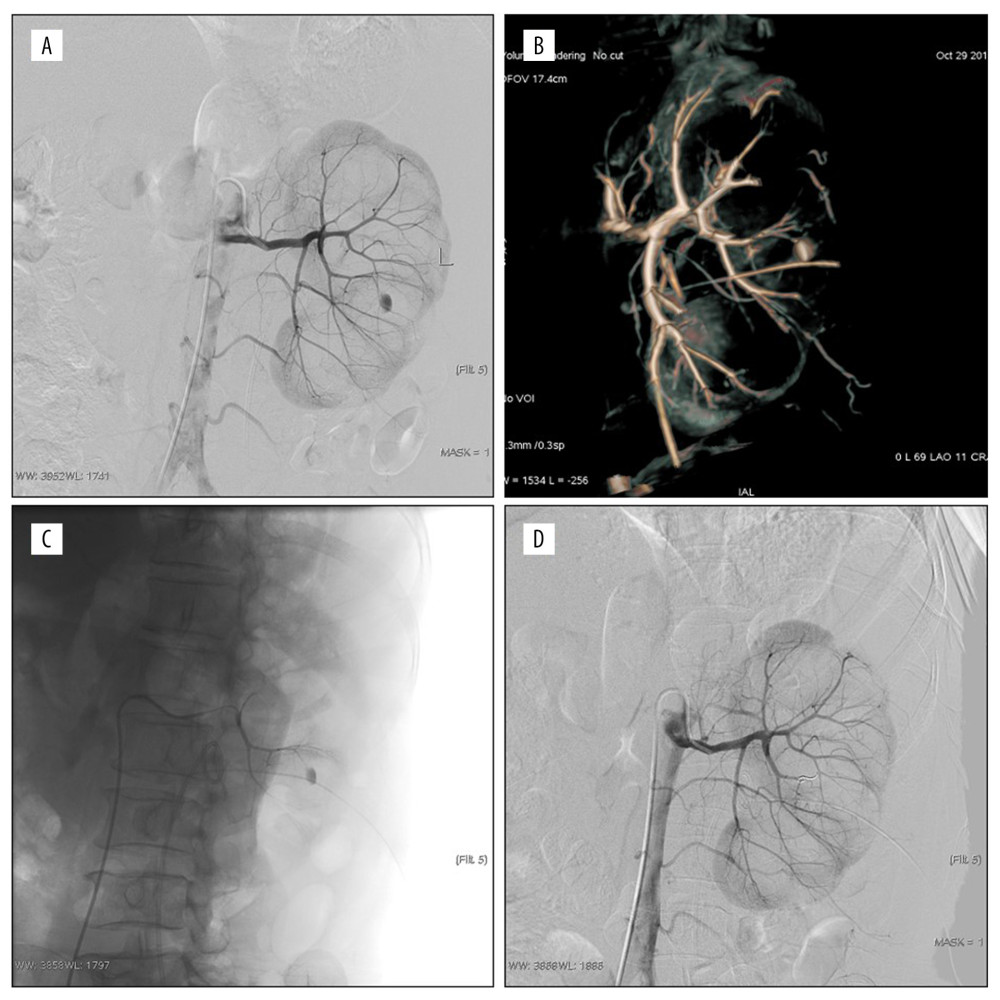 Figure 2. Persistent hematuria after the left percutaneous nephrolithotomy (PCNL); (A, B) Pseudoaneurysm of posterior segment artery was formed, digital subtraction angiography and volume rendering showed the origin of the parent artery; (C) Superselective to parent aneurysm angiography; (D) post-embolized by 100 μm polyvinyl alcohol and the coil. The pseudoaneurysm disappeared and the embolization volume was about 5%.
Figure 2. Persistent hematuria after the left percutaneous nephrolithotomy (PCNL); (A, B) Pseudoaneurysm of posterior segment artery was formed, digital subtraction angiography and volume rendering showed the origin of the parent artery; (C) Superselective to parent aneurysm angiography; (D) post-embolized by 100 μm polyvinyl alcohol and the coil. The pseudoaneurysm disappeared and the embolization volume was about 5%. 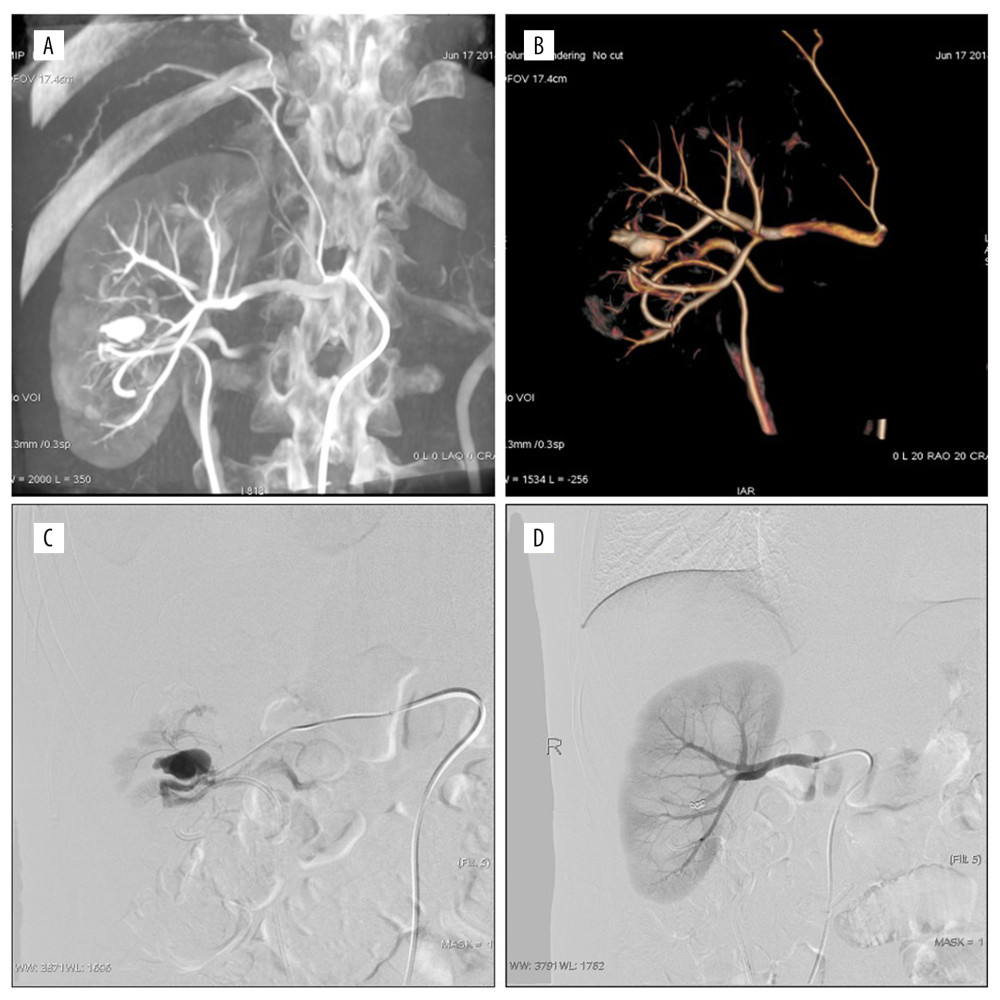 Figure 3. Persistent hematuria after the right percutaneous nephrolithotomy (PCNL). (A, B) Anterior inferior artery pseudoaneurysm (PA) and renal arteriovenous fistula (RAVF). Digital subtraction angiography and volume rendering showed blood supply artery and drainage vein; (C) The flow velocity of AVF was judged by superselective arteriography; (D) postembolized by the coil and 300 μm polyvinyl alcohol. The PA and RAVF disappeared and the embolization volume was about 15%.
Figure 3. Persistent hematuria after the right percutaneous nephrolithotomy (PCNL). (A, B) Anterior inferior artery pseudoaneurysm (PA) and renal arteriovenous fistula (RAVF). Digital subtraction angiography and volume rendering showed blood supply artery and drainage vein; (C) The flow velocity of AVF was judged by superselective arteriography; (D) postembolized by the coil and 300 μm polyvinyl alcohol. The PA and RAVF disappeared and the embolization volume was about 15%. Tables
Table 1. Details of the distribution of the lesions and outcomes of their intervention (n=26).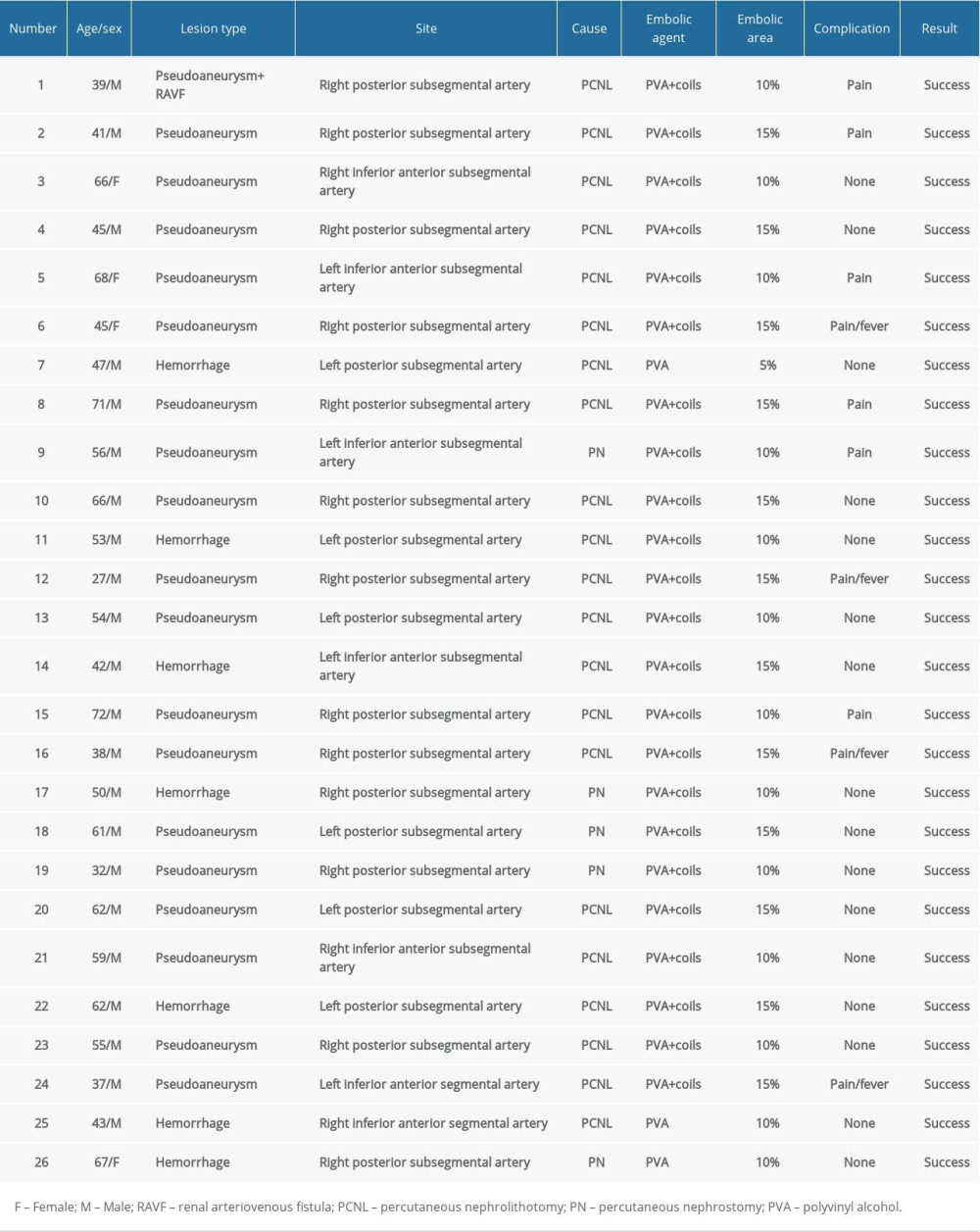 Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA).
Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA).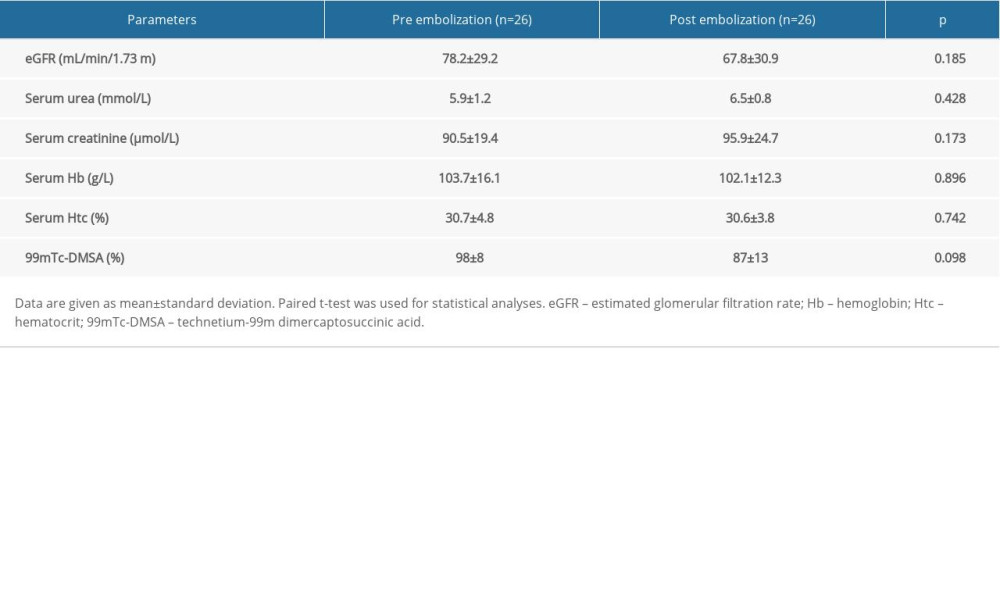 Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula.
Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula.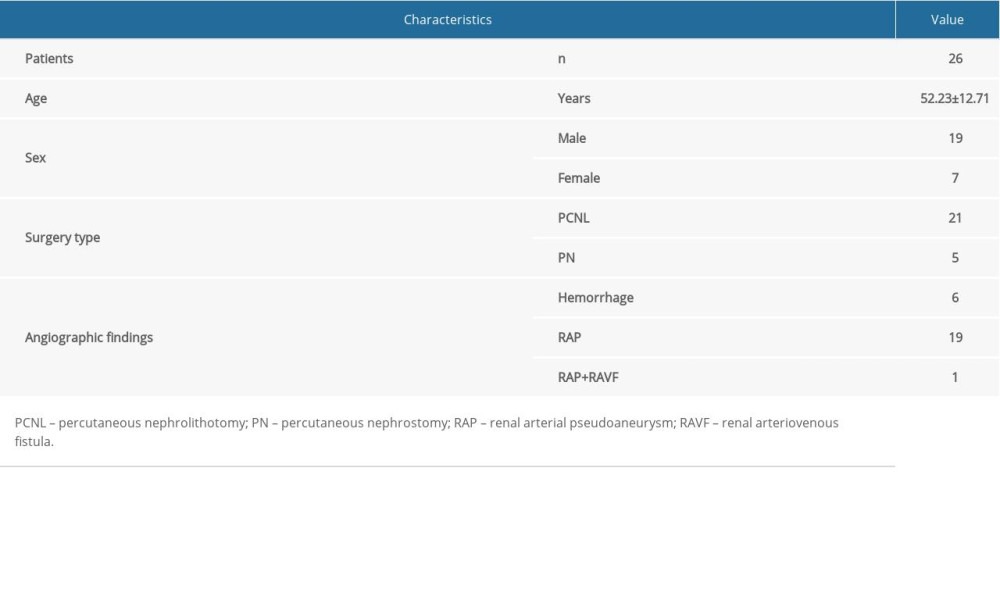
References
1. Chouhan JD, Winer AG, Johnson C, Contemporary evaluation and management of renal trauma: Can J Urol, 2016; 23; 8191-97
2. Güneyli S, Gök M, Bozkaya H, Endovascular management of iatrogenic renal arterial lesions and clinical outcomes: Diagn Interv Radiol, 2015; 21; 229-34
3. Summerton DJ, Kitrey ND, Lumen N, EAU guidelines on iatrogenic trauma: Eur Urol, 2012; 62; 628-39
4. Du N, Ma J, Luo J, The efficacy and safety of transcatheter arterial embolization to treat renal hemorrhage after percutaneous nephrolithotomy: Biomed Res Int, 2019; 2019 6265183
5. Sam K, Gahide G, Soulez G, Percutaneous embolization of iatrogenic arterial kidney injuries: safety, efficacy, and impact on blood pressure and renal function: J Vasc Interv Radiol, 2011; 22; 1563-68
6. Barbiero G, Groff S, Battistel M, Are iatrogenic renal artery pseudoaneurysms more challenging to embolize when associated with an arteriovenous fistula?: Radiol Med, 2018; 123; 742-52
7. Loffroy R, Chevallier O, Gehin S, Endovascular management of arterial injuries after blunt or iatrogenic renal trauma: Quant Imaging Med Surg, 2017; 7; 434-42
8. Schwartz MJ, Smith EB, Trost DW, Vaughan ED, Renal artery embolization: Clinical indications and experience from over 100 cases: BJU Int, 2007; 99; 881-86
9. Jacobson AI, Amukele SA, Marcovich R, Efficacy and morbidity of therapeutic renal embolization in the spectrum of urologic disease: J Endourol, 2003; 17; 385-91
10. Angle JF, Siddiqi NH, Wallace MJ, Quality improvement guidelines for percutaneous transcatheter embolization: Society of Interventional Radiology Standards of Practice Committee: J Vasc Interv Radiol, 2010; 21; 1479-86
11. Ginat DT, Saad WE, Turba UC, Transcatheter renal artery embolization: Clinical applications and techniques: Tech Vasc Interv Radiol, 2009; 12; 224-39
12. Sommer CM, Stampfl U, Bellemann N, Patients with life-threatening arterial renal hemorrhage: CT angiography and catheter angiography with subsequent superselective embolization: Cardiovasc Intervent Radiol, 2010; 33; 498-508
13. Morey AF, Brandes S, Dugi DR, Urotrauma: AUA guideline: J Urol, 2014; 192; 327-35
14. Sauk S, Zuckerman DA, Renal artery embolization: Semin Intervent Radiol, 2011; 28; 396-406
15. Lewis S, Patel U, Major complications after percutaneous nephrostomy – lessons from a department audit: Clin Radiol, 2004; 59; 171-79
16. Song P, Wang MQ, Liu FY, Iatrogenic renovascular injuries treated by transarterial embolization: Eur Rev Med Pharmacol Sci, 2013; 17; 3398-404
17. Leppaniemi A, Lamminen A, Tervahartiala P, MRI and CT in blunt renal trauma: An update: Semin Ultrasound CT MR, 1997; 18; 129-35
18. Lin WC, Lin CH, Chen JH, Computed tomographic imaging in determining the need of embolization for high-grade blunt renal injury: J Trauma Acute Care Surg, 2013; 74; 230-35
19. Charbit J, Manzanera J, Millet I, What are the specific computed tomography scan criteria that can predict or exclude the need for renal angioembolization after high-grade renal trauma in a conservative management strategy?: J Trauma, 2011; 70; 1219-28
20. Nouralizadeh A, Ziaee SA, Hosseini SS, Delayed postpercutaneous nephrolithotomy hemorrhage: Prevalence, predictive factors and management: Scand J Urol, 2014; 48; 110-15
21. Venkateswarlu J, Kumar MS, Babu RP, Abkari A, Endovascular management of iatrogenic renal vascular injuries complicating percutaneous nephrolithotomy: Role of renal angiography and embolization; An analysis of 159 cases: Indian J Radiol Imaging, 2017; 27(3); 293-97
22. Cimsit NC, Baltacioglu F, Cengic I, Transarterial glue embolization in iatrogenic renovascular injuries: Int Urol Nephrol, 2008; 40; 875-79
23. Wang D, Su L, Fan X, Cardiovascular collapse and disseminated intravascular coagulation as complications of ethanol embolization of arteriovenous malformations in the upper lip: Case report and literature review: J Oral Maxillofac Surg, 2014; 72; 346-51
24. Muller A, Rouviere O, Renal artery embolization-indications, technical approaches and outcomes: Nat Rev Nephrol, 2015; 11; 288-301
25. Sakr MA, Desouki SE, Hegab SE, Direct percutaneous embolization of renal pseudoaneurysm: J Endourol, 2009; 23; 875-78
26. Cantasdemir M, Adaletli I, Cebi D, Emergency endovascular embolization of traumatic intrarenal arterial pseudoaneurysms with N-butyl cyanoacrylate: Clin Radiol, 2003; 58; 560-65
27. Kervancioglu S, Gelebek Yilmaz F, Erturhan S, Endovascular management of vascular complications after percutaneous nephrolithotomy: Vasa, 2014; 43(6); 459-64
28. Guo H, Wang C, Yang M, Management of iatrogenic renal arteriovenous fistula and renal arterial pseudoaneurysm by transarterial embolization: Medicine, 2017; 96; e8187
29. Gahan JC, Gaitonde M, Wadskier L, Renal function outcomes following selective angioembolization for iatrogenic vascular lesions after partial nephrectomy: J Endourol, 2013; 27; 1516-19
30. El-Nahas AR, Shokeir AA, Mohsen T, Functional and morphological effects of postpercutaneous nephrolithotomy superselective renal angiographic embolization: Urology, 2008; 71; 408-12
31. Oderich GS, Panneton JM, Hofer J, Iatrogenic operative injuries of abdominal and pelvic veins: a potentially lethal complication: J Vasc Surg, 2004; 39; 931-36
32. Vora A, Brodsky R, Nolan J, Incidence of postembolization syndrome after complete renal angioinfarction: A single-institution experience over four years: Scand J Urol, 2014; 48; 245-51
33. Collins CS, Eggert CH, Stanson AJ, Garovic VD, Long-term follow-up of renal function and blood pressure after selective renal arterial embolization: Perspect Vasc Surg Endovasc Ther, 2010; 22; 254-60
Figures
 Figure 1. Active bleeding areas and persistent hematuria after the left percutaneous nephrolithotomy (PCNL). (A) No definite abnormality; (B, C) multiple spikes of contrast media in puncture tract were observed by magnification angiography and superselective anterior inferior segment angiography; (D) postembolized by 300 μm polyvinyl alcohol, the embolization volume was about 15%.
Figure 1. Active bleeding areas and persistent hematuria after the left percutaneous nephrolithotomy (PCNL). (A) No definite abnormality; (B, C) multiple spikes of contrast media in puncture tract were observed by magnification angiography and superselective anterior inferior segment angiography; (D) postembolized by 300 μm polyvinyl alcohol, the embolization volume was about 15%. Figure 2. Persistent hematuria after the left percutaneous nephrolithotomy (PCNL); (A, B) Pseudoaneurysm of posterior segment artery was formed, digital subtraction angiography and volume rendering showed the origin of the parent artery; (C) Superselective to parent aneurysm angiography; (D) post-embolized by 100 μm polyvinyl alcohol and the coil. The pseudoaneurysm disappeared and the embolization volume was about 5%.
Figure 2. Persistent hematuria after the left percutaneous nephrolithotomy (PCNL); (A, B) Pseudoaneurysm of posterior segment artery was formed, digital subtraction angiography and volume rendering showed the origin of the parent artery; (C) Superselective to parent aneurysm angiography; (D) post-embolized by 100 μm polyvinyl alcohol and the coil. The pseudoaneurysm disappeared and the embolization volume was about 5%. Figure 3. Persistent hematuria after the right percutaneous nephrolithotomy (PCNL). (A, B) Anterior inferior artery pseudoaneurysm (PA) and renal arteriovenous fistula (RAVF). Digital subtraction angiography and volume rendering showed blood supply artery and drainage vein; (C) The flow velocity of AVF was judged by superselective arteriography; (D) postembolized by the coil and 300 μm polyvinyl alcohol. The PA and RAVF disappeared and the embolization volume was about 15%.
Figure 3. Persistent hematuria after the right percutaneous nephrolithotomy (PCNL). (A, B) Anterior inferior artery pseudoaneurysm (PA) and renal arteriovenous fistula (RAVF). Digital subtraction angiography and volume rendering showed blood supply artery and drainage vein; (C) The flow velocity of AVF was judged by superselective arteriography; (D) postembolized by the coil and 300 μm polyvinyl alcohol. The PA and RAVF disappeared and the embolization volume was about 15%. Tables
 Table 1. Details of the distribution of the lesions and outcomes of their intervention (n=26).
Table 1. Details of the distribution of the lesions and outcomes of their intervention (n=26). Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA).
Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA). Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula.
Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula. Table 1. Details of the distribution of the lesions and outcomes of their intervention (n=26).
Table 1. Details of the distribution of the lesions and outcomes of their intervention (n=26). Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA).
Table 2. Comparison of pre- and postprocedural estimated glomerular filtration rate (eGFR), serum parameters, and technetium-99m dimercaptosuccinic acid (99mTc-DMSA). Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula.
Table 3. Characteristics of 26 patients undergoing transcatheter arterial embolization for iatrogenic renal arterial pseudoaneurysm/renal arteriovenous fistula. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








