01 January 2022: Clinical Research
A Statistical Prediction Model for Survival After Kidney Transplantation from Deceased Donors
Jia-shan Pan1AE, Yi-ding Chen1BC, Han-dong Ding1B, Tian-chi Lan1B, Fei Zhang1B, Jin-biao Zhong1A*, Gui-yi Liao1AFDOI: 10.12659/MSM.933559
Med Sci Monit 2022; 28:e933559
Abstract
BACKGROUND: In an environment of limited kidney donation resources, patient recovery and survival after kidney transplantation (KT) are highly important. We used pre-operative data of kidney recipients to build a statistical model for predicting survivability after kidney transplantation.
MATERIAL AND METHODS: A dataset was constructed from a pool of patients who received a first KT in our hospital. For allogeneic transplantation, all donated kidneys were collected from deceased donors. Logistic regression analysis was used to change continuous variables into dichotomous ones through the creation of appropriate cut-off values. A regression model based on the least absolute shrinkage and selection operator (LASSO) algorithm was used for dimensionality reduction, feature selection, and survivability prediction. We used receiver operating characteristic (ROC) analysis, calibration, and decision curve analysis (DCA) to evaluate the performance and clinical impact of the proposed model. Finally, a 10-fold cross-validation scheme was implemented to verify the model robustness.
RESULTS: We identified 22 potential variables from which 30 features were selected as survivability predictors. The model established based on the LASSO regression algorithm had shown discrimination with an area under curve (AUC) value of 0.690 (95% confidence interval: 0.557-0.823) and good calibration result. DCA demonstrated clinical applicability of the prognostic model when the intervention progressed to the possibility threshold of 2%. An average AUC value of 0.691 was obtained on the validation data.
CONCLUSIONS: Our results suggest that the proposed model can predict the mortality risk for patients after kidney transplants and could help kidney specialists choose kidney recipients with better prognosis.
Keywords: Kidney Transplantation, Predictive Value of Tests, Tissue Donors, Cadaver, Female, Humans, Kidney Failure, Chronic, Male, Models, Statistical, Patient Selection, Risk Assessment
Background
Kidney transplantation (KT) is the best treatment option for patients with end-stage renal disease (ESRD) [1], whose numbers have significantly increased worldwide in parallel with the increase in the average life expectancy over the past decades [2]. This preference over other options (such as kidney dialysis) is due to the increased survival rates and improved quality of life associated with KT procedures [3–5]. Kidneys to be transplanted mostly come from living healthy donors or through deceased donors (DD), including donors of cardiac death (DCD) and donors of brain death (DBD) [6–8]. However, not all patients can get kidneys from living donors due to matching problems, ethical considerations, and other reasons. The alternative source of transplanted kidneys would then be DD [9].
Therefore, one key goal that should be considered in the donor-recipient matching process is to minimize the post-operative risk (eg, recipient mortality or graft failure). Previous studies have confirmed that such risks can be influenced by numerous factors, including donor status (alive vs dead), donor race, donor age, recipient age, recipient race, and ethnicity, as well as chronic medical conditions, such as diabetes [10], hypertension [11,12], gastroenterological complications [10], comorbidities, acute rejection, delayed graft function, and post-transplantation proteinuria [13–20]. Consequently, it would be beneficial to collectively consider such risk factors for prognosis and risk assessment after KT operations. In fact, the literature abounds with research work intended to evaluate the various outcomes of kidney transplantation. Porrini et al [23] used 11 predictors to create risk prediction tools that both patients and clinicians can conveniently use in order to understand the absolute and relative risks leading to graft loss within 5 years after transplantation. Foucher et al [24] devised a composite clinical score, the Kidney Transplant Failure Score (KTFS), to predict the dialysis risk 1 year after the kidney transplantation. This score takes into account multiple well-recognized pre- and post-transplantation risk factors of graft loss. Numerous similar studies have been conducted [21–23]. However, these studies investigated either too many or too few risk factors. Few studies used pre-operative data to predict the post-operative mortality risk in kidney transplant patients. Obviously, building a comprehensive prediction model based on pre-operative donor and recipient data is vital for the prediction of patient survivability after KT.
To create a model for achieving these goals, we collected pre-operative laboratory test indicators. Specifically, we collected biochemical blood indices, including kidney function, liver function, electrolytes, fasting blood-glucose (FBG) and blood routine), underlying diseases (including high blood pressure (HBP), pre-transplant diabetes mellitus (pt-DM), and demographics. Then, we utilized these indicators to build a predictive model for post-operative prognosis. In particular, we used a series of statistical tests to verify the accuracy and applicability of this prediction model. With the rapid worldwide spread of solid-organ transplantation procedures, understanding the risk factors and possible KT consequences can help in clinical decision making. In addition, the proposed predictive model might help the kidney transplantation surgeons reach better overall prognosis.
Material and Methods
STUDY POPULATION:
We obtained the necessary study approval from the Ethics Committee of the First Affiliated Hospital of Anhui Medical University. We retrospectively searched the pre-operative medical records of 263 kidney recipients from April 1, 2016 to December 31, 2019. During that time interval, these recipients were all undergoing KT for the first time, and they were from various parts of Anhui Province, China. All donated kidneys came from deceased donors.
DATA COLLECTION, DIAGNOSTIC CRITERIA, AND OBSERVATION GUIDELINES:
For each selected patient, 30 indicators were collected, including (in addition to the donor age) 29 indicators of the recipient: 24 blood indicators, age, sex, pre-transplant diabetes mellitus (pt-DM), high blood pressure (HBP), and the distance between the patient’s home and the hospital. All relevant data of each patient was collected within 3 days before the kidney transplant operation. As for the diagnostic criteria, only KT-related deaths were included in the study, while deaths due to other causes (such as car accidents) were excluded. The following observation guidelines were followed: (1) The medical condition of each kidney recipient was followed up until January 1, 2021 after surgery; (2) If the recipient died after surgery before January 1, 2021, the observation was ended at the death date.
STATISTICAL ANALYSIS:
Statistical data analysis and visualization were performed using R software (version 4.0.3;
We applied logistic regression analysis to 26 continuous variables (including 24 blood indicators, donor age, and recipient age), and converted these continuous variables into dichotomous ones for the best separation method. At the same time, single-factor logistic regression analysis was performed on the above variables. The 26 variables were augmented with 4 categorical variables (recipient sex, pt-DM, HBP, and the home-hospital distance). Then, the least absolute shrinkage and selection operator (LASSO) method was applied to select the most predictive variables and hence reduce the complexity of the prediction model [24,25]. In particular, features with nonzero coefficients in the LASSO regression model were selected [26] based on an odds ratio (OR) statistic with a two-sided 95% confidence interval. For the kidney recipients, risk scores were calculated with nonzero coefficients. These scores were then used to construct receiver operating characteristic (ROC) curves and obtain the values of the area under the curve (AUC) with a 95% confidence interval. Calibration curves were plotted to assess the predictive model standardization. A significance test shows that the model is not ideally calibrated [27]. While traditional diagnostic indicators only reflect the diagnostic accuracy but not the clinical utility of a particular predictive model, decision curve analysis (DCA) accounts for patient features [28]. Moreover, this type of analysis determines the clinical validity of a prediction model by quantifying the net benefit under different probability thresholds [29]. This net benefit is calculated by subtracting the proportion of false-positive recipients from the proportion of true-positive recipients, and weighing the relative harm of giving up the intervention against the negative consequences of an unnecessary intervention [30]. In the last step, the dataset was evenly and randomly divided into 2 groups: Group I and Group II (in R software). This procedure was repeated 10 times. Thus, we performed testing with the internal dataset for 10 times, and for each division, the 2 subsets produced a collection of AUC values, false-positive rates, and true-positive rates. The overall workflow is demonstrated in Figure 1.
Results
DEMOGRAPHIC AND CLINICAL PATIENT CHARACTERISTICS:
In total, we considered 263 kidney recipients who had kidneys transplanted from deceased donors. Among these patients, only 151 subjects had no missing data for 3 days before the transplant and were thus included in the study. Among these included patients, 18 recipients had kidneys donated from DCD, 12 recipients had kidneys donated from DBD, and 121 recipients had kidneys donated from heart-brain death donors. By January 1, 2021, 15 of the 151 patients had died. The selected patients consisted of 133 males and 18 females, with a mean age of 40.7±9.9 years (and an age range of 15–63 years). Table 1 shows a list of all 30 independent variables of the recipients including demographics, blood indicators, pt-DM, and BHP (the groups are created based on death and non-death). Figure 2 shows the K-M survival curve of patients who received kidneys from deceased donors. The curve shows that the survival rate of the kidney recipients at 1743 days after surgery was 89.9%.
FEATURE SELECTION:
Firstly, to maximize the potential of each pertinent variable in predicting the renal transplantation outcomes, we transformed 26 continuous variables into dichotomous ones by the best separation method. Secondly, LASSO-based feature selection and the 151-patient dataset were employed to select the 22 best potential predictors from the original 30 predictors (Figure 3). The selected features are the concentrations of creatinine, aspartate aminotransferase (AST), alanine aminotransferase (ALT), total bilirubin (TBil), albumin (ALB), globulin (GLOB), the ratio of albumin to globulin (A/G), the concentrations of K, Na, Cl, and HCO3, the white blood cell (WBC) count, neutrophile (NEUT), neutrophile percent, lymph, monocytes (MONO), eosinophilic (EO), hemoglobin (HB), sex, pre-transplant diabetes mellitus (pt-DM), the donor age, and the recipient age (Table 2).
MODEL PERFORMANCE IN POST-TRANSPLANT MORTALITY PREDICTION:
The prediction model had an AUC of 0.690 (based on 22 potential predictors) for the cohort. This result is shown in the ROC curve plot of Figure 4A, where the x-axis and y-axis represent the false-positive and true-positive rates for mortality prediction after kidney transplants. The calibration curve of the post-transplant mortality prediction model for the ESRD patients who received kidney from DD demonstrated agreement in the studied cohort (Figure 4B).
CLINICAL APPLICABILITY:
The DCA curve of the mortality prediction model is shown in Figure 4C. This curve shows that if the probability thresholds of a patient and a doctor are >0.02 and <0.24, respectively, then the proposed predictive model will be more beneficial than the original plan.
INTERNAL VALIDATION:
Table 3 shows the internal validation results with an average AUC of 0.691 (with a range of 0.557–0.823) for a 10-fold cross-validation scheme.
Discussion
Predicting post-KT functional recovery and survival is crucial, especially with the critically limited kidney donor pool. Currently, there are only 2 treatment options for patients with ESRD: dialysis and KT. Comparative studies have consistently demonstrated that KT generally outperforms dialysis in improving the patient’s quality of life [31], reducing financial expenses [32], and extending the residual life of a patient [1]. Therefore, KT is the best choice for ESRD patients. Also, with recent KT developments, donor-recipient matching outcomes and KT success rates have been gradually improved through modern techniques such as the ABO agglutination test [33,34], lymphocytotoxicity crossmatch (LCT-XM) [35], human leukocyte antigen (HLA) testing [36,37], and panel-reactive antibody (PRA) testing [38–40]. However, prognosis is equally important. In fact, the number of patients awaiting kidney transplantation has grown significantly over the past few decades, while the number of available kidney donors has increased only modestly [41]. With these constraints, the limited KT resources should be efficiently utilized through optimal donor-recipient matching and selecting patients with good prognoses. Many studies have focused on risk assessment based on post-operative laboratory testing results or general information from the donors or recipients [42–45]. Nevertheless, there has been little focus on assessing risk based on pre-operative information [46]. In particular, we seek an answer to the question: Can a model be established to assess the post-operative risk by analyzing pre-operative laboratory test factors?
To address this question, we investigated the correlation between pre-operative clinical indicators and clinical outcomes after kidney transplantation. To construct the associated predictive model without involving variations in the source of the kidney transplantation, we collected data from recipients who received kidneys from deceased donors. This particular choice was made because earlier studies suggested that the 1-year, 3-year, and 5-year survival rates of recipients who received kidneys from living donors are different from the survival rates of recipients of kidneys from deceased donors [47,48]. To boost the prediction performance in our work, logistic regression analysis was employed to translate 26 independent continuous variables into dichotomous ones, which were also supplemented with 4 categorical variables. Then, a LASSO method was employed to further select the 22 best variables among the 30 original ones. Finally, we evaluated the modeling and prediction performance using calibration, ROC, and DCA curves. Our results showed good prediction based on the variables of creatinine, AST, ALT, Tbil, ALB, GLOB, A/G, K, Na, Cl, HCO3, WBC, NEUT, NEUT percent, lymph, MONO, EO, HB, sex, pt-DM, donor age, and recipient age. We also evaluated the proposed model via internal validation, which has also shown good results. In our work, the LASSO algorithm was introduced on the basis of logistic regression analysis of the pre-operative continuous variables. This LASSO algorithm is used to optimize feature selection for the risk model. Subsequently, the coefficients of the relatively unimportant variables become 0, and these variables are thus excluded from the prediction model. Finally, only the best predictor variables are retained. Because the LASSO algorithm can achieve optimal selection of categorical variables, we used the best separation method to find the cut-off value (where single-factor logistic regression analysis is performed on each continuous variable during the operation) to transform the continuous variables into categorical variables and hence adapt to the LASSO algorithm.
Our model involved 18 laboratory parameters. In particular, creatinine, ALB, and lymph concentrations are more important variables than the other laboratory factors. These 3 variables are easily obtainable in clinical settings. In addition, these 3 variables have the largest regression coefficient magnitudes in the fit prediction model (Table 2). Currently, creatinine level is a key indicator of renal function, and studies had been conducted to investigate the relationship between the creatinine level and the prognosis of kidney dialysis patients. Also, Gallinat et al demonstrated that the serum creatinine level of a kidney donor affected the recipient life after KT [49]. Foucher et al built a prediction model based on the Kidney Transplant Failure Score, and found the creatinine levels after 6–12 months of KT to be good predictors of survival 8 years after the kidney transplant [50]. These results indicate a positive association between the serum creatinine levels and survival after KT. Based on the prediction of the recipients’ pre-operative serum creatinine levels, our study further complemented the findings on the correlation between the creatinine level and prognosis. In addition, albumin level is a measure of liver function, as reported by Molnar et al [51], and lower blood albumin concentrations in ESRD patients are associated with higher mortality risks. This observation is consistent with our conclusions. However, earlier studies verified the connection between the lymphocytes and post-transplant prognosis. Bruminhent et al [52] reported a positive correlation between the absolute lymphocyte count and survival after KT. Indeed, high absolute counts of lymphocytes could inhibit human adenovirus infection, and hence improve the chances of survival of kidney recipients. Sun et al [53] found that the lymphocyte count can be used as a predictor of ARDS in KT patients with cytomegalovirus pneumonia. The increase in the numbers of CD4 + T and CD8 + T cells will increase the patient survival rate. Therefore, creatinine, ALB, and lymphocytes have strong correlations with the recipient prognosis. Also, there is a definite correlation between pre-operative blood indicators and survival rate.
Demographic databases also play an important role in forecasting models. Laging et al [54] reported that donor age is a key independent risk factor for post-operative mortality. Moosa et al also confirmed that the younger the kidney donors, the longer the survival times of the recipients with transplanted kidneys [55]. Furthermore, Moers et al [55] quantified the extent to which the donor age negatively affected the survival of kidney recipients. To meet the increasing KT demand, the internationally proposed designation of an expanded criteria donor (ECD) was introduced as a high-risk donor [56]. One of the subsequent criteria is that a donor over 60 years old is categorized as ECD and this increases the risk for poor prognosis. The proportion of such old donors in our database was approximately 14% (21 out of 151), and this “risk ratio” coincided with the high value of the coefficient of the donor age predictor in our prediction model. Other studies addressed the effects of donor and recipient sex. Tent et al [58] have shown that the donor sex had no effect on renal function after KT operations. Hence, we focused on the sex disparity for the recipients. Abou-Jaoude et al [57] confirmed that the recipient sex is a key independent risk factor for KT success. In our dataset, the male-to-female ratio of the recipients was 133 to 18, and the regression coefficient of the female recipients was higher than that of the males. This is consistent with the results reported by Moosa [54] that the recipient mortality for females is higher than that for males [55]. In conclusion, the predictive factors of our model cover multiple aspects and dimensions, including experimental factors and population data. Prior to our work, few earlier studies collected so many pieces of information for prognosis after KT. Therefore, we hope our method will help doctors match donors to recipients with better long-term prognosis. At the same time, doctors can employ our predictive model to take precautions for recipients with high post-operative mortality risk.
Admittedly, our proposed prediction model still has some limitations. Firstly, this model does not consider several key donor factors, such as the donor sex, and the warm/cold ischemia time for kidney transplant. As suggested by earlier studies, these types of donor information are influential for post-transplant prognosis [58–60]. Future directions for enhancing this work include expanding the sample size, collecting more pre-operative data of KT patients, and extending the follow-up time to improve the prediction model accuracy. The second limitation is that both the sample size and the follow-up period were not large enough. Indeed, our sample has only a mean follow-up time of 1.5 years and 151 recipients (including 16 dead subjects). Thirdly, although the prediction model robustness has been extensively validated internally through a randomized cross-validation scheme, this model could not be externally validated in datasets of other kidney recipients in other regions and countries. Hence, there is still some uncertainty about the model’s universality. External evaluation is actually required in a larger test group to ensure the suitability, reliability, and practicality of our prediction model.
Conclusions
We proposed a novel prognostic model for patients who received kidney donations from deceased donors. This model has 22 predictors that could be applied to facilitate the prediction of individual post-operative survivability in recipients. Predictive models based on donor and recipient pre-operative data should be assessed and utilized by kidney specialists in clinical practice.
Figures
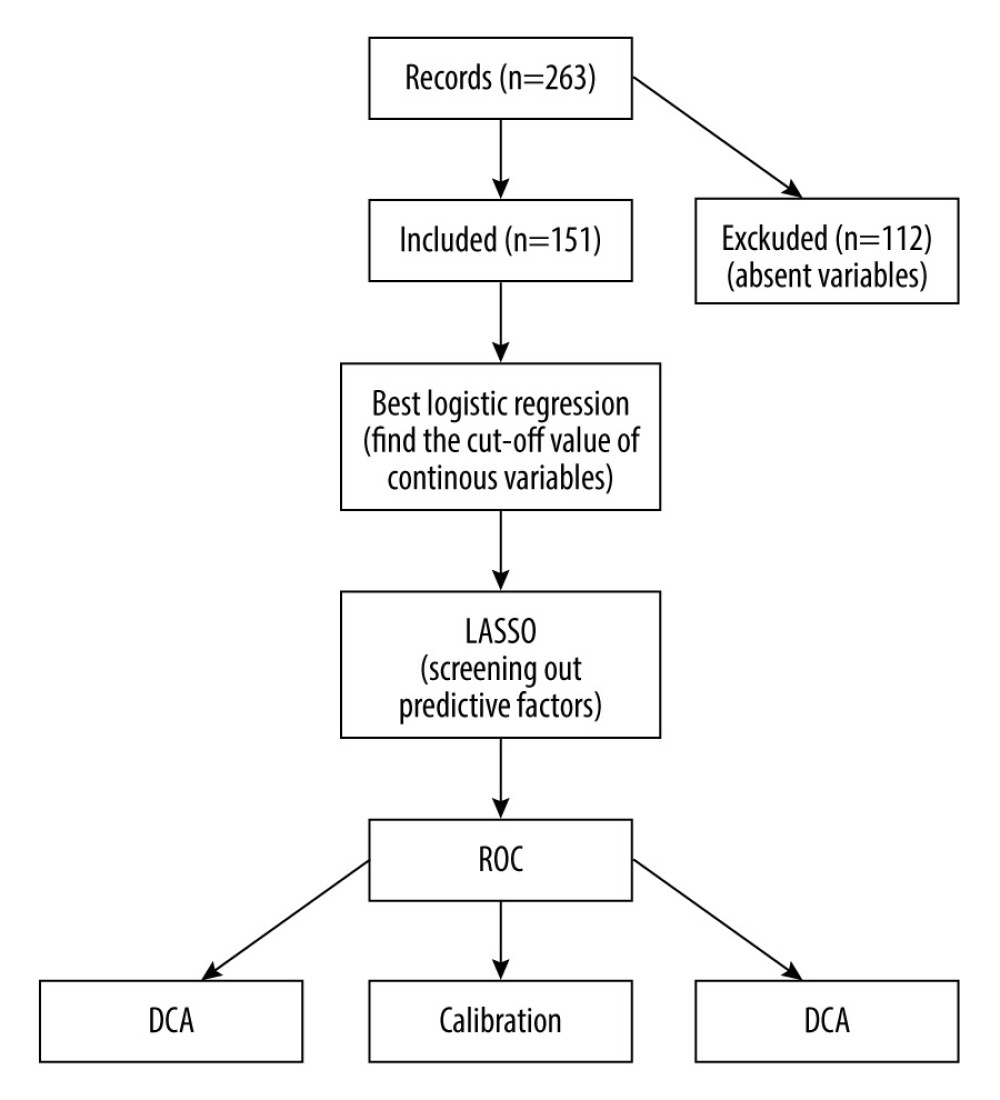 Figure 1. A flowchart of the proposed post-KT mortality risk assessment framework.The figure was created using WPS Office software (version 11.1.0.10700-release).
Figure 1. A flowchart of the proposed post-KT mortality risk assessment framework.The figure was created using WPS Office software (version 11.1.0.10700-release). 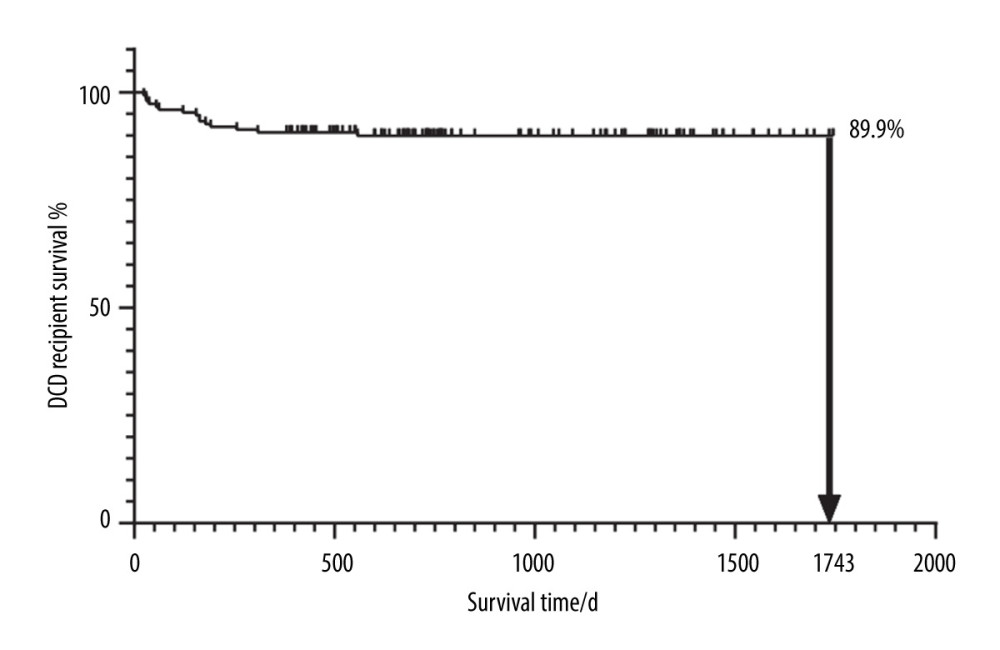 Figure 2. The K-M curve reflecting the survival conditions of patients receiving kidneys from deceased donors.The figure was created using GraphPad Prism software (version 8.0).
Figure 2. The K-M curve reflecting the survival conditions of patients receiving kidneys from deceased donors.The figure was created using GraphPad Prism software (version 8.0). 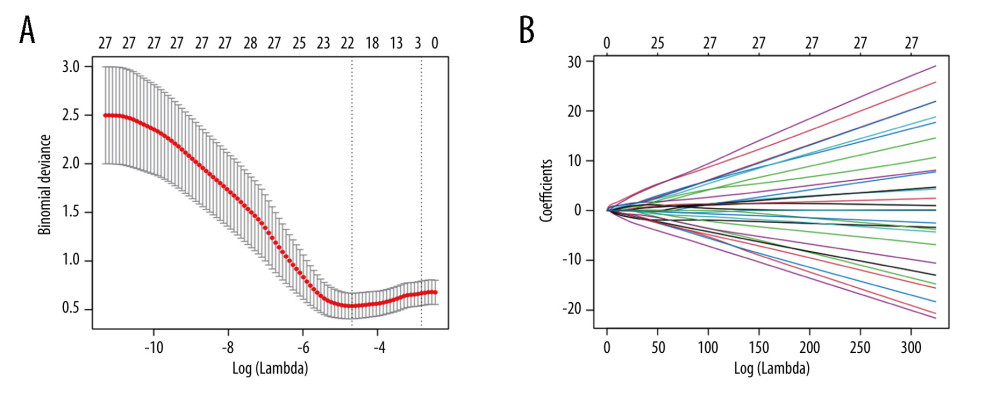 Figure 3. Demographics and clinical feature selection using the LASSO binary regression model.(A) LASSO coefficient profiles for 30 features. Each coefficient profile was plotted against the logarithm of the lambda parameter. The vertical line was drawn at the value selected using 5-fold cross-validation, where the optimal lambda value resulted in 22 features with nonzero coefficients. (B) Optimal lambda parameter selection in the LASSO model using 5-fold cross-validation via minimum criteria. The curve of the partial likelihood deviance (binomial deviance) was plotted versus the logarithm of the lambda parameter. Dotted vertical lines were drawn at the optimal values by using the minimum criteria and the I-SE of the minimum criteria (the I-SE criteria). The figures use the R software (version 4.0.3; https://www.R-project.org). LASSO – least absolute shrinkage and selection operator; SE – standard error.
Figure 3. Demographics and clinical feature selection using the LASSO binary regression model.(A) LASSO coefficient profiles for 30 features. Each coefficient profile was plotted against the logarithm of the lambda parameter. The vertical line was drawn at the value selected using 5-fold cross-validation, where the optimal lambda value resulted in 22 features with nonzero coefficients. (B) Optimal lambda parameter selection in the LASSO model using 5-fold cross-validation via minimum criteria. The curve of the partial likelihood deviance (binomial deviance) was plotted versus the logarithm of the lambda parameter. Dotted vertical lines were drawn at the optimal values by using the minimum criteria and the I-SE of the minimum criteria (the I-SE criteria). The figures use the R software (version 4.0.3; https://www.R-project.org). LASSO – least absolute shrinkage and selection operator; SE – standard error. 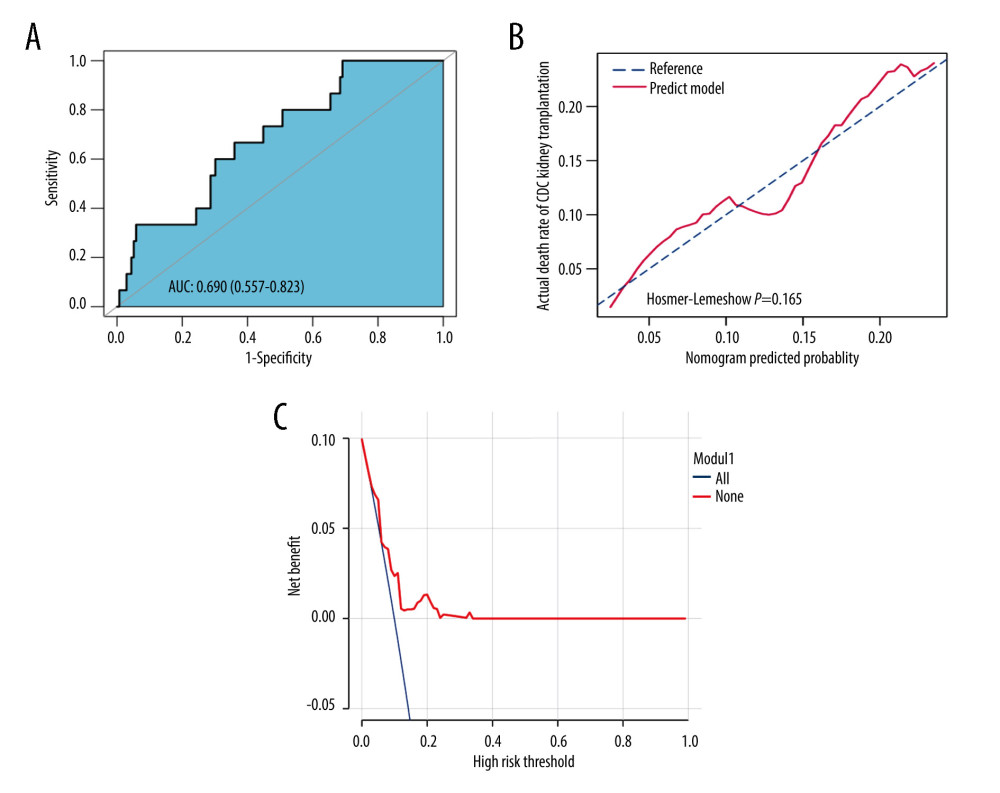 Figure 4. Assessment of the clinical effectiveness of the proposed model.(A) Receiver operating characteristic (ROC) curves of the nomogram of the post-KT mortality risk prediction. The x-axis and y-axis represent the false-positive and true-positive rates of the post-KT mortality risk prediction. The area under the ROC curve (AUC) is 0.690. (B) Calibration curves of the nomogram of the post-KT mortality risk prediction for the study cohort. The x-axis represents the post-KT death predictions, while the y-axis represents the actual death outcomes. The diagonal dotted line corresponds to perfect prediction outcomes by an ideal model. The solid line represents the nomogram performance, where a closer fit to the diagonal dotted line represents better prediction. (C) Decision curve analysis (DCA) for post-KT death. The y-axis measures the net benefit. The red line represents the nomogram of the post-KT death risk. The thin solid line represents the assumption of survival of all kidney recipients after kidney transplantation, while the thick solid line represents the assumption of the death of all kidney recipients after kidney transplantation. The decision curve shows that if the threshold probabilities of a patient and a doctor are (>0.02) and (<0.24), respectively, using the mortality risk nomogram in the current study for post-KT mortality risk prediction is more beneficial than the intervention-for-all-recipients scheme or the no-intervention scheme. The figures use R software (version 4.0.3; https://www.R-project.org).
Figure 4. Assessment of the clinical effectiveness of the proposed model.(A) Receiver operating characteristic (ROC) curves of the nomogram of the post-KT mortality risk prediction. The x-axis and y-axis represent the false-positive and true-positive rates of the post-KT mortality risk prediction. The area under the ROC curve (AUC) is 0.690. (B) Calibration curves of the nomogram of the post-KT mortality risk prediction for the study cohort. The x-axis represents the post-KT death predictions, while the y-axis represents the actual death outcomes. The diagonal dotted line corresponds to perfect prediction outcomes by an ideal model. The solid line represents the nomogram performance, where a closer fit to the diagonal dotted line represents better prediction. (C) Decision curve analysis (DCA) for post-KT death. The y-axis measures the net benefit. The red line represents the nomogram of the post-KT death risk. The thin solid line represents the assumption of survival of all kidney recipients after kidney transplantation, while the thick solid line represents the assumption of the death of all kidney recipients after kidney transplantation. The decision curve shows that if the threshold probabilities of a patient and a doctor are (>0.02) and (<0.24), respectively, using the mortality risk nomogram in the current study for post-KT mortality risk prediction is more beneficial than the intervention-for-all-recipients scheme or the no-intervention scheme. The figures use R software (version 4.0.3; https://www.R-project.org). References
1. Wolfe RA, Ashby VB, Milford EL, Comparison of mortality in all patients on dialysis, patients on dialysis awaiting transplantation, and recipients of a first cadaveric transplant: N Engl J Med, 1999; 341(23); 1725-30
2. Saran R, Robinson B, Abbott KC, US Renal Data System 2016 Annual Data Report: Epidemiology of kidney disease in the United States: Am J Kidney Dis, 2017; 69(3 Suppl 1); A7-A8
3. Womer KL, Vella JP, Sayegh MH, Chronic allograft dysfunction: mechanisms and new approaches to therapy: Semin Nephrol, 2000; 20(2); 126-47
4. Merion RM, Ashby VB, Wolfe RA, Deceased-donor characteristics and the survival benefit of kidney transplantation: JAMA, 2005; 294(21); 2726-33
5. Hernandez-Fuentes MP, Lechler RI, Chronic graft loss. Immunological and non-immunological factors: Contrib Nephrol, 2005; 146; 54-64
6. Moers C, Leuvenink HGD, Ploeg RJ, Donation after cardiac death: Evaluation of revisiting an important donor source: Nephrol Dial Transplant, 2010; 25(3); 666-73
7. Magliocca JF, Magee JC, Rowe SA, Extracorporeal support for organ donation after cardiac death effectively expands the donor pool: J Trauma, 2005; 58(6); 1095-101 discussion 1101–2
8. Li Y, Li J, Fu Q, Kidney transplantation from brain-dead donors: Initial experience in China: Transplant Proc, 2016; 48(8); 2592-95
9. Zhang L, Zeng L, Gao X, Transformation of organ donation in China: Transpl Int, 2015; 28(4); 410-15
10. Calogero A, Gallo M, Sica A, Gastroenterological complications in kidney transplant patients: Open Med (Wars), 2020; 15(1); 623-34
11. Tantisattamo E, Molnar MZ, Ho BT, Approach and management of hypertension after kidney transplantation: Front Med (Lausanne), 2020; 7; 229
12. Thomas B, Taber DJ, Srinivas TR, Hypertension after kidney transplantation: A pathophysiologic approach: Curr Hypertens Rep, 2013; 15(5); 458-69
13. Ojo AO, Hanson JA, Meier-Kriesche H, Survival in recipients of marginal cadaveric donor kidneys compared with other recipients and wait-listed transplant candidates: J Am Soc Nephrol, 2001; 12(3); 589-97
14. Troppmann C, Gillingham KJ, Benedetti E, Delayed graft function, acute rejection, and outcome after cadaver renal transplantation. The multivariate analysis: Transplantation, 1995; 59(7); 962-68
15. Almond PS, Matas A, Gillingham K, Risk factors for chronic rejection in renal allograft recipients: Transplantation, 1993; 55(4); 752-56 discussion 756–67
16. Cosio FG, Hickson LJ, Griffin MD, Patient survival and cardiovascular risk after kidney transplantation: The challenge of diabetes: Am J Transplant, 2008; 8(3); 593-99
17. Molnar MZ, Langer RM, Remport A, Roma ethnicity and clinical outcomes in kidney transplant recipients: Int Urol Nephrol, 2012; 44(3); 945-54
18. Molnar-Varga M, Molnar MZ, Szeifert L, Health-related quality of life and clinical outcomes in kidney transplant recipients: Am J Kidney Dis, 2011; 58(3); 444-52
19. Molnar MZ, Streja E, Kovesdy CP, Age and the associations of living donor and expanded criteria donor kidneys with kidney transplant outcomes: Am J Kidney Dis, 2012; 59(6); 841-48
20. Roodnat JI, Mulder PG, Rischen-Vos J, Proteinuria after renal transplantation affects not only graft survival but also patient survival: Transplantation, 2001; 72(3); 438-44
21. Moore J, He X, Shabir S, Development and evaluation of a composite risk score to predict kidney transplant failure: Am J Kidney Dis, 2011; 57(5); 744-51
22. Israni AK, Snyder JJ, Skeans MA, Predicting coronary heart disease after kidney transplantation: Patient Outcomes in Renal Transplantation (PORT) study: Am J Transplant, 2010; 10(2); 338-53
23. Porrini E, Ruggenenti P, Luis-Lima S, Author correction: Estimated GFR: Time for a critical appraisal: Nat Rev Nephrol, 2019; 15(2); 121
24. Sauerbrei W, Royston P, Binder H, Selection of important variables and determination of functional form for continuous predictors in multivariable model building: Stat Med, 2007; 26(30); 5512-28
25. Friedman J, Hastie T, Tibshirani R, Regularization paths for generalized linear models via coordinate descent: J Stat Softw, 2010; 33(1); 1-22
26. Kidd AC, McGettrick M, Tsim S, Survival prediction in mesothelioma using a scalable Lasso regression model: Instructions for use and initial performance using clinical predictors: BMJ Open Respir Res, 2018; 5(1); e000240
27. Kramer AA, Zimmerman JE, Assessing the calibration of mortality benchmarks in critical care: The Hosmer-Lemeshow test revisited: Crit Care Med, 2007; 35(9); 2052-56
28. Fitzgerald M, Saville BR, Lewis RJ, Decision curve analysis: JAMA, 2015; 313(4); 409-10
29. Vickers AJ, Cronin AM, Elkin EB, Gonen M, Extensions to decision curve analysis, a novel method for evaluating diagnostic tests, prediction models and molecular markers: BMC Med Inform Decis Mak, 2008; 8; 53
30. Huang Y-Q, Liang C-H, He L, Development and validation of a radiomics nomogram for preoperative prediction of lymph node metastasis in colorectal cancer: J Clin Oncol, 2016; 34(18); 2157-64
31. Purnell TS, Auguste P, Crews DC, Comparison of life participation activities among adults treated by hemodialysis, peritoneal dialysis, and kidney transplantation: A systematic review: Am J Kidney Dis, 2013; 62(5); 953-73
32. Silva SB, Caulliraux HM, Araújo CAS, Rocha E, Cost comparison of kidney transplant versus dialysis in Brazil: Cad Saude Publica, 2016; 32(6); S0102-311 X2016000605005
33. Basu A, Prieto M, Kosberg C, Ten years of kidney paired donation at Mayo Clinic: The benefits of incorporating ABO/HLA compatible pairs: Transplantation, 2020; 104(6); 1229-38
34. Couzi L, Manook M, Perera R, Difference in outcomes after antibody-mediated rejection between abo-incompatible and positive cross-match transplantations: Transpl Int, 2015; 28(10); 1205-15
35. Deepa R, Balapriya P, Venkatesh KG, Complement-dependent lymphocytotoxicity crossmatch in deceased donor renal transplant: A single institutional experience: Indian J Med Microbiol, 2011; 29(4); 414-17
36. South AM, Grimm PC, Transplant immuno-diagnostics: Crossmatch and antigen detection: Pediatr Nephrol, 2016; 31(6); 897-905
37. Kransdorf EP, Pando MJ, Gragert L, Kaplan B, HLA population genetics in solid organ transplantation: Transplantation, 2017; 101(9); 1971-76
38. Karadeniz ST, Akgul SU, Ogret Y, Corrected panel-reactive antibody positivity rates for hypersensitized patients in Turkish population with calculated panel-reactive antibody software: Transplant Proc, 2017; 49(3); 445-47
39. Lee N, Park HS, In JW, Association of HLA types with non-specific binding of negative control beads in luminex panel reactive antibody (PRA) screening assay: Clin Lab, 2017; 63(1); 169-74
40. Huber L, Lachmann N, Niemann M, Pretransplant virtual PRA and long-term outcomes of kidney transplant recipients: Transpl Int, 2015; 28(6); 710-19
41. Wu DA, Watson CJ, Bradley JA, Global trends and challenges in deceased donor kidney allocation: Kidney Int, 2017; 91(6); 1287-99
42. Kaboré R, Ferrer L, Couchoud C, Dynamic prediction models for graft failure in paediatric kidney transplantation: Nephrol Dial Transplant, 2021; 36(5); 927-35
43. Kabore R, Haller MC, Harambat J, Risk prediction models for graft failure in kidney transplantation: a systematic review: Nephrol Dial Transplant, 2017; 32(Suppl 2); ii68-76
44. Maier HT, Ashraf MI, Denecke C, Prediction of delayed graft function and long-term graft survival by serum and urinary neutrophil gelatinase-associated lipocalin during the early postoperative phase after kidney transplantation: PLoS One, 2018; 13(1); e0189932
45. Kers J, Peters-Sengers H, Heemskerk MBA, Prediction models for delayed graft function: external validation on The Dutch Prospective Renal Transplantation Registry: Nephrol Dial Transplant, 2018; 33(7); 1259-68
46. Matsukuma Y, Masutani K, Tanaka S, Development and validation of a new prediction model for graft function using preoperative marginal factors in living-donor kidney transplantation: Clin Exp Nephrol, 2019; 23(11); 1331-40
47. Zhang H, Wei Y, Liu L, Different risk factors for graft survival between living-related and deceased donor kidney transplantation: Transplant Proc, 2018; 50(8); 2416-20
48. Roldán-Reina AJ, Martín-Villén L, Palomo-López N, Living donor in renal transplantation: Minimizing risks: Transplant Proc, 2018; 50(2); 543-45
49. Gallinat A, Leerhoff S, Paul A, Kidney transplantation from deceased donors with elevated serum creatinine: Langenbecks Arch Surg, 2016; 401(8); 1211-17
50. Foucher Y, Daguin P, Akl A, A clinical scoring system highly predictive of long-term kidney graft survival: Kidney Int, 2010; 78(12); 1288-94
51. Molnar MZ, Kovesdy CP, Bunnapradist S, Associations of pretransplant serum albumin with post-transplant outcomes in kidney transplant recipients: Am J Transplant, 2011; 11(5); 1006-15
52. Bruminhent J, Apiwattanakul N, Hongeng S, Absolute lymphocyte count and human adenovirus-specific T-cell immune restoration of human adenovirus infection after kidney transplantation: J Med Virol, 2019; 91(8); 1432-39
53. Sun Q, Li L, Ji S, Variation of CD4+ and CD8+ T lymphocytes as predictor of outcome in renal allograft recipients who developed acute respiratory distress syndrome caused by cytomegalovirus pneumonia: Transplant Proc, 2005; 37(5); 2118-21
54. Laging M, Kal-van Gestel JA, van de Wetering J, The relative importance of donor age in deceased and living donor kidney transplantation: Transpl Int, 2012; 25(11); 1150-57
55. Port FK, Bragg-Gresham JL, Metzger RA, Donor characteristics associated with reduced graft survival: An approach to expanding the pool of kidney donors: Transplantation, 2002; 74(9); 1281-86
56. Port FK, Bragg-Gresham JL, Metzger RA, Donor characteristics associated with reduced graft survival: An approach to expanding the pool of kidney donors: Transplantation, 2002; 74(9); 1281-86
57. Abou-Jaoude MM, El-Helou E, Nasser HA, Kansoun AH, The impact of gender matching between donor and recipient on the outcome of kidney transplant patients: A retrospective study: Saudi J Kidney Dis Transpl, 2019; 30(6); 1254-65
58. van der Vliet JA, Warlé MC, The need to reduce cold ischemia time in kidney transplantation: Curr Opin Organ Transplant, 2013; 18(2); 174-78
59. van Es A, Hermans J, van Bockel JH, Effect of warm ischemia time and HLA (A and B) matching on renal cadaveric graft survival and rejection episodes: Transplantation, 1983; 36(3); 255-58
60. Wong G, Teixeira-Pinto A, Chapman JR, The impact of total ischemic time, donor age and the pathway of donor death on graft outcomes after deceased donor kidney transplantation: Transplantation, 2017; 101(6); 1152-58
Figures
 Figure 1. A flowchart of the proposed post-KT mortality risk assessment framework.The figure was created using WPS Office software (version 11.1.0.10700-release).
Figure 1. A flowchart of the proposed post-KT mortality risk assessment framework.The figure was created using WPS Office software (version 11.1.0.10700-release). Figure 2. The K-M curve reflecting the survival conditions of patients receiving kidneys from deceased donors.The figure was created using GraphPad Prism software (version 8.0).
Figure 2. The K-M curve reflecting the survival conditions of patients receiving kidneys from deceased donors.The figure was created using GraphPad Prism software (version 8.0). Figure 3. Demographics and clinical feature selection using the LASSO binary regression model.(A) LASSO coefficient profiles for 30 features. Each coefficient profile was plotted against the logarithm of the lambda parameter. The vertical line was drawn at the value selected using 5-fold cross-validation, where the optimal lambda value resulted in 22 features with nonzero coefficients. (B) Optimal lambda parameter selection in the LASSO model using 5-fold cross-validation via minimum criteria. The curve of the partial likelihood deviance (binomial deviance) was plotted versus the logarithm of the lambda parameter. Dotted vertical lines were drawn at the optimal values by using the minimum criteria and the I-SE of the minimum criteria (the I-SE criteria). The figures use the R software (version 4.0.3; https://www.R-project.org). LASSO – least absolute shrinkage and selection operator; SE – standard error.
Figure 3. Demographics and clinical feature selection using the LASSO binary regression model.(A) LASSO coefficient profiles for 30 features. Each coefficient profile was plotted against the logarithm of the lambda parameter. The vertical line was drawn at the value selected using 5-fold cross-validation, where the optimal lambda value resulted in 22 features with nonzero coefficients. (B) Optimal lambda parameter selection in the LASSO model using 5-fold cross-validation via minimum criteria. The curve of the partial likelihood deviance (binomial deviance) was plotted versus the logarithm of the lambda parameter. Dotted vertical lines were drawn at the optimal values by using the minimum criteria and the I-SE of the minimum criteria (the I-SE criteria). The figures use the R software (version 4.0.3; https://www.R-project.org). LASSO – least absolute shrinkage and selection operator; SE – standard error. Figure 4. Assessment of the clinical effectiveness of the proposed model.(A) Receiver operating characteristic (ROC) curves of the nomogram of the post-KT mortality risk prediction. The x-axis and y-axis represent the false-positive and true-positive rates of the post-KT mortality risk prediction. The area under the ROC curve (AUC) is 0.690. (B) Calibration curves of the nomogram of the post-KT mortality risk prediction for the study cohort. The x-axis represents the post-KT death predictions, while the y-axis represents the actual death outcomes. The diagonal dotted line corresponds to perfect prediction outcomes by an ideal model. The solid line represents the nomogram performance, where a closer fit to the diagonal dotted line represents better prediction. (C) Decision curve analysis (DCA) for post-KT death. The y-axis measures the net benefit. The red line represents the nomogram of the post-KT death risk. The thin solid line represents the assumption of survival of all kidney recipients after kidney transplantation, while the thick solid line represents the assumption of the death of all kidney recipients after kidney transplantation. The decision curve shows that if the threshold probabilities of a patient and a doctor are (>0.02) and (<0.24), respectively, using the mortality risk nomogram in the current study for post-KT mortality risk prediction is more beneficial than the intervention-for-all-recipients scheme or the no-intervention scheme. The figures use R software (version 4.0.3; https://www.R-project.org).
Figure 4. Assessment of the clinical effectiveness of the proposed model.(A) Receiver operating characteristic (ROC) curves of the nomogram of the post-KT mortality risk prediction. The x-axis and y-axis represent the false-positive and true-positive rates of the post-KT mortality risk prediction. The area under the ROC curve (AUC) is 0.690. (B) Calibration curves of the nomogram of the post-KT mortality risk prediction for the study cohort. The x-axis represents the post-KT death predictions, while the y-axis represents the actual death outcomes. The diagonal dotted line corresponds to perfect prediction outcomes by an ideal model. The solid line represents the nomogram performance, where a closer fit to the diagonal dotted line represents better prediction. (C) Decision curve analysis (DCA) for post-KT death. The y-axis measures the net benefit. The red line represents the nomogram of the post-KT death risk. The thin solid line represents the assumption of survival of all kidney recipients after kidney transplantation, while the thick solid line represents the assumption of the death of all kidney recipients after kidney transplantation. The decision curve shows that if the threshold probabilities of a patient and a doctor are (>0.02) and (<0.24), respectively, using the mortality risk nomogram in the current study for post-KT mortality risk prediction is more beneficial than the intervention-for-all-recipients scheme or the no-intervention scheme. The figures use R software (version 4.0.3; https://www.R-project.org). Tables
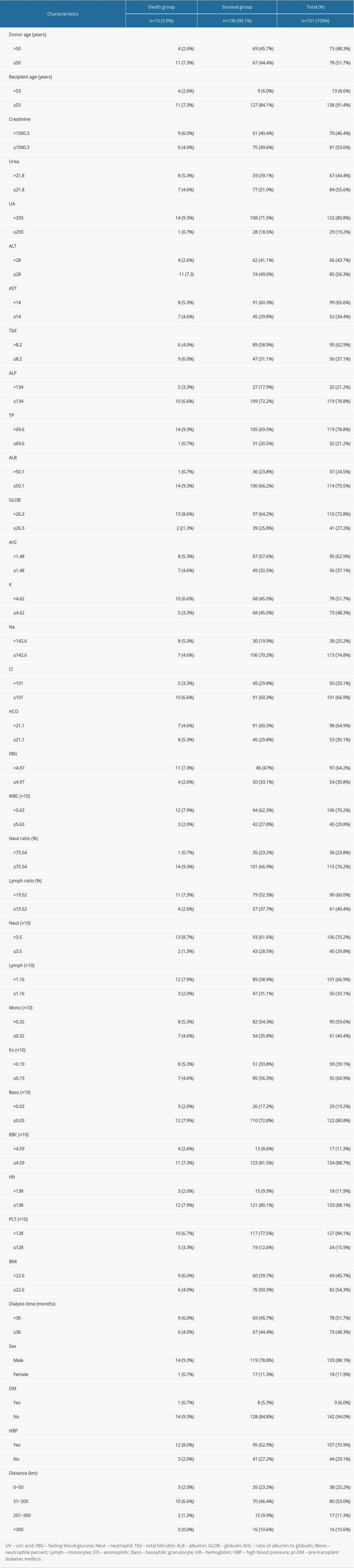 Table 1. Demographics and clinical characteristics of the death and survival groups.
Table 1. Demographics and clinical characteristics of the death and survival groups.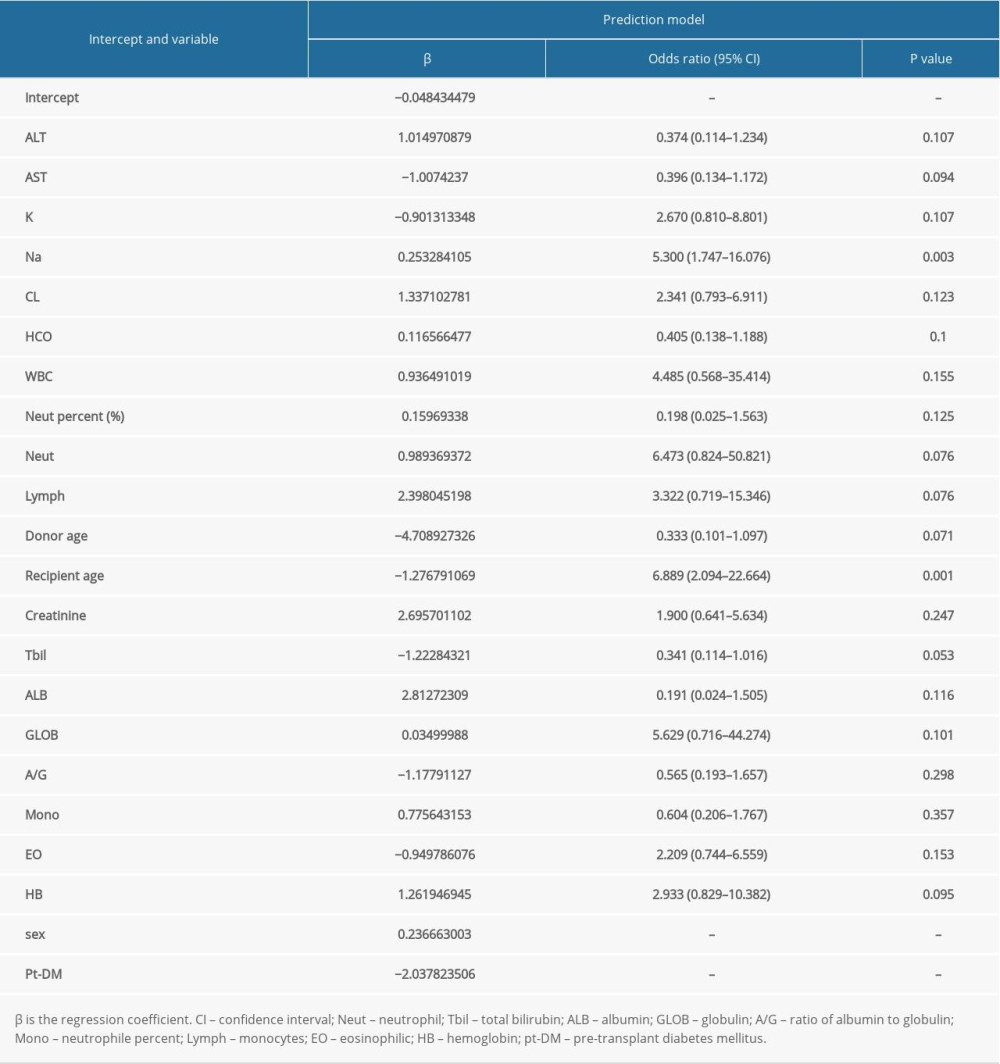 Table 2. Mortality prediction factors after renal transplantation from deceased donors.
Table 2. Mortality prediction factors after renal transplantation from deceased donors.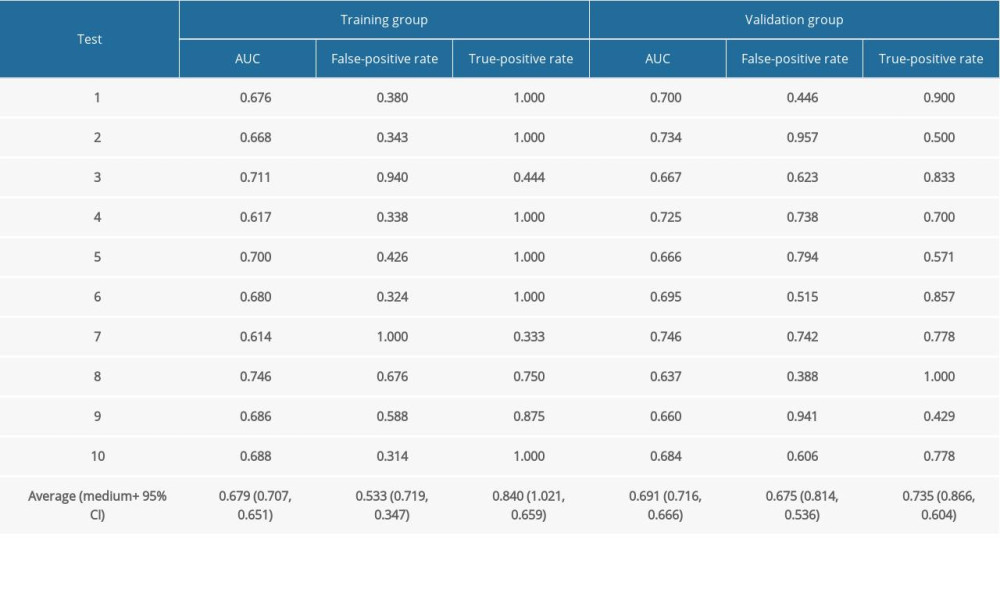 Table 3. Evaluation results for the proposed model among 10 randomly selected independent subgroups.
Table 3. Evaluation results for the proposed model among 10 randomly selected independent subgroups. Table 1. Demographics and clinical characteristics of the death and survival groups.
Table 1. Demographics and clinical characteristics of the death and survival groups. Table 2. Mortality prediction factors after renal transplantation from deceased donors.
Table 2. Mortality prediction factors after renal transplantation from deceased donors. Table 3. Evaluation results for the proposed model among 10 randomly selected independent subgroups.
Table 3. Evaluation results for the proposed model among 10 randomly selected independent subgroups. In Press
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








