11 January 2022: Clinical Research
Association Between Hyperhomocysteinemia Combined with Metabolic Syndrome and Higher Prevalence of Stroke in Chinese Adults Who Have Elevated Blood Pressure
Feng Hu12BCDEF, Shichao Yu3BCD, Juan Li4ABCD, Wei Zhou25BDEF, Tao Wang25BCDE, Xiao Huang125AF*, Huihui Bao125AG, Xiaoshu Cheng125AGDOI: 10.12659/MSM.934100
Med Sci Monit 2022; 28:e934100
Abstract
BACKGROUND: Hyperhomocysteinemia (HHcy) and metabolic syndrome (MS) are established cardiovascular risk factors of stroke and are frequently associated with hypertension. However, studies on the association between HHcy combined with MS and stroke risk in hypertensive patients were absent.
MATERIAL AND METHODS: In 14 059 selected participants with elevated blood pressure, we assessed the prevalence of the MS and stroke. We defined HHcy as plasma total homocysteine >15 μmol/L. MS was defined according to the Chinese Diabetes Society (CDS) criterion. Multivariable analysis was used to examine the association of HHcy or (and) MS with stroke risk in different models.
RESULTS: The prevalence rates of HHcy and MS were 49.96% and 42.21%, respectively. Patients with stroke had higher plasma total homocysteine levels and a higher prevalence of MS (P<0.001). Multivariable analyses indicated that HHcy and MS are independently associated with higher prevalence of stroke (adjusted-odds ratio (OR): 1.36, 95% CI 1.17 to 1.58, P<0.001; adjusted-OR: 1.68, 95% CI 1.44 to 1.96, P<0.001, respectively). Those with combined HHcy and MS had higher odds of stroke than those with isolated HHcy or MS (adjusted-OR: 1.78, 95% CI 1.47 to 2.15, P<0.001; adjusted-OR: 1.39, 95% CI 1.13 to 1.70, P=0.002, respectively).
CONCLUSIONS: HHcy combined with MS was associated with higher prevalence of stroke in Chinese adults with elevated blood pressure.
Keywords: Hyperhomocysteinemia, metabolic syndrome, Stroke, Blood Pressure Determination, Comorbidity, Correlation of Data, Female, Heart Disease Risk Factors, homocysteine, Humans, Hypertension, Male, Prevalence, Risk Assessment
Background
Globally, stroke is the second leading cause of death, and its prevalence in adults is 2.7% in the United States [1]. Stroke in Chinese adults has increased over the past 30 years, and the age-standardized prevalence, incidence, and mortality rates are 1114.8/100 000 people, 246.8, and 114.8/100 000 person-years, respectively [2]. There are some modifiable risk factors in the prevention and treatment of stroke, including obesity, hypertension, type 2 diabetes mellitus (T2DM), dyslipidemia, and hyperhomocysteinemia (HHcy) [1–4].
Homocysteine is a sulfurated amino acid that is synthesized during the methionine metabolic cycle, and the pathogenic elevation of the plasma total homocysteine (tHcy) levels above 10–15 μmol/L is defined as HHcy [3,4]. HHcy is an independent risk factor for cardiovascular diseases (CVD) such as hypertension, atherosclerosis, coronary heart disease (CHD), and stroke [5,6] via various pathways, including oxidative stress, endoplasmic reticulum (ER) stress, epigenetic modulation, and protein homocysteinylation [7]. The prevalence of hypertension in the Chinese adult population (≥18 years of age) is 23.2% (about 245 million) [8], and approximately 75% of Chinese patients with hypertension also have HHcy, which is associated with a higher risk of stroke [4].
Metabolic syndrome (MS), which is a global public health problem of great concern, is a cluster of clinical and metabolic factors that significantly increase the risk for T2DM, CHD, and stroke [9]. The interrelated risk factors include central obesity, dyslipidemias, hypertension, hypercoagulable state, and insulin resistance (IR) [10]. According to the International Diabetes Federation (IDF) criteria, the prevalence of MS in the USA is 33–39%, with many being female, while the prevalence of MS in Europe varies widely among countries, ranging from 18% to 30% [11,12]. The estimated prevalence of MS in China is 34.0% according to the revised ATP III criteria and 26.9% by IDF criteria, which is higher in women and older adults [13].
HHcy and MS are both established risk factors of stroke and are frequently associated with hypertension. However, there are no published studies on the combined effect of HHcy and MS on stroke risk in hypertensive patients, a cohort at particularly high risk for stroke [1,2,14]. China bears the huge burden of hypertension and stroke [2]. In this study, we investigated the association of HHcy combined with MS with the prevalence of stroke in Chinese adults with elevated blood pressure.
Material and Methods
STUDY POPULATION:
The China H-type Hypertension Registry Study was a real-world, multicenter, observational study, conducted in March 2018 at Wuyuan, Jiangxi province of China. The enrolled population consisted of hypertensive patients aged 18 years and older. The exclusion criteria included psychological or nervous system impairment resulting in an inability to provide informed consent, unable to participate in long-term followed-up according to the study protocol, or plans to relocate in the near future. The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the Institute of Biomedicine, Anhui Medical University. Signed informed consent was obtained from all participants [15].
We assessed data on a total of 14 268 individuals. Elevated blood pressure was defined as the 130/85 mmHg threshold [16,17] or having been diagnosed with hypertension and previously started treatment. After excluding those without elevated blood pressure (n=10) or with missing value of blood lipids (n=7), 1 patient with maximum value of Hcy (182.92 μmol/L), and 191 patients with atrial fibrillation that could cause cardiogenic stroke, 14 059 individuals were included in the analysis (Supplementary Figure 1).
DATA COLLECTION AND INDEXES DETERMINATION:
Data on individuals’ general characteristics (age, sex, and smoking and drinking status), past medical history (hypertension, diabetes, dyslipidemia, cerebral stroke [ischemic stroke or intracerebral hemorrhage]), and medication usage were gathered by professional researchers through a questionnaire survey. Atrial fibrillation was assessed by past medical history and electrocardiograms.
The anthropometric examinations included systolic blood pressure (SBP), diastolic blood pressure (DBP), body height, weight, height, waistline, and hipline. BP was measured, with the participant in a sitting position using the Omron HBP-1300 Professional Portable Blood Pressure Monitor (Kyoto, Japan) on the right arm, which was supported at the heart level. After a 5-min rest period, BP was measured 4 times, and SBP and DBP were calculated as the average of the last 3 readings. Waistline and hipline were measured using an inelastic measuring tape. Body mass index (BMI) was calculated as the weight in kilograms divided by height in meters squared (kg/m2).
The levels of plasma tHcy, fasting blood glucose (FBG), total cholesterol (TC), total triglyceride (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), serum uric acid and creatinine, blood urea nitrogen (BUN), total and direct bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) were assessed using biochemical analysis instruments (Beckman Coulter). tHcy was quantified by high-performance liquid chromatography using a fluorescence detector [18]. All laboratory measurements were performed according to the criteria of a standardization and certification program. In our study, HHcy was defined as Hcy >15 μmol/L, in consideration of the fact that the ratio of tHcy levels ≤10 μmol/L was 1.16%. T2DM was defined as FBG levels over 7.0 mmol/L or on appropriate hypoglycemic medication [17]. Dyslipidemia was defined as having 1 of the following features: elevated TG (≥2.3 mmol/L), elevated TC (≥6.2 mmol/L), elevated LDL-C (≥4.1 mmol/L), and reduced HDL-C (<1.0 mmol/L) or on appropriate lipid-lowering medication [19]. The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation was used to evaluate estimated glomerular filtration rate (eGFR) [20].
DEFINITION OF MS:
Although the definition of MS is ambiguous, the most popular definition of MS consider ethnicity for assessing waistline, given differences in visceral adipose tissue [16,21]. In our study, MS was defined according to the Chinese Diabetes Society (CDS) criterion [17], as presence of at least 3 of the following 5 criteria: abdominal obesity (waistline ≥90 cm in adult male and 85 cm in adult female subjects), elevated blood pressure (SBP ≥130 mmHg and/or DBP ≥85 mmHg, or on treatment for diagnosed hypertension formerly), hyperglycemia (FBG ≥6.1 mmol/L, or on appropriate hypoglycemic medication), elevated TG (≥1.7 mmol/L, or on appropriate lipid-lowering medication) and reduced HDL-C (<1.03 mmol/L in adult male and <1.30 mmol/L in adult female subjects, or on appropriate lipid-lowering medication). In addition, considering this distinction of sex-specific reduced HDL-C criterion at current popular international guidelines [16], we did not choose the threshold of <1.04 mmol/L for definition of reduced HDL-C according to the CDS criterion in our study [17].
STATISTICAL ANALYSIS:
Continuous variables are presented as the mean±standard deviation or the median (IQR), as appropriate, and were compared using the
Multivariable logistic regression analyses were used to examine the associations of tHcy or (and) MS with prevalence of stroke. Model I was only adjusted for age and sex. Model II (confounder model) screened covariates including age, sex, BMI, heart rate, smoking and drinking status, tHcy, AST, ALT, serum uric acid, eGFR, total and direct bilirubin, abdominal obesity, hyperglycemia, elevated TG, and reduced HDL-C, except for the independent variable itself. We selected these confounders on the basis of a more than 10% association with the outcomes of interest. Supplementary Table 1 show the associations of each confounder with stroke occurrence. We considered the confounder model to be the main model. In addition, the generalized additive model and smooth curve fitting (penalized spline method) were used to visually show the relationship between the subduplicate Hcy levels and stroke probability. In addition, subgroup analysis was executed by stratified and interaction test to investigate the robustness in this association.
The R statistical package (Te R Foundation;
Results
CLINICAL CHARACTERISTICS:
This study analyzed 14 059 adults with elevated blood pressure (age: 63.76±9.35 years, range 27–96 years; male, 47.30%), and the prevalence rates of HHcy and MS were 49.96% and 42.21%, respectively (Table 1). The participants with stroke were more likely to be older, male, smoker; had lower BMI, SBP, DBP, TC, TG, HDL-C, LDL-C, eGFR, and AST, had higher levels of tHcy and serum creatinine, had a higher prevalence of current T2DM, elevated TG, reduced HDL-C, dyslipidemia, and MS, and had a lower prevalence of current abdominal obesity compared to those without stroke (P<0.05).
ASSOCIATIONS BETWEEN PLASMA THCY LEVELS OR MS AND CEREBRAL STROKE:
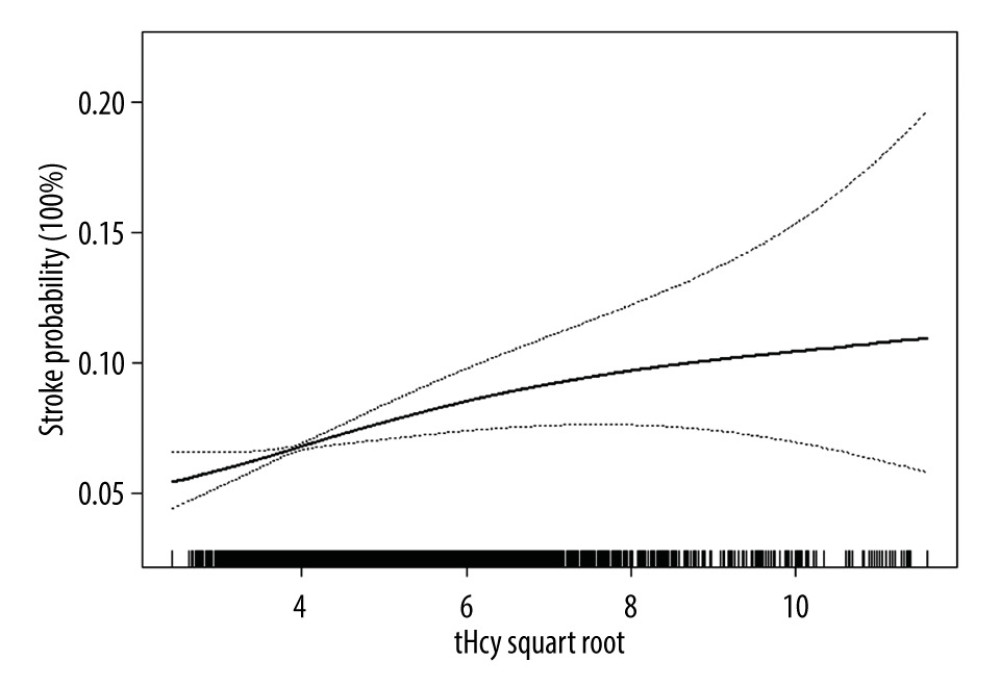
The multivariable analyses indicated that plasma tHcy level was positively associated with the prevalence of stroke (adjusted odds ratio [OR] per SD increase: 1.08, 95% CI 1.02 to 1.14, P=0.010; Table 2). Compared to the group without HHcy, participants with HHcy had a higher prevalence of stroke (adjusted-OR: 1.36, 95% CI 1.17 to 1.58, P<0.001). Compared to tHcy concentrations ≤12.46 μmol/L, there were significantly higher prevalence rates of stroke in the third and highest tHcy quartiles (adjusted-OR: 1.37, 95% CI 1.11 to 1.69, P=0.003; adjusted-OR: 1.49, 95% CI 1.19 to 1.85, P <0.001, respectively; P for trend < 0.001). The generalized additive model and penalized spline method were used to find a positive relationship between the subduplicate tHcy levels and stroke probability (Figure 1).
Similarly, the multivariable analyses also showed that, compared to the group without MS, participants with MS had a higher prevalence of stroke (adjusted-OR: 1.68, 95% CI 1.44 to 1.96, P<0.001; Table 2). When the group with only 1 component (hypertension) of MS was used as the reference, a statistically significant higher prevalence of stroke was found in the group with 3, 4 or 5 components of MS (adjusted-OR: 1.74, 95% CI 1.41 to 2.13, P<0.001; adjusted-OR: 1.99, 95% CI 1.56 to 2.54, P<0.001; adjusted-OR: 2.60, 95% CI 1.84 to 3.67, P<0.001, respectively; P for trend <0.001; Table 2).
ASSOCIATION BETWEEN HHCY COMBINED WITH MS AND STROKE RISK IN HYPERTENSIVE PATIENTS:
Clinical characteristics of participants grouped by HHcy and MS are presented in Supplementary Table 2. Compared to the control group, the prevalence of stroke was markedly increased in the isolated HHcy group, isolated MS group, and combined HHcy and MS group (adjusted-OR: 1.32, 95% CI 1.07 to 1.62, P=0.008; adjusted-OR: 1.78, 95% CI 1.41 to 2.23, P<0.001; adjusted-OR: 2.46, 95% CI 1.97 to 3.08, P <0.001, respectively; P for trend <0.001; Supplementary Table 3). Individuals with both HHcy and MS had increased odds of stroke than those with only HHcy or only MS (adjusted-OR: 1.78, 95% CI 1.47 to 2.15, P<0.001; adjusted-OR: 1.39, 95% CI 1.13 to 1.70, P=0.002, respectively; Table 3).
SUBGROUP ANALYSES BY POTENTIAL EFFECT MODIFIERS:
To explore whether the association between HHcy combined with MS and stroke risk in hypertensive patients was still stable, we conducted subgroup analyses, which suggested that there were no interactions in any of the subgroups, including age (<60 vs ≥60 years), sex (male vs female), smoking habit (no vs yes), drinking habit (no vs yes), BMI (≤24 vs >24 kg/m2), and eGFR (≥60 vs <60 ml/min) (all P for interaction >0.05; Supplementary Figure 2).
Discussion
The findings of this research demonstrate that HHcy and MS are independently and positively associated with the prevalence of cerebral stroke in Chinese adults with elevated blood pressure. Homocysteine is a non-essential, possibly cytotoxic amino acid and HHcy caused by its metabolic imbalance is an independent risk factor for various vascular diseases [7,14], especially stroke [5,6,22,23]. Many possible mechanisms linking HHcy with atherogenesis have been suggested, including pro-thrombotic and pro-inflammatory effects, endothelial dysfunction, and smooth muscle cell proliferation [24]. Celermajer et al [25] found that HHcy was associated with impaired endothelial function of systemic arteries and decreased nitric oxide bioavailability in hyperhomocystinuria children. Cheng et al [26] found that HHcy impaired endothelium-dependent micro-vasculature relaxation to acetylcholine in mouse small mesenteric artery via oxidation of SKCa/IKCa. Intermediate HHcy potentiated diabetes impaired endothelium-derived hyperpolarizing factor (EDHF)-induced microvasculature relaxation via hydrogen sulfide (H2S)-downregulation and IKCa tyrosine nitration in T2DM/HHcy mice [27]. Recent research showed HHcy can cause mitochondrial dysfunction and systemic tissue damage, mostly by inhibiting mitochondrial complex I gene expression, which leads to impaired electron transfer, complex assembly, and redox homeostasis dysregulation in human disease and experimental models [28]. Therefore, we speculate that HHcy can cause intracranial vascular endothelial dysfunction and tissue damage through oxidative stress and mitochondrial dysfunction, which can lead to a stroke.
MS is associated with significantly higher risk of CVD such as CHD, stroke, and peripheral arterial diseases [9,16]. Similar to previous reports, we also found that the participants with MS were inclined to be female (non-MS vs MS: 45.2% vs 63.0%) and above the age of 50 years due to disappearance of estrogen protection after menopause [13,29], and had a higher prevalence of formerly cerebral stroke and current T2DM [9] (Supplementaty Table 1). Moreover, MS is independently associated with higher prevalence of stroke.
The molecular mechanisms underlying IR, MS, and CVD are intricate and confusing. IR is triggered by cellular inflammation, lipotoxicity, mitochondrial dysfunction, and endoplasmic stress, which lead to insulin signal transduction disorder, dysregulation of genes, and modifications of regulatory proteins [16]. Central obesity, with disturbances in lipid metabolism with deposition of fat in the waist and internal organs, is also linked to IR and CVD [30]. Dyslipidemia characterized by elevated TG and lowered HDL-C increased the risk for CVD in obese children with MS [31,32]. Vascular IR is a potential link between metabolic and CVD. In IR, the effect of insulin on endothelial cells is minimized with reduced nitric oxide production, making endothelial cells more susceptible to oxidized lipid-induced injury and atherogenesis [33]. In patients with IR, the process of lipolysis within adipose tissue is accelerated, resulting in increased free fatty acid release into the portal circulation. Moreover, macrophage infiltration and polarization (increased ratio of M1 to M2) of adipose tissue, altered immune response, and imbalance in the synthesis of pro- and anti-inflammatory cytokines affect the activity of insulin in the liver and muscles [34,35].
Previous study has found that patients with MS and HHcy or smoking are at particularly high cardiovascular risk compared with those with MS but with normal tHcy levels [36]. There was an interaction reported between HHcy and MS in individuals at risk for atherosclerosis [36]. In our research, those individuals with combined HHcy and MS had higher odds of stroke than those with only HHcy or only MS in Chinese adults with elevated blood pressure, thus showing an association between HHcy combined with MS and stroke risk in hypertensive patients.
Targeted stroke prevention should include healthy lifestyle improvement and treatment of MS and HHcy in an individualized and comprehensive way [37]. Among Chinese hypertensive adults without a history of CVD but with low baseline folate levels, folic acid supplementation significantly reduced the risk of first stroke [38], and elevated total cholesterol levels modified this benefit [39]. The reduction of stroke was proportional to the reduction of tHcy [40], and the reduction of stroke was greater in participants with HHcy [41] or elevated LDL-C [39]. Notably, folic acid therapy was beneficial among those with impaired renal function [42]. Although B vitamin therapy might reduce the stroke risk, this benefit might have been obscured in early trials by increased risk of CVD in participants with renal failure receiving high-dose cyanocobalamin [43]. In countries where folate fortification is in place, vitamin B12 deficiency is the main nutritional determinant of HHcy [44,45]. The potential benefits of methylcobalamin supplementation with folic acid therapy to lower plasma tHcy levels in populations at high risk of stroke warrant further investigation [43].
However, in the Insulin Resistance Intervention after Stroke (IRIS) randomized clinical trial, pioglitazone reduced new-onset diabetes by half and reduced stroke or myocardial infarction by 24% over 5 years among patients with IR [46], particularly in those with good adherence [47]. Pioglitazone also significantly lowered BP and TG and raised HDL-C, indicated that the benefits from this drug outweigh the risks of fracture and fluid retention [47]. Therefore, treatment with pioglitazone in patients with MS needs to be reconsidered given the large effects of pioglitazone in prediabetes [48].
This study had several limitations. The hypertensive population at Wuyuan had relatively higher and wider tHcy concentrations (median: 14.99 μmol/L), which may have led to differences in odds ratios of tHcy on the prevalence of stroke compared to other studies [49,50]. Additionally, we cannot draw any causal relationship between HHcy, MS, and stroke considering the cross-sectional design. Moreover, the subtype of stroke was not exactly classified due to the lack of imaging evidence originally. Finally, we did not measure the levels of glycated hemoglobin and insulin, which could have better evaluated the glycometabolism of our hypertensive patients.
Conclusions
HHcy and MS are independently associated with higher prevalence of stroke in Chinese adults with elevated blood pressure. Those with combined HHcy and MS had increased odds of stroke than those with only HHcy or only MS. We found an association between HHcy combined with MS and stroke risk in hypertensive patients.
Tables
Table 1. Clinical characteristics of participants grouped by stroke.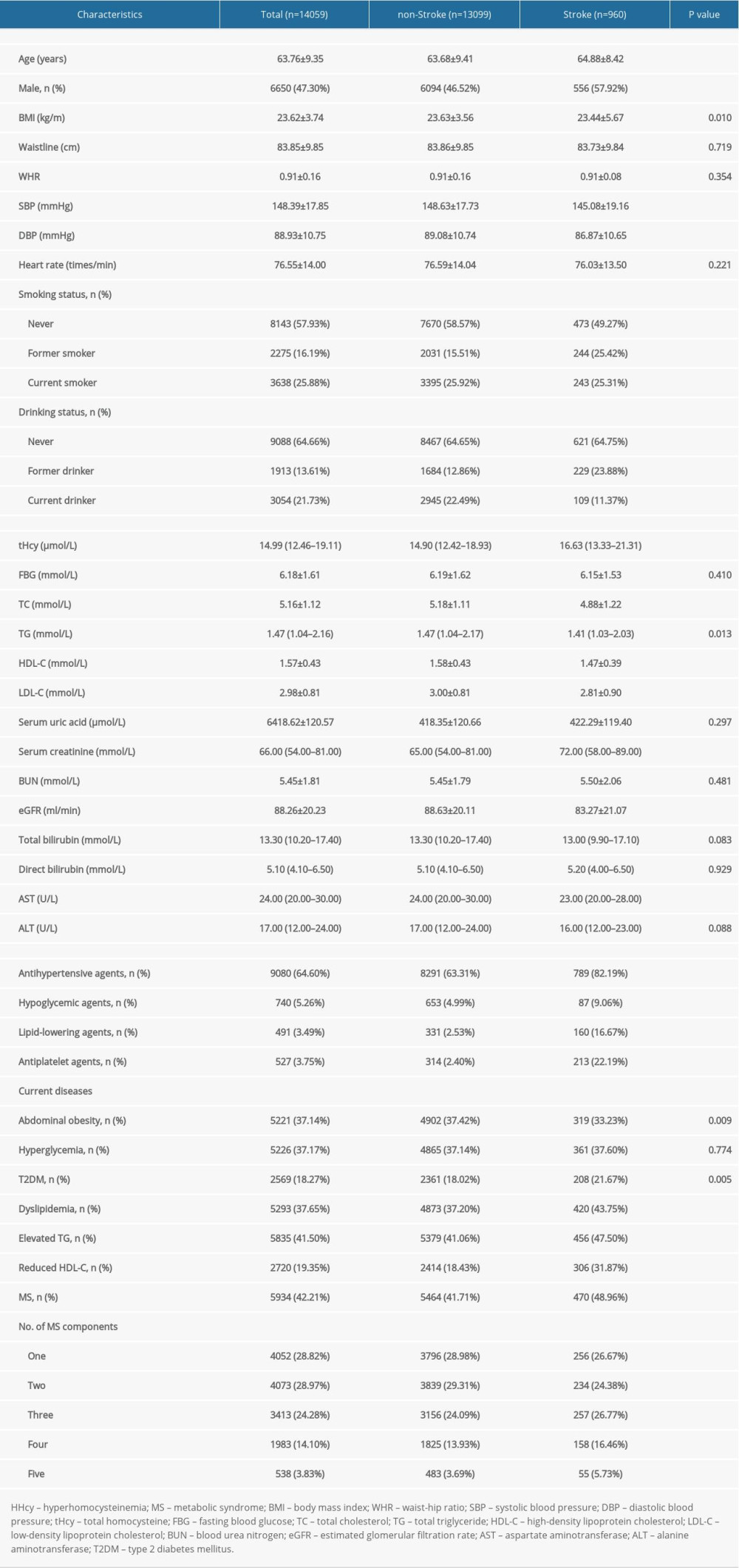 Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models.
Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models.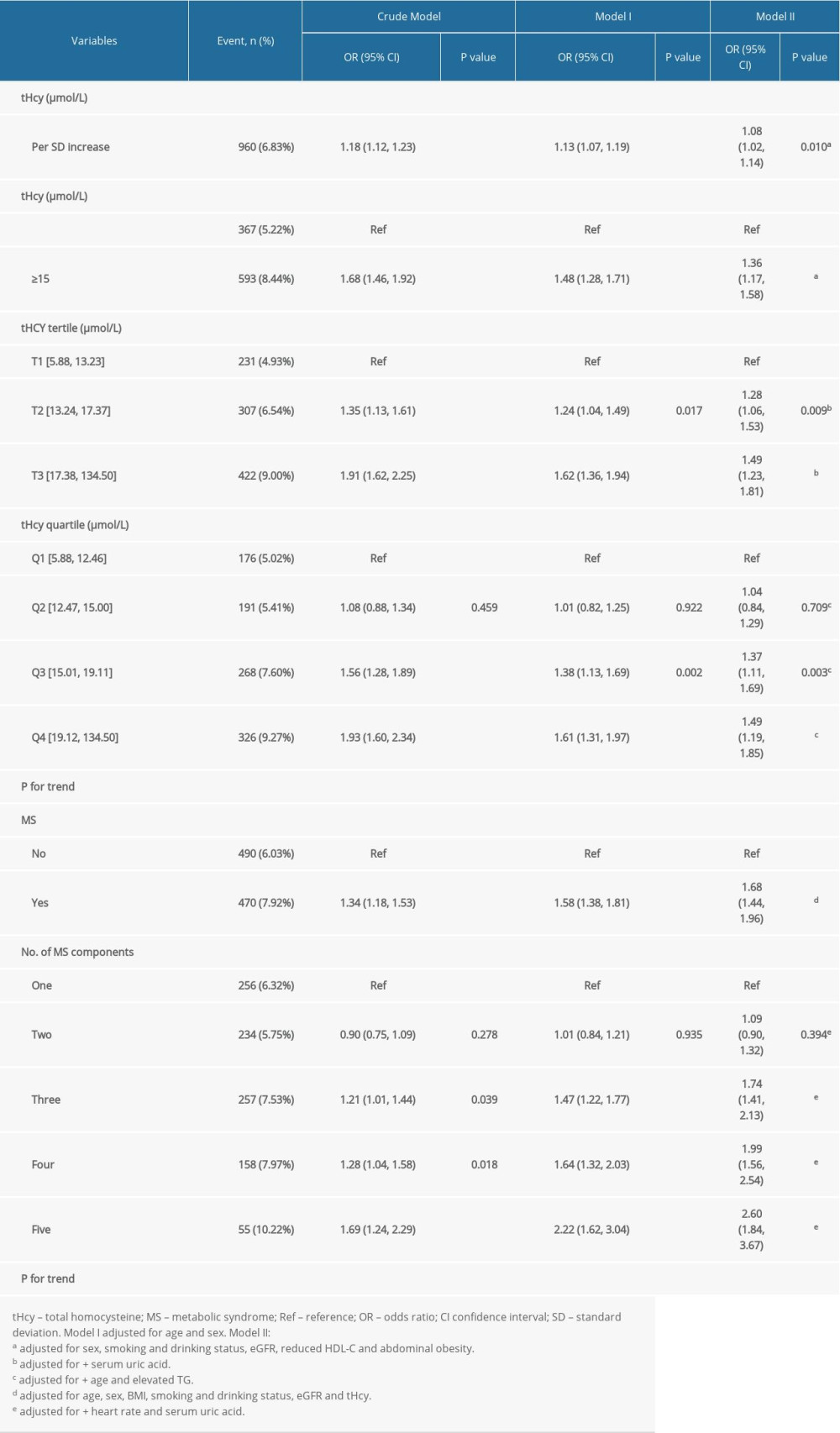 Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms.
Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms.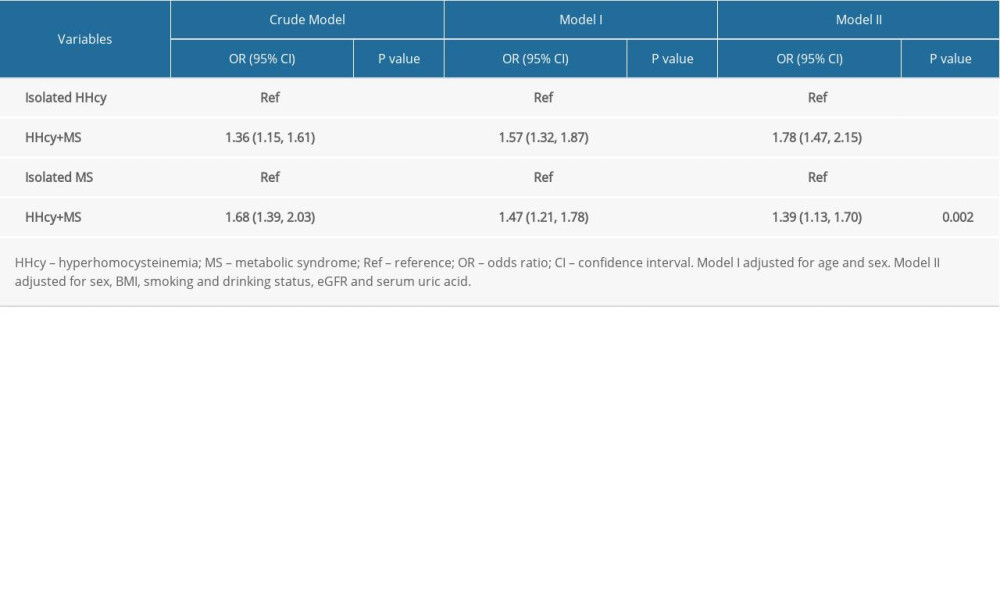 Supplementary Table 1. Associations of covariates with stroke occurrence.
Supplementary Table 1. Associations of covariates with stroke occurrence.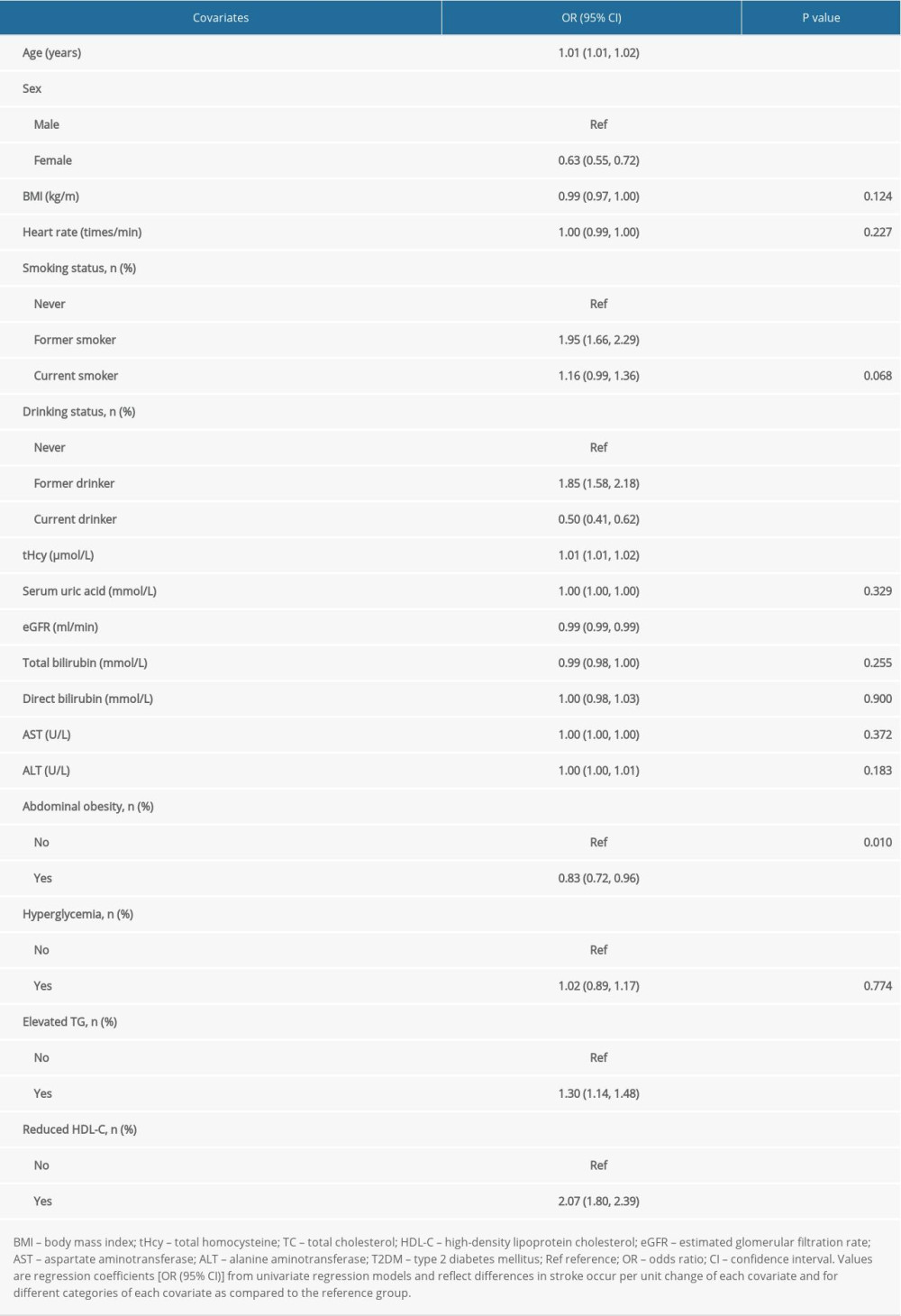 Supplementary Table 2. Clinical characteristics of participants grouped by HHcy and MS.
Supplementary Table 2. Clinical characteristics of participants grouped by HHcy and MS.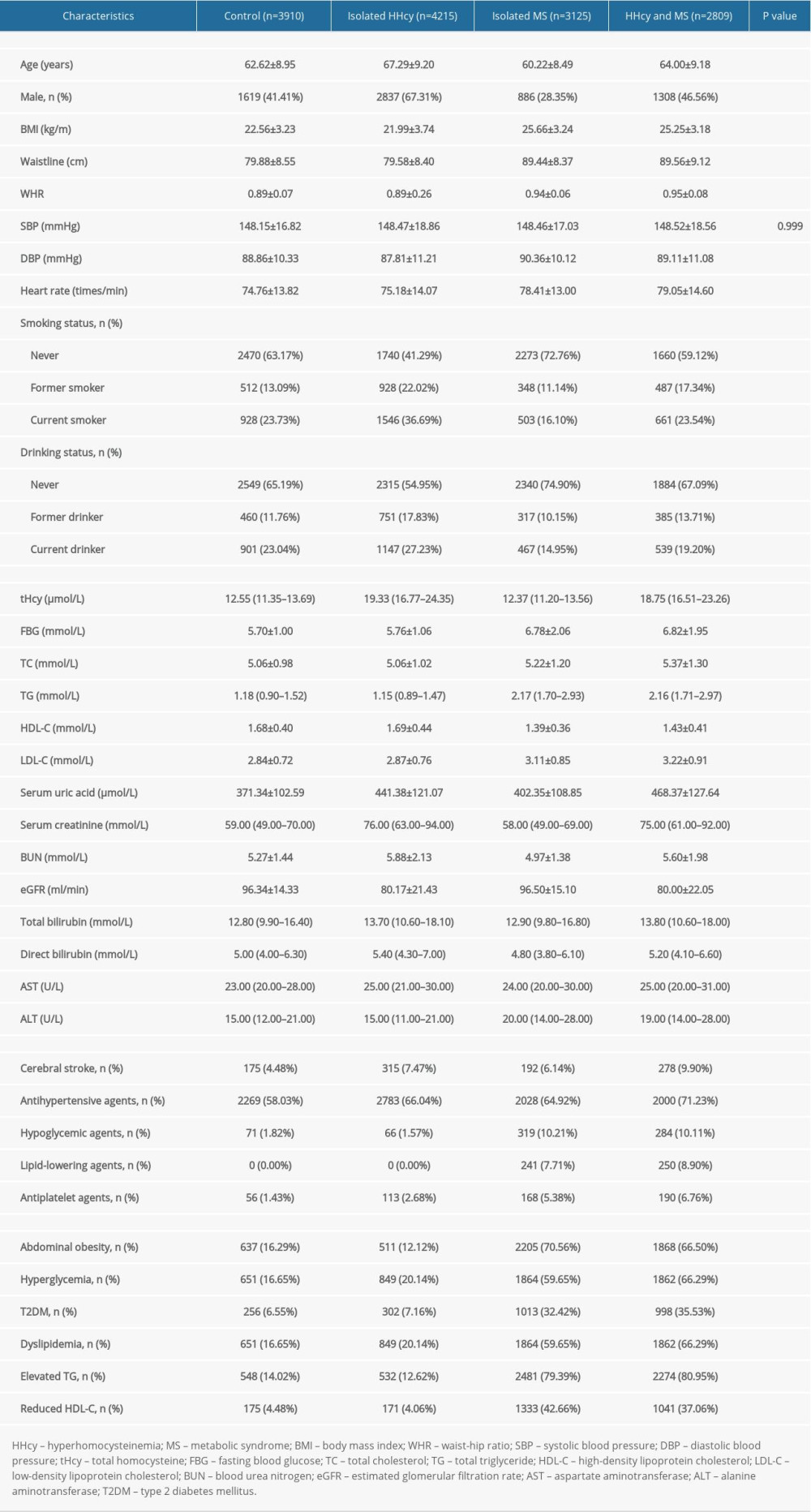 Supplementary Table 3. Effect size of HHcy and MS on cerebral stroke in different models.
Supplementary Table 3. Effect size of HHcy and MS on cerebral stroke in different models.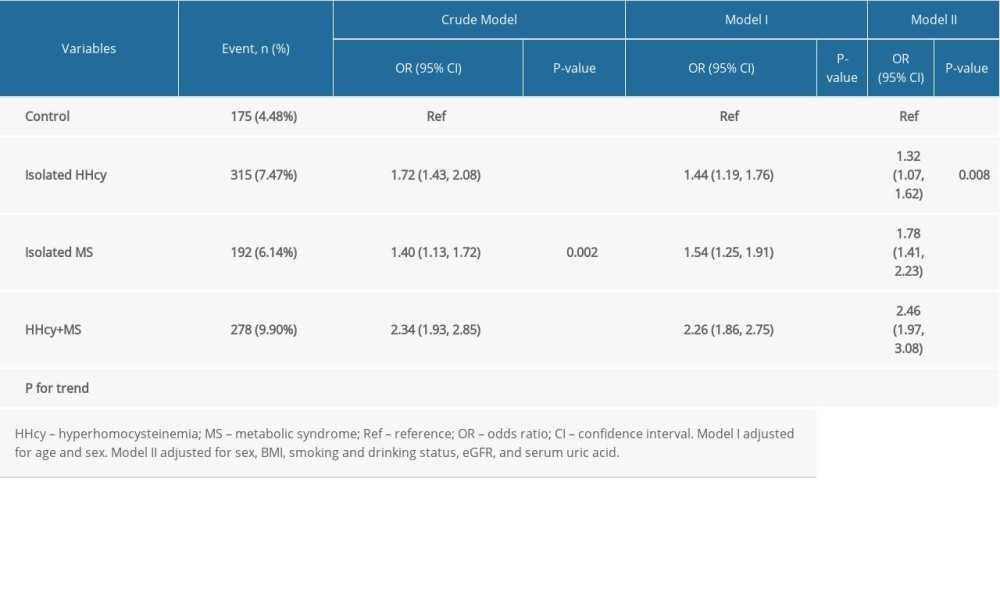
References
1. Virani SS, Alonso A, Benjamin EJ, Heart Disease and Stroke Statistics – 2020 update: A Report from the American Heart Association: Circulation, 2020; 141; 139-596
2. Wang W, Jiang B, Sun H, Prevalence, incidence, and mortality of stroke in China: Results from a nationwide population-based survey of 480 687 adults: Circulation, 2017; 135; 759-71
3. Ji C, Kaplowitz N, Hyperhomocysteinemia, endoplasmic reticulum stress, and alcoholic liver injury: World J Gastroenterol, 2004; 10; 1699-708
4. Qin X, Huo Y, H-Type hypertension, stroke and diabetes in China: Opportunities for primary prevention: J Diabetes, 2016; 8; 38-40
5. Gungor L, Polat M, Ozberk MB, Which ischemic stroke subtype is associated with hyperhomocysteinemia?: J Stroke Cerebrovasc Dis, 2018; 27; 1921-29
6. Larsson SC, Traylor M, Markus HS, Homocysteine and small vessel stroke: A mendelian randomization analysis: Ann Neurol, 2019; 85; 495-501
7. Fu Y, Wang X, Kong W, Hyperhomocysteinaemia and vascular injury: Advances in mechanisms and drug targets: Br J Pharmacol, 2018; 175; 1173-89
8. Wang Z, Chen Z, Zhang L, Status of hypertension in China: Results from the China hypertension survey, 2012–2015: Circulation, 2018; 137; 2344-56
9. Alberti KG, Zimmet P, Shaw J, The metabolic syndrome – a new worldwide definition: Lancet, 2005; 366; 1059-62
10. Weiss R, Bremer AA, Lustig RH, What is metabolic syndrome, and why are children getting it?: Ann N Y Acad Sci, 2013; 1281; 123-40
11. Aguilar M, Bhuket T, Torres S, Prevalence of the metabolic syndrome in the United States, 2003–2012: JAMA, 2015; 313; 1973-74
12. O’Neill S, O’Driscoll L, Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies: Obes Rev, 2015; 16; 1-12
13. Li W, Song F, Wang X, Prevalence of metabolic syndrome among middle-aged and elderly adults in China: Current status and temporal trends: Ann Med, 2018; 50; 345-53
14. Catena C, Colussi G, Nait F, Elevated homocysteine levels are associated with the metabolic syndrome and cardiovascular events in hypertensive patients: Am J Hypertens, 2015; 28; 943-50
15. Li M, Zhan A, Huang X, Positive association between triglyceride glucose index and arterial stiffness in hypertensive patients: The China H-type Hypertension Registry Study: Cardiovasc Diabetol, 2020; 19; 139
16. Zafar U, Khaliq S, Ahmad HU, Metabolic syndrome: An update on diagnostic criteria, pathogenesis, and genetic links: Hormones, 2018; 17; 299-313
17. Jia W, Weng J, Zhu D, Standards of medical care for type 2 diabetes in China 2019: Diabetes Metab Res Rev, 2019; 35; e3158
18. Minniti G, Piana A, Armani U, Determination of plasma and serum homocysteine by high-performance liquid chromatography with fluorescence detection: J Chromatogr A, 1998; 828; 401-5
19. Chinese guidelines on prevention and treatment of dyslipidemia in adults: Zhonghua Xin Xue Guan Bing Za Zhi, 2007; 35(5); 390-419 [in Chinese]
20. Levey AS, Stevens LA, Schmid CH, A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12
21. Lear SA, Gasevic D, Ethnicity and metabolic syndrome: Implications for assessment, management and prevention: Nutrients, 2019; 12; 15
22. Hu DY, Xu XPPrevention of stroke relies on valid control “H” type hypertension: Zhonghua Xin Xue Guan Bing Za Zhi, 2008; 47; 976-77 [in Chinese]
23. Zhang Q, Qiu DX, Fu RL, H-type hypertension and C reactive protein in recurrence of ischemic stroke: Int J Environ Res Public Health, 2016; 13; 477
24. Hayden MR, Tyagi SC, Homocysteine and reactive oxygen species in metabolic syndrome, type 2 diabetes mellitus, and atheroscleropathy: The pleiotropic effects of folate supplementation: Nutr J, 2004; 3; 4
25. Celermajer DS, Sorensen K, Ryalls M, Impaired endothelial function occurs in the systemic arteries of children with homozygous homocystinuria but not in their heterozygous parents: J Am Coll Cardiol, 1993; 22; 854-58
26. Cheng Z, Jiang X, Kruger WD, Hyperhomocysteinemia impairs endothelium-derived hyperpolarizing factor-mediated vasorelaxation in transgenic cystathionine beta synthase-deficient mice: Blood, 2011; 118; 1998-2006
27. Cheng Z, Shen X, Jiang X, Hyperhomocysteinemia potentiates diabetes-impaired EDHF-induced vascular relaxation: Role of insufficient hydrogen sulfide: Redox Biol, 2018; 16; 215-25
28. Cueto R, Zhang L, Shan HM, Identification of homocysteine-suppressive mitochondrial ETC complex genes and tissue expression profile – novel hypothesis establishment: Redox Biol, 2018; 17; 70-88
29. Pucci G, Alcidi R, Tap L, Sex- and gender-related prevalence, cardiovascular risk and therapeutic approach in metabolic syndrome: A review of the literature: Pharmacol Res Pharmacol Res, 2017; 120; 34-42
30. Cornier MA, Dabelea D, Hernandez TL, The metabolic syndrome: Endocr Rev, 2008; 29; 777-822
31. Urbina EM, Khoury PR, McCoy CE, Triglyceride to HDL-C ratio and increased arterial stiffness in children, adolescents, and young adults: Pediatrics, 2013; 131; e1082-90
32. Burns SF, Lee SJ, Arslanian SA, Surrogate lipid markers for small dense low-density lipoprotein particles in overweight youth: J Pediatr, 2012; 161; 991-96
33. Schulman IH, Zhou MS, Vascular insulin resistance: A potential link between cardiovascular and metabolic diseases: Curr Hypertens Rep, 2009; 11; 48-55
34. Gustafson B, Hammarstedt A, Andersson CX, Inflamed adipose tissue: A culprit underlying the metabolic syndrome and atherosclerosis: Arterioscler Thromb Vasc Biol, 2007; 27; 2276-83
35. Heilbronn LK, Campbell LV, Adipose tissue macrophages, low grade inflammation and insulin resistance in human obesity: Curr Pharm Des, 2008; 14; 1225-30
36. Azarpazhooh MR, Andalibi MSS, Hackam DG, Interaction of smoking, hyperhomocysteinemia, and metabolic syndrome with carotid atherosclerosis: A cross-sectional study in 972 non-diabetic patients: Nutrition, 2020; 79–80; 110874
37. Pandian JD, Gall SL, Kate MP, Prevention of stroke: A global perspective: Lancet, 2018; 392; 1269-78
38. Huo Y, Li J, Qin X, Efficacy of folic acid therapy in primary prevention of stroke among adults with hypertension in China: The CSPPT randomized clinical trial: JAMA, 2015; 313; 1325-35
39. Qin X, Li J, Spence JD, Folic acid therapy reduces the first stroke risk associated with hypercholesterolemia among hypertensive patients: Stroke, 2016; 47; 2805-12
40. Huang X, Li Y, Li P, Association between percent decline in serum total homocysteine and risk of first stroke: Neurology, 2017; 89; 2101-741
41. Kong X, Huang X, Zhao M, Platelet count affects efficacy of folic acid in preventing first stroke: J Am Coll Cardiol, 2018; 71; 2136-46
42. Xu X, Qin X, Li Y, Efficacy of folic acid therapy on the progression of chronic kidney disease: The renal substudy of the China Stroke Primary Prevention Trial: JAMA Intern Med, 2016; 176; 1443-50
43. Spence JD, Yi Q, Hankey GJ, B vitamins in stroke prevention: Time to reconsider: Lancet Neurol, 2017; 16; 750-60
44. Robertson J, Iemolo F, Stabler SP, Vitamin B12, homocysteine and carotid plaque in the era of folic acid fortification of enriched cereal grain products: CMAJ, 2005; 172; 1569-73
45. Mathiesen EB, Johnsen SH, Wilsgaard T, Carotid plaque area and intima-media thickness in prediction of first-ever ischemic stroke: A 10-year follow-up of 6584 men and women: the Tromsø Study: Stroke, 2011; 42; 972-78
46. Kernan WN, Viscoli CM, Furie KL, Pioglitazone after ischemic stroke or transient ischemic attack: N Engl J Med, 2016; 374; 1321-31
47. Spence JD, Viscoli CM, Inzucchi SE, Pioglitazone therapy in patients with stroke and prediabetes: A post hoc analysis of the IRIS randomized clinical trial: JAMA Neurol, 2019; 76; 526-35
48. Pantoni L, Potential new horizons for the prevention of cerebrovascular diseases and dementia: JAMA Neurol, 2019; 76; 521-22
49. Wang CY, Chen ZW, Zhang T, Elevated plasma homocysteine level is associated with ischemic stroke in Chinese hypertensive patients: Eur J Intern Med, 2014; 25; 538-44
50. Pang H, Han B, Fu Q, Association between homocysteine and conventional predisposing factors on risk of stroke in patients with hypertension: Sci Rep, 2018; 8; 3900
Tables
 Table 1. Clinical characteristics of participants grouped by stroke.
Table 1. Clinical characteristics of participants grouped by stroke. Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models.
Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models. Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms.
Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms. Table 1. Clinical characteristics of participants grouped by stroke.
Table 1. Clinical characteristics of participants grouped by stroke. Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models.
Table 2. Relationship between plasma tHcy levels or MS and the prevalence of stroke in different models. Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms.
Table 3. Effect size of combined HHcy and MS on the prevalence of stroke in different models compared to its isolated forms. Supplementary Table 1. Associations of covariates with stroke occurrence.
Supplementary Table 1. Associations of covariates with stroke occurrence. Supplementary Table 2. Clinical characteristics of participants grouped by HHcy and MS.
Supplementary Table 2. Clinical characteristics of participants grouped by HHcy and MS. Supplementary Table 3. Effect size of HHcy and MS on cerebral stroke in different models.
Supplementary Table 3. Effect size of HHcy and MS on cerebral stroke in different models. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952