03 November 2021: Review Articles
A Review of the Role of Hypoxia in Radioresistance in Cancer Therapy
Wafa BouleftourDOI: 10.12659/MSM.934116
Med Sci Monit 2021; 27:e934116
Abstract
ABSTRACT: Hypoxia involves neoplastic cells. Unlike normal tissue, solid tumors are composed of aberrant vasculature, leading to a hypoxic microenvironment. Hypoxia is also known to be involved in both metastasis initiation and therapy resistance. Radiotherapy is the appropriate treatment in about half of all cancers, but loco-regional control failure and a disease recurrence often occur due to clinical radioresistance. Hypoxia induces radioresistance through a number of molecular pathways, and numerous strategies have been developed to overcome this. Nevertheless, these strategies have resulted in disappointing results, including adverse effects and limited efficacy. Additional clinical studies are needed to achieve a better understanding of the complex hypoxia pathways. This review presents an update on the mechanisms of hypoxia in radioresistance in solid tumors and the potential therapeutic solutions.
Keywords: Cancer, Familial, with In Vitro Radioresistance, Hypoxia-Inducible Factor 1, Radiotherapy, Female, Humans, Male, Neoplasms, Radiation Tolerance
Background
The tumor microenvironment is composed of extracellular matrix, blood vessels, signaling molecules, and non-malignant cells such as stromal cells, fibroblasts, and immune cells [1]. Unlike normal tissue, solid tumors contain dysfunctional vasculature. Stroma composition, rate of tumor growth, and a pathological vasculature lead to a hypoxic environment and impede immune cell functions in tumors. Moreover, hypoxia causes a selection pressure promoting growth of cells with genetic machinery for malignant potential [2]. Therefore, hypoxia leads to epithelial-to-mesenchymal cell transition (EMT), resulting in cell mobility and thus metastasis [3]. Furthermore, hypoxia often alters tumor cell metabolism by inducing cell quiescence. It also induces an alteration in transport and/or distribution of chemotherapy, immunotherapy, and radiotherapy, leading to resistance to therapies [4]. While proliferating tumor cells in normoxic regions of the tumor respond to chemotherapy and radiotherapy, hypoxic cells manage to survive these antineoplastic therapies; thus, they multiply, which leads to treatment failure [5]. In addition, tumor cell sensitivity to X-rays is about 3 times greater when irradiated in a well-oxygenated environment vs under anoxic conditions [6,7].
Hypoxia induces: (1) increased potential of malignancy [8], (2) resistance to chemotherapy, immunotherapy, and radiation therapies [9,10], and (3) increased metastasis occurrence, which all lead to a poor prognosis [11]. Elucidating these resistance mechanisms would therefore improve cancer patients’ outcomes.
Extensive studies have focused on the impact of this hostile environment on tumor cells, therapeutic responses, and clinical outcomes. Many strategies have been developed to overcome this feature [12,13]. This review presents an update on the mechanisms of hypoxia in radioresistance in solid tumors, and presents some potential therapeutic solutions.
Hypoxia-Regulated Programs in Cancer
When pO2 level is below 10 mmHg, solid cell tumors grow rapidly due to hypoxia, which has long been associated with chemotherapy and radiotherapy resistance [14,15]. The proportion of hypoxic cells is heterogeneous within the tumor mass and among tumors. In normal tissues, consumption and supply of oxygen are balanced, whereas this balance is disrupted in tumoral tissue. Indeed, within this disordered development of cancer cells and expanding tumor tissue, oxygen demand is surpassed by oxygen supply. Therefore, a combination of multiple factors, as well as the increasing vasculature, prevents oxygen diffusion, leading to an increase of hypoxic region area [3,16].
In solid tumors, 2 subtypes of tumor hypoxia are well documented. Acute and chronic hypoxia leads to different hypoxia-related responses within the tumor [17]. Acute hypoxia or perfusion-limited hypoxia is mainly due to a transient occlusion, narrowing of vessels, and arteriolar vasomotion, which cause local disturbances in perfusion and thus a disruption in oxygen supply [18]. Conversely, chronic hypoxia, also called diffusion-limited hypoxia, is caused by a critical limitation in oxygen diffusion from tumor microvessels to surrounding tissues [18]. Anemic hypoxia is due to the decreased oxygen transport induced by chemotherapy [19,20]. Acute and chronic hypoxia used to be considered as having similar cellular biological effects, but recent analyses have proved otherwise. Although there is no consensus, acute hypoxia is associated with a more aggressive phenotype by increasing cell migration and leading to possible tumor metastasis [17]. Cairns et al studied the contribution of acute and chronic hypoxia in the process of lung metastases in murine models of fibromas [21]. Mice with acute hypoxia (intermittently inhaling 5–7% oxygen for 10 min 12 times a day) had significantly more pulmonary metastasis than in the control group (inhaling normal air) [21]. Conversely, there was no significant difference between mice exposed to chronic hypoxia (inhaling 5–7% oxygen for 2 h each day) and the control group [21].
At the cellular level, the effects of hypoxia on tumor cells are multiple and ambivalent. Indeed, hypoxia causes proteomic modification by stopping the cell cycle and thus causing necrosis and apoptosis [22], or induces proteomic changes by stimulating tumor growth, invasion, and metastasis [23]. At the molecular level, transcription factors such as hypoxia-induced factors (HIFs) play an essential role in these shifts. Under hypoxic conditions, the HIF-1α subunit translocates to the nucleus and binds HIF-1β, which results in transcriptional activation of many genes and plays a role in tumor progression. Thus, HIF-1 induces the expression of angiogenic vascular endothelial growth factor (VEGF), which is one of the most important stimulators of angiogenesis. Glucose transporters like GLUT-1 facilitate anaerobic glycolysis metabolism and enable cancer cell survival by limiting sufficient ATP (Adenosine 5-triphosphate) production and hematopoietic factors such as erythropoietin (EPO) [24–26].
HIF-1α is overexpressed in the vast majority of patients with squamous cell cancer of the oropharynx. Its degree of expression has predictive and prognostic significance in patients undergoing curative radiation therapy [27]. For breast and endometrioid ovarian cancer, carbonic anhydrase IX (CAIX) is a biomarker of poor prognosis for metastasis and survival. CAIX is a hypoxia-inducible protein that regulates cellular pH to promote cancer cell survival and invasion in hypoxic microenvironments [28,29]. Therefore, the evaluation of CAIX in breast cancer is a robust indicator of hypoxia.
Tumor Hypoxia and Radiation Efficacy
In 1909, Schwarz demonstrated that skin response to radiation decreased when blood flow in the irradiated area was compressed. Mottram, Crabtree, and Cramer and many others later explored the issue of oxygen and radiation effects more fully [30]. Indeed, they highlighted that cells were much more sensitive to X-rays in the presence of oxygen [30]. This was measured by the oxygen enhancement ratio (OER). OER is a ratio of radiation doses delivered under hypoxic to aerobic conditions to achieve the same biological endpoint. Indeed, hypoxic cells required higher radiation doses to reach the same death rate as normoxic cells. In hypoxic areas, the ability of tumor cells to survive ionizing radiation was 2–3 times higher than in normoxic areas [31]. Concentration and time of exposure to oxygen are crucial. Thus, to produce an apoptosis effect, molecular oxygen must be present during radiotherapy treatment or at least during the lifetime of the free radicals generated by the radiation. Only a small quantity of oxygen was required for radiosensitization; 0.5% oxygen, pO2 of about 3 mmHg [32].
Hypoxic tumors are more resistant to radiotherapy because of the absence of oxygen. Radiation can directly create DNA (deoxyribonucleic acid) strand breaks and induce tumor cell death. Water radiolysis induced the formation of reactive oxygen species (ROS; eg, hydroxyl free radical and superoxide radical) and provoked DNA cells damage [33]. Multiple unrepaired DNA damage then results in cell death. In an oxygen-free environment, ROS formation is limited and leads to DNA damage. Moreover, oxygen reacts with fractured DNA to generate stable peroxides and prevent cells from repairing DNA in an aerobic environment. In a hypoxic environment, tumor cells remove hydrogen from free sulfhydryl groups and are then able to repair DNA damage [31].
In addition, tumor radiosensitivity depends on cell cycle. In late S (chromosome replication) and G0 phases, tumor cells are more radiotherapy-resistant, whereas in G2/M phase (separation of replicated chromosome) cells are more sensitive [34]. Indeed, radiation cell sensitivity changes during the cell cycle. Thus, OER increases with the aging of cells. In G1 phase, cells are more radiosensitive and they thus have a lower OER than in the S phase. At lower oxygen levels, HIF-1α can prevent cells from entering S phase by upregulating genes coding proteins involved in cell cycle regulation. Thus, Waf1 (p21) and Kip1 (p27), which are 2 major cyclin-dependent kinase inhibitors, are upregulated under hypoxic conditions and cause cell growth arrest [35,36]. Several other factors are activated under low oxygen tension, including transcription factors such as NF-κb (nuclear factor-kappa B), oncogenes, growth factors, and free radicals [37]. Hypoxia can induce apoptosis through a number of pathways (HIF-dependent or not), but tumor cells survive under hypoxic conditions. Induction of anti-apoptotic genes such as IAP-2 or the downregulation of pro-apoptotic proteins of the Bcl2 family are part of these mechanisms (Bid, Bax) [38,39].
In addition, some studies showed that hypoxia in cancer was associated with shorter disease-free and overall survival. Understanding the mechanisms of radioresistance is a major step to develop strategies to finally overcome this problem [15].
Overcoming Radioresistance: A Never-Ending Issue?
Hypoxia is currently used as a potential biomarker for radiation oncology in addition to tumor size and positivity for human papilloma virus in head and neck squamous cell carcinoma [40]. Thus, hypoxia must be considered as an ultimate target to enhance therapeutic effects of radiation [41]. Many clinical studies tried to solve this problem by developing physical or chemical approaches.
Radiotherapy is based on the “5 Rs” of radiobiology: repair of sublethal damage, reassortment of cells within the cell cycle, repopulation, intrinsic radioresistance, and reoxygenation. This must be taken into account to develop efficient strategies [32,42]. Thus, many studies focused on testing altered fractionation and/or increased doses to overcome the radioresistance due to hypoxia. Increasing radiation doses are necessary to kill more hypoxic cells. However, normal tissues have a limited tolerance to radiation [43]. Use of focal dose escalation on hypoxic areas was developed to improve local control. Theoretically, it can improve radiation efficacy. However, the literature is conflicting and limited on this particular topic [44,45]. In cervical cancer patients, hypoxia can be imaged by positron emission tomography (PET) [46]. Dose escalation was performed on hypoxic areas, resulting in increased survival [45]. However, some studies showed that increasing radiation doses in hypoxic areas had a very limited impact on acute hypoxic areas, but could partly improve the local control rate of chronic hypoxic areas [44]. The ESCALOX protocol concluded that dose escalation to large parts of the tumor was associated with the risk of more acute and late toxicity [47]. In prostate cancer, the estimated dose escalation to overcome tumor hypoxia was 165 Gy [48]. A recent study reported that hypoxia biomarkers can predict outcome and benefit of dose escalation in prostate cancer patients receiving external beam radiotherapy (EBRT) alone or combined with high-dose-rate brachytherapy boost (HDR-BTb) [49].
Stereotaxic body radiation therapy (SBRT) differs from other EBRTs as it involves the delivery of higher radiation doses to the cancer site. Unconventional SBRT was developed to target hypoxic segments in hypoxic areas [50]. The first data from SBRT-PATHY (PArtial Tumor HYpoxic segment) showed very encouraging results for very large unresectable tumors, but prospective studies are necessary to confirm this [51]. It was also demonstrated that SBRT could induce tumor hypoxia after initial treatment [52].
Accelerated radiotherapy (1 fraction every 12 h instead of every 24 h) was tested in head and neck cancer patients, inducing tumor oxygenation in 70–80% of patients and thus causing tumor radiosensitivity [53]. Other altered fractionation schemes were unsuccessfully tested. The large randomized British CHART (Continuous Hyper-fractionated Accelerated Radiotherapy) study was carried out in 918 head and neck cancer patients. It used a total dose of 54 Gy, with 1.5Gy x 3 per day over a 12-day period. This protocol failed to prove its efficacy versus a normo-fractionated standard radiotherapy, possibly because reoxygenation was insufficient to radiosensitize cancer cells [54]. Thus, post-radiotherapy biological effects such as cell cycle redistribution, blood reperfusion, and reoxygenation are complex to model and trigger.
Regarding synergic action, many studies evaluated radiotherapy-combined treatments, also called radiation-sensitizers. Historically, nitroimidazole was first studied in the 1950s and several generations of drugs generations have emerged since. The radiation-sensitizer behaves like water when irradiated. This generates free radicals that damage DNA and lead to tumor cell death. First- and second-generation drugs turned very toxic, whereas third-generation drugs showed positive but inconsistent results in head and neck and cervical tumors [57–59]. Although such drugs showed efficacy, the effect was not significant enough to stimulate a broader interest in this area. To limit toxicities, bioreductive drugs were developed to target hypoxic tumor cells only [59]. The results of tirapazamine in pre-clinical studies were encouraging, but when combined with radiotherapy it was less efficient than expected. Moreover, no bioreductive drug was approved for use in clinical practice [56–59].
In view of the predominant role of major pathways in hypoxia such as HIF, targeting therapies emerged to overcome hypoxic resistance. Recent reviews focused on the action of HIF-2 inhibitors in glioblastoma, neuroblastoma, and clear cell renal carcinoma [60]. Indeed, a study tested HIF-2 Alpha Inhibitor PT2385 in patients with recurrent glioblastoma [61]. Only a few studies attempted to understand the biological mechanisms by which HIF-2α regulates radiation response in cancer [62]. HIF-2α inhibition enhanced radiation sensitivity in a cellular model of lung cancer by promoting apoptotic activity via the p53 pathway [63]. Likewise, targeting HIF-1 and tumor glucose metabolism sensitized various solid tumors to irradiation [64].
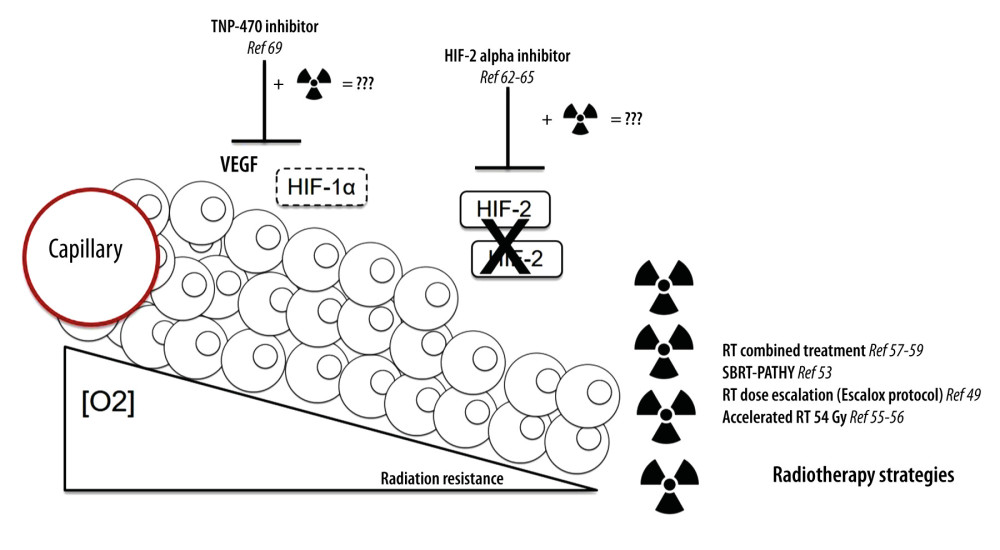
In an in vitro lung cancer model, inhibition of the HIF-1α/VEGF pathway reversed the effect of hypoxia on cell viability, invasion, and migration, thus inhibiting resistance to radiotherapy [65]. Radiotherapy and anti-VEGF interaction are not yet well validated [66]. In vitro, TNP-470, an angiogenesis inhibitor, could increase tumor oxygenation and radiosensitivity [67]. ROS are considered the main cause of radiation-induced cell death. Therefore, disruption of ROS homeostasis can overcome hypoxic radioresistance (Figure 1) [68].
In the tumor microenvironment, hypoxia influences the interaction between cancers and the immune system on every level: downregulation of major histocompatibility complex (MHC) molecules, upregulation of immune checkpoint inhibitors such as HLA-G (Human leukocyte antigen-G), CTLA-4 (cytotoxic T-lymphocyte antigen 4), and PD-L1 (programmed death ligand-1) [69,70]. The rational to combine radiotherapy and immunotherapy is based on increased T cell infiltration in irradiated tumors and mitigation of radiation-induced immunosuppressive mechanisms such as PD-L1 upregulation by immune checkpoint inhibition. Thus, hypoxic cells might be a specific target of immune checkpoint inhibitors combined with radiotherapy. Such inhibitors improve local control and induce abscopal effects, which leads to better systemic tumor control [71]. The tumor microenvironment and stromal reaction should be considered, as they clearly play a critical role in resistance and may offer novel therapeutic perspectives [72].
More recently, gene therapy strategies targeting hypoxic cells were developed. Studies evaluated the double gene knockout of GLUT-1 and HIF-1α, which are 2 major genes involved in hypoxic resistance. The technique used was the developed genome editing system CRISPR (clustered regularly interspaced short palindromic repeat)/Cas (CRISPR-associated proteins), which permitted multigene knockout. In modified cells, the proliferation, migration, and invasion significantly decreased [72]. Research is warranted on adding radiotherapy to this model.
Conclusions
This review has shown that hypoxia is a major characteristic of the microenvironment of solid tumors. Tumor tissue hypoxia affects the response to radiation therapy, chemotherapy, and immunotherapy. The mechanism of hypoxia is via the selection of more aggressive tumor cell clones that affect patient prognosis. Currently, multiple therapeutic strategies have failed to overcome hypoxia-related radioresistance. Therefore, the pathophysiological mechanisms and molecular pathways involved need to be investigated.
References
1. Balkwill FR, Capasso M, Hagemann T, The tumor microenvironment at a glance: J Cell Sci, 2012; 125; 5591-96
2. Vaupel P, Mayer A, Höckel M, Tumor hypoxia and malignant progression: Meth Enzymol, 2004; 381; 335-54
3. Muz B, de la Puente P, Azab F, Azab AK, The role of hypoxia in cancer progression, angiogenesis, metastasis, and resistance to therapy: Hypoxia (Auckl), 2015; 3; 83-92
4. Carmeliet P, Jain RK, Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases: Nat Rev Drug Discov, 2011; 10; 417-27
5. Brizel DM, Dodge RK, Clough RW, Dewhirst MW, Oxygenation of head and neck cancer: Changes during radiotherapy and impact on treatment outcome: Radiother Oncol, 1999; 53; 113-17
6. Gray LH, Conger AD, Ebert M, The concentration of oxygen dissolved in tissues at the time of irradiation as a factor in radiotherapy: Br J Radiol, 1953; 26; 638-48
7. Rockwell S, Dobrucki IT, Kim EY, Hypoxia and radiation therapy: Past history, ongoing research, and future promise: Curr Mol Med, 2009; 9; 442-58
8. Emami Nejad A, Najafgholian S, Rostami A, The role of hypoxia in the tumor microenvironment and development of cancer stem cell: A novel approach to developing treatment: Cancer Cell Int, 2021; 21; 62
9. Begg K, Tavassoli M, Inside the hypoxic tumour: Reprogramming of the DDR and radioresistance: Cell Death Discov, 2020; 6; 77
10. Sørensen BS, Horsman MR, Tumor hypoxia: Impact on radiation therapy and molecular pathways: Front Oncol, 2020; 10; 562
11. Vaupel P, Hypoxia and aggressive tumor phenotype: Implications for therapy and prognosis: Oncologist, 2008; 13(Suppl 3); 21-26
12. Dewhirst MW, A potential solution for eliminating hypoxia as a cause for radioresistance: Proc Natl Acad Sci USA, 2018; 115; 10548-50
13. Benej M, Hong X, Vibhute S, Papaverine and its derivatives radiosensitize solid tumors by inhibiting mitochondrial metabolism: Proc Natl Acad Sci USA, 2018; 115; 10756-61
14. Mottram J, Experiments on the radioation of tumours: Br Med J, 1927; 1; 275-77
15. Vaupel P, Mayer A, Hypoxia in cancer: Significance and impact on clinical outcome: Cancer Metastasis Rev, 2007; 26; 225-39
16. Semenza GL, Hypoxia, clonal selection, and the role of HIF-1 in tumor progression: Crit Rev Biochem Mol Biol, 2000; 35; 71-103
17. Bayer C, Vaupel P, Acute versus chronic hypoxia in tumors: Controversial data concerning time frames and biological consequences: Strahlenther Onkol, 2012; 188; 616-27
18. Bayer C, Shi K, Astner ST, Acute versus chronic hypoxia: Why a simplified classification is simply not enough: Int J Radiat Oncol Biol Phys, 2011; 80; 965-68
19. Vaupel P, Kallinowski F, Okunieff P, Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: A review: Cancer Res, 1989; 49; 6449-65
20. Dewhirst MW, Concepts of oxygen transport at the microcirculatory level: Semin Radiat Oncol, 1998; 8; 143-50
21. Cairns RA, Kalliomaki T, Hill RP, Acute (cyclic) hypoxia enhances spontaneous metastasis of KHT murine tumors: Cancer Res, 2001; 61; 8903-8
22. Al Tameemi W, Dale TP, Al-Jumaily RMK, Forsyth NR, Hypoxia-modified cancer cell metabolism: Front Cell Dev Biol, 2019; 7; 4
23. Vaupel P, Harrison L, Tumor hypoxia: Causative factors, compensatory mechanisms, and cellular response: Oncologist, 2004; 9(Suppl 5); 4-9
24. Vaupel P, The role of hypoxia-induced factors in tumor progression: Oncologist, 2004; 9(Suppl 5); 10-17
25. Wang GL, Jiang B-H, Rue EA, Semenza GL, Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular 02 tension: Proc Natl Acad Sci USA, 1995; 92; 5510-14
26. Semenza GL, Wang GL, A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation: Mol Cell Biol, 1992; 12; 5447-54
27. Aebersold DM, Burri P, Beer KT, Expression of hypoxia-inducible factor-1alpha: A novel predictive and prognostic parameter in the radiotherapy of oropharyngeal cancer: Cancer Res, 2001; 61; 2911-16
28. Choschzick M, Oosterwijk E, Müller V, Overexpression of carbonic anhydrase IX (CAIX) is an independent unfavorable prognostic marker in endometrioid ovarian cancer: Virchows Arch, 2011; 459; 193-200
29. Lock FE, McDonald PC, Lou Y, Targeting carbonic anhydrase IX depletes breast cancer stem cells within the hypoxic niche: Oncogene, 2013; 32; 5210-19
30. Crabtree HG, Cramer W, Murray JA, The action of radium on cancer cells. II. – Some factors determining the susceptibility of cancer cells to radium. Proceedings of the Royal Society of London Series B, Containing Papers of a Biological Character: Royal Society, 1933; 113; 238-50
31. Brown JM, Wilson WR, Exploiting tumour hypoxia in cancer treatment: Nat Rev Cancer, 2004; 4; 437-47
32. Hall EJ, Giaccia AJ: Radiobiology for the radiologist, 2012, Philadelphia, Wolters Kluwer Health/Lippincott Williams & Wilkins
33. Pouget J-P, Frelon S, Ravanat J-L, Formation of modified DNA bases in cells exposed either to gamma radiation or to high-LET particles: Radiat Res, 2002; 157; 589-95
34. Pawlik TM, Keyomarsi K, Role of cell cycle in mediating sensitivity to radiotherapy: Int J Radiat Oncol Biol Phys, 2004; 59; 928-42
35. Green SL, Freiberg RA, Giaccia AJ, p21Cip1 and p27Kip1 regulate cell cycle reentry after hypoxic stress but are not necessary for hypoxia-induced arrest: Mol Cell Biol, 2001; 21; 1196-206
36. Krtolica A, Krucher NA, Ludlow JW, Hypoxia-induced pRB hypophosphorylation results from downregulation of CDK and upregulation of PP1 activities: Oncogene, 1998; 17; 2295-304
37. Chandel NS, McClintock DS, Feliciano CE, Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: A mechanism of O2 sensing: J Biol Chem, 2000; 275; 25130-38
38. Greijer AE, van der Wall E, The role of hypoxia inducible factor 1 (HIF-1) in hypoxia induced apoptosis: J Clin Pathol, 2004; 57; 1009-14
39. Erler JT, Cawthorne CJ, Williams KJ, Hypoxia-mediated down-regulation of Bid and Bax in tumors occurs via hypoxia-inducible factor 1-dependent and -independent mechanisms and contributes to drug resistance: Mol Cell Biol, 2004; 24; 2875-89
40. Krause M, Dubrovska A, Linge A, Baumann M, Cancer stem cells: Radioresistance, prediction of radiotherapy outcome and specific targets for combined treatments: Adv Drug Deliv Rev, 2017; 109; 63-73
41. Brown JM, Tumor hypoxia in cancer therapy: Meth Enzymol, 2007; 435; 297-321
42. Steel GG, McMillan TJ, Peacock JH, The 5Rs of radiobiology: Int J Radiat Biol, 1989; 56; 1045-48
43. Emami B, Lyman J, Brown A, Tolerance of normal tissue to therapeutic irradiation: Int J Radiat Oncol Biol Phys, 1991; 21; 109-22
44. Popple RA, Ove R, Shen S, Tumor control probability for selective boosting of hypoxic subvolumes, including the effect of reoxygenation: Int J Radiat Oncol Biol Phys, 2002; 54; 921-27
45. Chao KS, Bosch WR, Mutic S, A novel approach to overcome hypoxic tumor resistance: Cu-ATSM-guided intensity-modulated radiation therapy: Int J Radiat Oncol Biol Phys, 2001; 49; 1171-82
46. Han K, Shek T, Vines D, Measurement of tumor hypoxia in patients with locally advanced cervical cancer using positron emission tomography with 18F-Fluoroazomyin arabinoside: Int J Radiat Oncol Biol Phys, 2018; 102; 1202-9
47. Pigorsch SU, Wilkens JJ, Kampfer S, Do selective radiation dose escalation and tumour hypoxia status impact the loco-regional tumour control after radio-chemotherapy of head & neck tumours? The ESCALOX protocol: Radiat Oncol, 2017; 12; 45
48. Wang JZ, Li XA, Mayr NA, Dose escalation to combat hypoxia in prostate cancer: A radiobiological study on clinical data: Br J Radiol, 2006; 79; 905-11
49. Bhattacharya IS, Taghavi Azar Sharabiani M, Alonzi R, Hoskin PJ, Hypoxia and angiogenic biomarkers in prostate cancer after external beam radiotherapy (EBRT) alone or combined with high-dose-rate brachytherapy boost (HDR-BTb): Radiother Oncol, 2019; 137; 38-44
50. Tubin S, Popper HH, Brcic L, Novel stereotactic body radiation therapy (SBRT)-based partial tumor irradiation targeting hypoxic segment of bulky tumors (SBRT-PATHY): Improvement of the radiotherapy outcome by exploiting the bystander and abscopal effects: Radiat Oncol, 2019; 14; 21
51. Tubin S, Popper HH, Brcic L, Novel stereotactic body radiation therapy (SBRT)-based partial tumor irradiation targeting hypoxic segment of bulky tumors (SBRT-PATHY): Improvement of the radiotherapy outcome by exploiting the bystander and abscopal effects: Radiat Oncol, 2019; 14; 21
52. Qian Y, Von Eyben R, 18F-EF5 PET-based imageable hypoxia predicts local recurrence in tumors treated with highly conformal radiation therapy: Int J Radiat Oncol Biol Phys, 2018; 102; 1183-92
53. Lartigau E, Lusinchi A, Weeger P, Variations in tumour oxygen tension (pO2) during accelerated radiotherapy of head and neck carcinoma: Eur J Cancer, 1998; 34; 856-61
54. Koukourakis MI, Bentzen SM, Giatromanolaki A, Endogenous markers of two separate hypoxia response pathways (hypoxia inducible factor 2 alpha and carbonic anhydrase 9) are associated with radiotherapy failure in head and neck cancer patients recruited in the CHART randomized trial: J Clin Oncol, 2006; 24; 727-35
55. Overgaard J, Hansen HS, Overgaard M, A randomized double-blind phase III study of nimorazole as a hypoxic radiosensitizer of primary radiotherapy in supraglottic larynx and pharynx carcinoma. Results of the Danish Head and Neck Cancer Study (DAHANCA) Protocol 5–85: Radiother Oncol, 1998; 46; 135-46
56. Bentzen J, Toustrup K, Eriksen JG, Locally advanced head and neck cancer treated with accelerated radiotherapy, the hypoxic modifier nimorazole and weekly cisplatin. Results from the DAHANCA 18 phase II study: Acta Oncologica, 2015; 54; 1001-7
57. Overgaard J, Hypoxic modification of radiotherapy in squamous cell carcinoma of the head and neck – a systematic review and meta-analysis: Radiother Oncol, 2011; 100; 22-32
58. Zeng Y-C, Yu L, Xiao Y-P, Radiation enhancing effects with the combination of sanazole and irinotecan in hypoxic HeLa human cervical cancer cell line: J BUON, 2013; 18; 713-16
59. McKeown SR, Cowen RL, Williams KJ, Bioreductive drugs: From concept to clinic: Clin Oncol, 2007; 19; 427-42
60. Yu T, Tang B, Sun X, Development of inhibitors targeting hypoxia-inducible factor 1 and 2 for cancer therapy: Yonsei Med J, 2017; 58; 489-96
61. Renfrow JJ, Soike MH, Debinski W, Hypoxia-inducible factor 2α: A novel target in gliomas: Future Med Chem, 2018; 10; 2227-36
62. Moreno Roig E, Groot AJ, Yaromina A, HIF-1α and HIF-2α differently regulate the radiation sensitivity of NSCLC cells: Cells, 2019; 8; 45
63. Bertout JA, Majmundar AJ, Gordan JD, HIF2alpha inhibition promotes p53 pathway activity, tumor cell death, and radiation responses: Proc Natl Acad Sci USA, 2009; 106; 14391-96
64. Meijer TWH, Kaanders JHAM, Span PN, Bussink J, Targeting hypoxia, HIF-1, and tumor glucose metabolism to improve radiotherapy efficacy: Clin Cancer Res, 2012; 18; 5585-94
65. Zhu H, Zhang S, Hypoxia inducible factor-1α/vascular endothelial growth factor signaling activation correlates with response to radiotherapy and its inhibition reduces hypoxia-induced angiogenesis in lung cancer: J Cell Biochem, 2018; 119; 7707-18
66. Kanthou C, Tozer G, Targeting the vasculature of tumours: Combining VEGF pathway inhibitors with radiotherapy: Br J Radiol, 2019; 92; 20180405
67. Teicher BA, Dupuis N, Kusomoto T, Antiangiogenic agents can increase tumor oxygenation and response to radiation therapy: Radiation Oncology Investigations, 1994; 2; 269-76
68. Wang H, Jiang H, Van De Gucht M, De Ridder M, Hypoxic radioresistance: Can ROS be the key to overcome it?: Cancers (Basel), 2019; 11; 112
69. Sethumadhavan S, Silva M, Philbrook P, Hypoxia and hypoxia-inducible factor (HIF) downregulate antigen-presenting MHC class I molecules limiting tumor cell recognition by T cells: PLoS One, 2017; 12; e0187314
70. Yaghi L, Poras I, Simoes RT, Hypoxia inducible factor-1 mediates the expression of the immune checkpoint HLA-G in glioma cells through hypoxia response element located in exon 2: Oncotarget, 2016; 7; 63690-707
71. Wigerup C, Påhlman S, Bexell D, Therapeutic targeting of hypoxia and hypoxia-inducible factors in cancer: Pharmacol Ther, 2016; 164; 152-69
72. Barker HE, Paget JTE, Khan AA, Harrington KJ, The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence: Nat Rev Cancer, 2015; 15; 409-25
In Press
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952