02 January 2022: Clinical Research
Retrospective Study of 573 Patients with Heart Failure Evaluated for Coronary Artery Disease at Toulouse University Center, France
Antoine Deney1ABCDEF, Vanessa Nader12ABCDEF, Anthony Matta13ABCDE, Romain Itier1CDF, Pauline Fournier1ABCD, Olivier LairezDOI: 10.12659/MSM.934804
Med Sci Monit 2022; 28:e934804
Abstract
BACKGROUND: Heart failure (HF) most commonly occurs due to ischemic heart disease from stenotic coronary artery disease (CAD). HF is classified into 3 groups based on the percentage of the ejection fraction (EF): reduced (HFrEF), mid-range (HFmrEF), and preserved (HFpEF). This retrospective study included 573 patients who presented with HF based on the evaluation of EF and were evaluated for CAD by coronary angiography before undergoing coronary angioplasty at a single center in Toulouse, France.
MATERIAL AND METHODS: This retrospective observational study included patients recently diagnosed with HF or acute decompensation of chronic HF and referred for coronary angiography at Toulouse University Hospital between January 2019 and May 2020.
RESULTS: Significant CAD was found in 55.8%, 55%, and 55% of the whole population, HFpEF, and HFrEF groups, respectively. Older age, male sex, and diabetes mellitus were the main risk factors for ischemic HF. Except for age and sex, patients with ischemic HFpEF were comparable to those with non-ischemic HFpEF, unlike the ischemic HFrEF group, which had more common cardiovascular risk factors than the non-ischemic HFrEF group. The ischemic HFpEF group had an older age and higher rate of dyslipidemia than the ischemic HFrEF group.
CONCLUSIONS: At our center, CAD was diagnosed in more than half of patients who presented with heart failure with preserved or reduced EF. Older age and male sex were the common risk factors in patients with HFpEF and HFrEF.
Keywords: Coronary Artery Disease, Heart Failure, Diastolic, Heart Failure, Systolic, Age Factors, Angioplasty, Balloon, Coronary, Coronary Angiography, Female, France, Heart Disease Risk Factors, Humans, Male, Risk Factors, Sex Factors, Stroke Volume
Background
Heart failure (HF) is a major medical concern accounting for a huge number of hospitalizations, emergency department visits, and cardiovascular deaths [1,2]. The growing prevalence of HF is the consequence of increased life expectancy, hypertension, obesity, aging of the population, prolonged HF survival, and advancement in diagnostic and therapeutic strategies [3,4]. Neuro-humoral activation is the cornerstone of the complex pathophysiology of HF syndrome. HF is currently classified into 3 categories based on the percentage of left ventricular ejection fraction (LVEF): reduced (HFrEF, LVEF ≤40%), mid-range (HFmrEF, 40%< LVEF <50%), and preserved (HFpEF, LVEF ≥50%) [5]. Unlike HFpEF, for which medical therapy is limited to diuretics, the treatment of HFrEF has been extensively investigated in research studies and clinical trials. Treatment includes different drug classes (β-blockers, angiotensin converting enzyme inhibitor/angiotensin II receptor blocker, angiotensin receptor-neprilysin inhibitor, mineralocorticoid receptor antagonist, ivabradine, SGLT2 inhibitors, and vericiguat), devices (implantable cardiac defibrillator), stem cell transplantation, and gene therapy [6,7].
While coronary artery disease (CAD) has been long recognized as a major cause and therapeutic target of HF, the beneficial role of percutaneous intervention for coronary revascularization in patients with HF remains controversial [8]. Indeed, the presence of hibernating myocardium is crucial to predict improvement in cardiac function after coronary revascularization [9]. The role of CAD is not limited to HFrEF but is also implicated in HFpEF [10], and a prevalence of 80% of significant CAD in patients with HFpEF was reported in a study using the coronary angiography approach [11]. Moreover, CAD and HFpEF share several common risk factors, such as age and hypertension [12]. It is worth mentioning that HFpEF is more common in women, and HFrEF is more common in men [12,13], which is explained by the fact that much HF in men is provoked by myocardial infarction [12,13]. Guidelines recommend the careful search of CAD in patients with HF [5], but in general, the prevalence of CAD is underestimated in patients with HF because noninvasive tests are commonly used for screening. Therefore, this retrospective study conducted at a single center in Toulouse, France, included 573 patients who presented with HF based on the evaluation of reduced EF and were evaluated for CAD by coronary angiography before undergoing coronary angioplasty.
Material and Methods
STUDY DESIGN AND POPULATION:
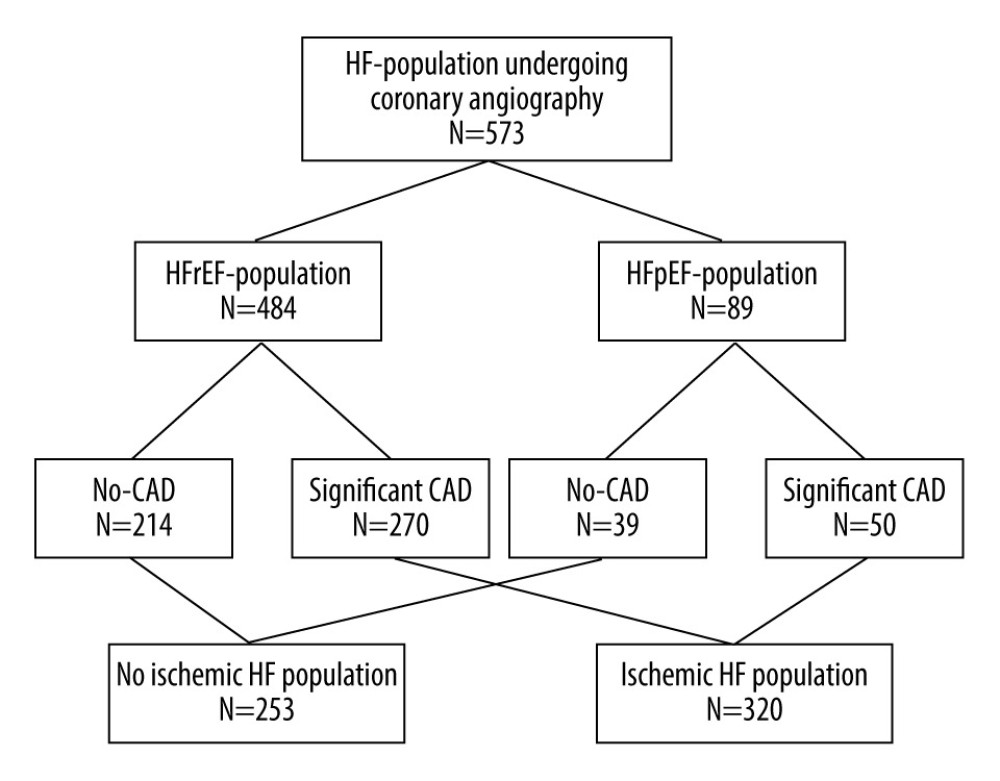
This observational retrospective study included 573 patients with HF who presented for coronary angiography at the Interventional Cardiology Department at the University Hospital of Toulouse, France, between January 2019 and May 2020. The indications of coronary angiography in the included patients were a recent diagnosis of HF or acute decompensation of chronic HF. Then, patients were divided into 2 groups, HFpEF and HFrEF, which were subsequently divided into 2 subgroups of those with ischemic HF (IHF) and those without IHF, according to the presence or absence of significant CAD (Figure 1). All patients were informed at admission that their clinical data could be used for research purposes and gave their informed consent. The cohort was registered by the Ministry of Research and the Regional Health Agency Occitanie (no. DC-2017-298).
DATA COLLECTION AND ENDPOINTS:
The baseline and demographic characteristics of the study participants, including age, sex, cardiovascular risk factors, medical treatment, prior medical history, and concomitant comorbidities, were collected by the study investigators. Also, transthoracic echocardiography parameters (left ventricular EF and valvulopathies), biological markers (troponin and NT-proBNP), and coronary angiography results were collected. All of these data were collected from the Orbis and Hemolia database used in our center. Significant CAD was defined as a ≥50% reduction in luminal coronary diameter. The purpose of this retrospective study was to assess the prevalence of significant CAD in the 573 patients who presented with HF and underwent coronary angiography at our center.
STATISTICAL ANALYSES:
Numbers and percentages were used to describe categorical variables, while means±standard deviations were used to describe continuous variables. Continuous variables were analyzed with the
Results
The mean age of the 573 study participants was 68.3±12 years. HFrEF was significantly more prevalent than HFpEF (84.5% vs 15.5%). Patients with HFpEF were older and had more cardiovascular risk factors than the other groups; there was no difference in smoking. Significant CAD was detected in 55.8% of patients, and 76.4% of patients were men. Tables 1 and 2 show the characteristics of the study population.
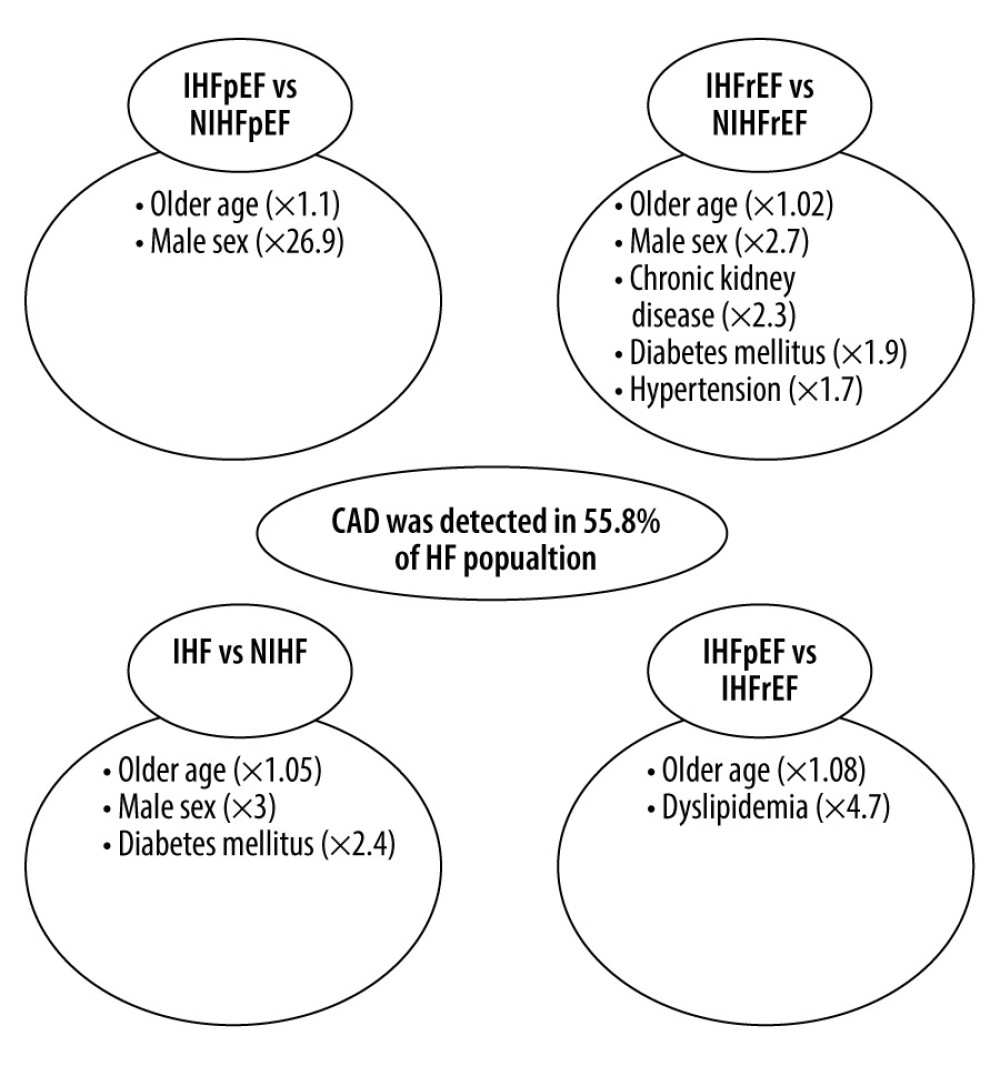
Compared with the non-IHFpEF subgroup (n=39), patients with IHFpEF (n=50) were older (74 vs 70.5 years), predominantly men (84% vs 53.8%), and had a lower mean baseline heart rate (74 vs 83 beats/min), New York Heart Association class, prevalence of associated atrial fibrillation (22% vs 35.9%), and right ventricular dysfunction (6% vs 7.7%). Except for the anti-thrombotic regimen, there were no differences in the received medical treatments (diuretics, β-blockers, mineralocorticoid, and ACEi/ARAII) (Table 3). Positive correlations between age (odds ratio [OR] 1.1, 95%CI 1.01–1.2,
Compared with the non-IHFrEF subgroup (n=214), the IHFrEF subgroup (n=270) had significantly higher age (70.3 vs 63.8 years), male sex (84.1% vs 69.2%) cardiovascular risk factors (chronic kidney disease [59.4% vs 31.9%], dyslipidemia [32.5% vs 24.7%], arterial hypertension [65.1% vs 48.3%], and diabetes mellitus [37.8% vs 27%]), implantable cardiac devices (7.4% vs 4.2%), and prescribed medical treatment (diuretics, β-blockers, ACEi/ARAII, and anti-thrombotic regimen). By contrast, mean baseline heart rate (80 vs 86 beats/min), diastolic blood pressure (74 vs 77 mmHg), and body mass index (BMI) (26.1 vs 30.3 kg/m2) were lower in IHFrEF subgroup (Table 4). The adjusted multivariate logistic regression on the previously cited confounding variables showed that IHFrEF was positively correlated with age (OR 1.02, 95%CI 1–1.05,
Male sex was more common in IHF (n=320) than in non-IHF (n=253) groups. The following were also more common in the IHF than in the non-IHF groups: implantable cardiac devices (8.1% vs 4.7%) and cardiovascular risk factors, including chronic kidney disease (58.5% vs 34.3%), dyslipidemia (35.8% vs 27.2%), diabetes mellitus (38.2% vs 27.2%), and arterial hypertension (66.9% vs 52.1%). Also, diuretics, β-blockers, ACEi/ARAII, and anti-thrombotic agents were used more often in patients with IHF. However, the prevalence of associated RVD (47.3% vs 38.4%), mean baseline heart rate (85.8 vs 79.2 bpm/min), and BMI (29.9 vs 26.3 kg/cm2) were higher in the non-IHF than IHF groups (Table 5). Lastly, the adjusted multivariate logistic regression showed a positive correlation between age (OR 1.05, 95%CI 1.02–1.09,
Lastly, patients with IHFpEF were older (74 vs 70.4 years) with a lower mean baseline heart rate (74 vs 80 beats/min) and higher systolic blood pressure (139.7 vs 127.9 mmHg), BMI (27.4 vs 26.1 kg/cm2), and rate of cardiovascular risk factors, including dyslipidemia (50% vs 30%) and arterial hypertension (72% vs 60%) than those with IHFrEF, which had more smokers (28.5% vs 16%) and higher NT-proBNP levels (6135 vs 3724) (Table 6). Compared with IHFrEF, the multivariate analysis showed that age and dyslipidemia were associated to IHFpEF (Figure 2).
Discussion
This study showed that more than half of patients referred for coronary angiography for a recent diagnosis of HF or acute decompensation of chronic HF presented with significant CAD. Age, male sex, and diabetes mellitus were independent predictors of IHF. Aside from age and sex, there were no differences between the IHFpEF and non-IHFpEF groups, unlike patients with IHFrEF, who had more cardiovascular risk factors, such as arterial hypertension, diabetes mellitus, and chronic kidney disease, than those with patent coronary arteries. Regardless, age and dyslipidemia, which were baseline characteristics of patients with HFpEF, were comparable to patients with HFrEF with CAD.
The literature has reported a similar rate of CAD in patients with HF after a systematic angiography approach, especially in those presenting with HFpEF [14–16]. Despite the known implications of CAD in the pathophysiology and development of HF, the effect of coronary revascularization on lowering the associated mortality and morbidity remains controversial [8]. Indeed, the co-existence of CAD in patients with HF was linked with poor long-term prognosis. CAD is usually underestimated in patients with HF, particularly in patients with HFpEF, in whom the role of CAD is under-recognized [17]. In agreement with our study result, Hwang et al showed that patients with HFpEF and those without CAD are comparable in medical treatment, laboratory markers, echocardiographic parameters, and baseline characteristics except for age and sex [10]. By contrast, cardiovascular risk factors in addition to older age and male sex were significantly more expressed in patients with IHFrEF compared with those with normal or near-normal coronary arteries. Regardless, this finding was included in a study conducted by Drissa et al [18]. It is well known that women are more predisposed to HFpEF, while men are more predisposed to HFrEF [12,19]. However, the present study showed that these sex differences vanished when comparing IHFpEF and IHFrEF. In view of the high prevalence of CAD in patients with HFpEF, absence of a difference in the distribution of cardiovascular risk factors, and poor outcomes attributed to the presence of CAD, searching for CAD in older men with HFpEF may improve prognosis and patient quality of life by preventing future ischemic heart events.
Compared with studies of HF in patients with non-obstructive CAD, the present study revealed that IHFpEF and IHFrEF share just 2 risk factors: older age and male sex. The relationship of sex and the development of ischemic cardiac diseases during the whole lifetime are well established in the literature. Furthermore, a recently published study investigated the role of sex in affecting the importance of risk factors for CAD [20]. Prospective trials based on the angiographic approach to screen and manage CAD in patients with HF and to evaluate the long-term impact on survival and quality of life are needed.
The main limitations of this study were the retrospective observational design and the lack of long-term follow-up data. Also, data concerning revascularization were not provided because we were interested in assessing the differences between various categories of HF according to the presence of significant CAD. The mean age of our study population was lower than that of the large registries of patients with HF, therefore explaining the ratio of HFrEF to HFpEF in these study participants from a tertiary referral hospital.
Conclusions
At our center, CAD was diagnosed in more than half of patients who presented with HF with preserved or reduced EF. Older age and male sex were the common risk factors in patients with HFpEF and HFrEF. Therefore, screening for CAD in patients recently diagnosed with HF or presenting with acute decompensation of HF is warranted. Future prospective studies investigating the impact of revascularization on long-term prognosis in patients with ischemic HFrEF and HFpEF compared with those without CAD are needed.
Tables
Table 1. Characteristics of the studied population stratified by type of heart failure.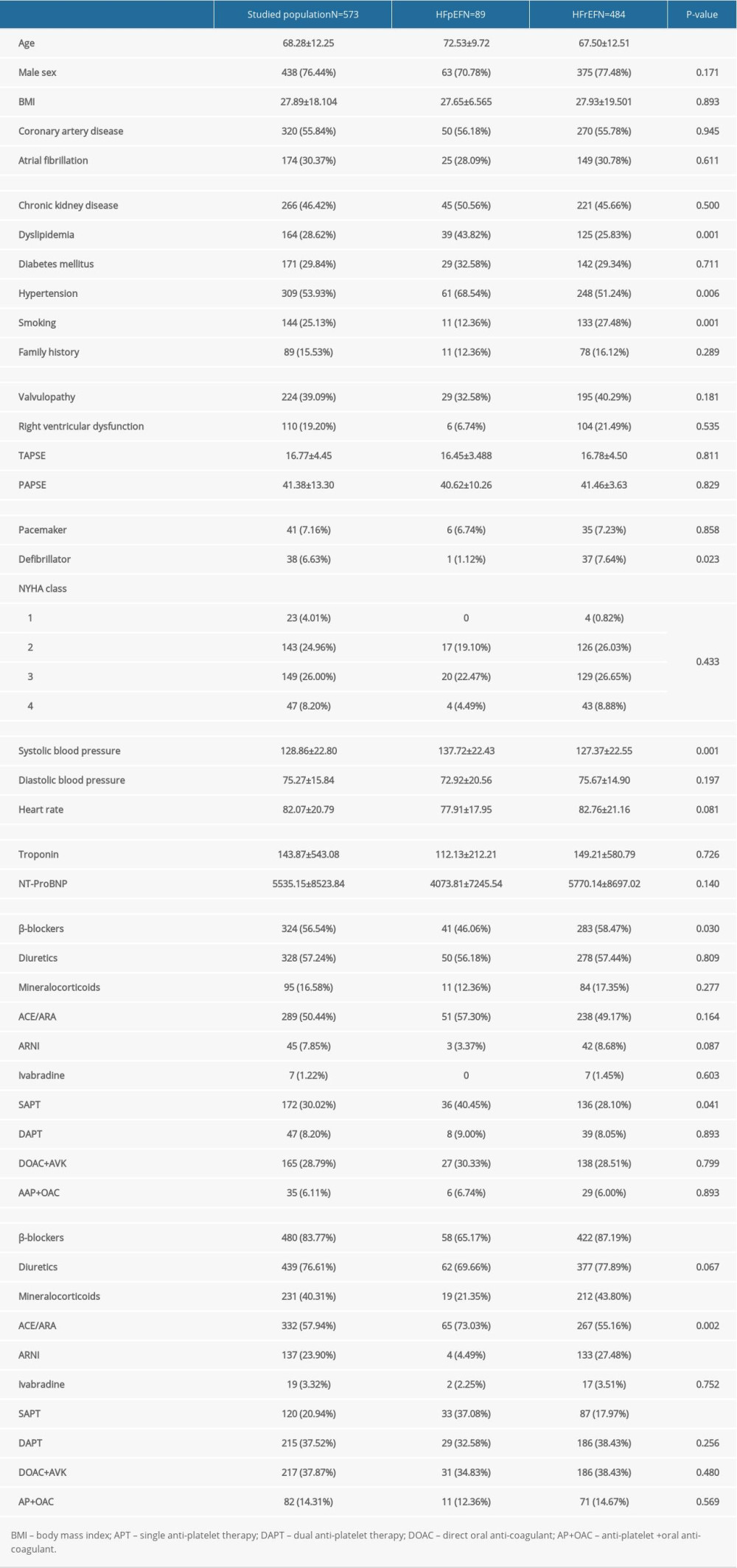 Table 2. The distribution of significant coronary artery disease.
Table 2. The distribution of significant coronary artery disease. Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease.
Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease.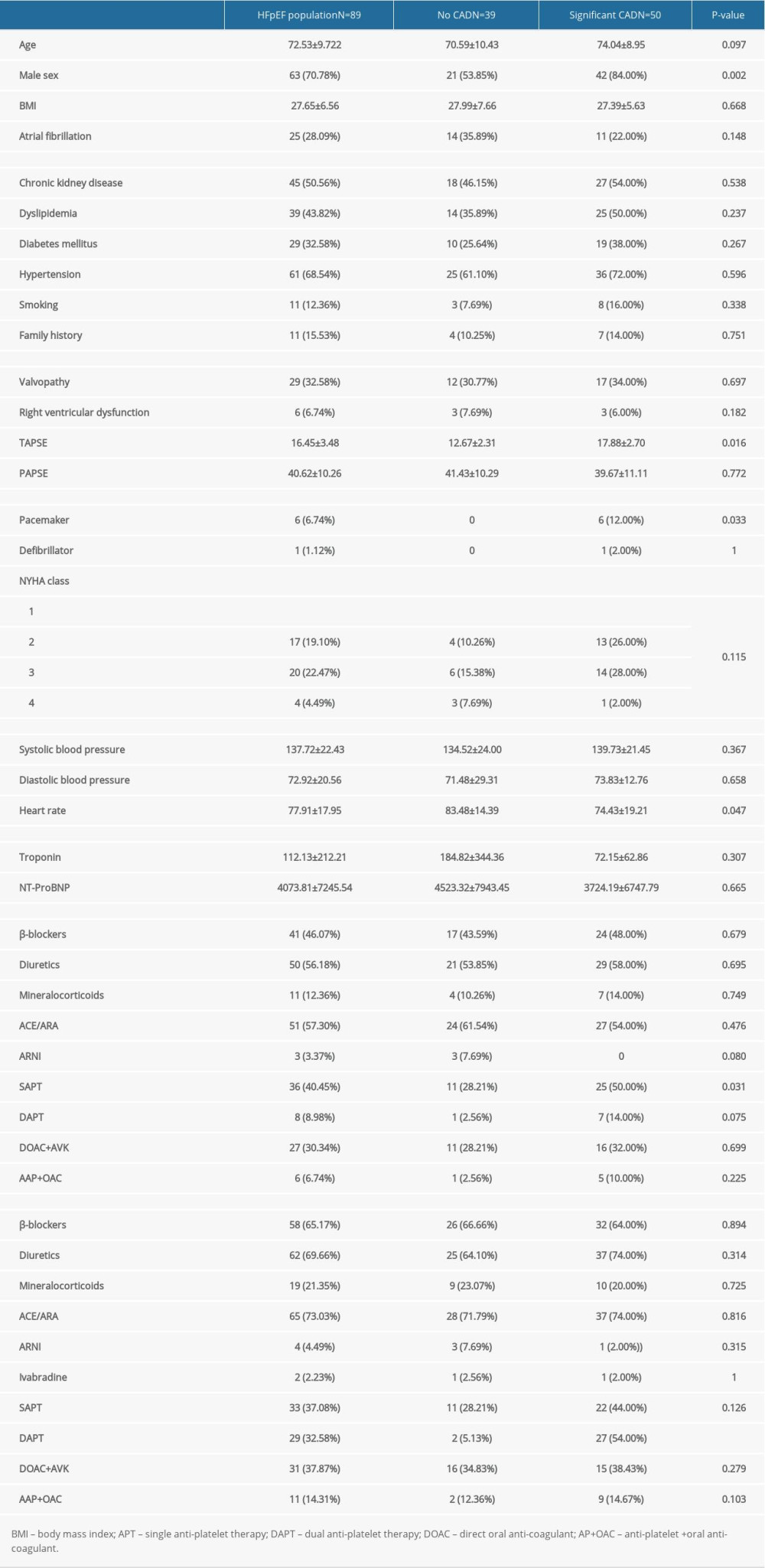 Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease.
Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease.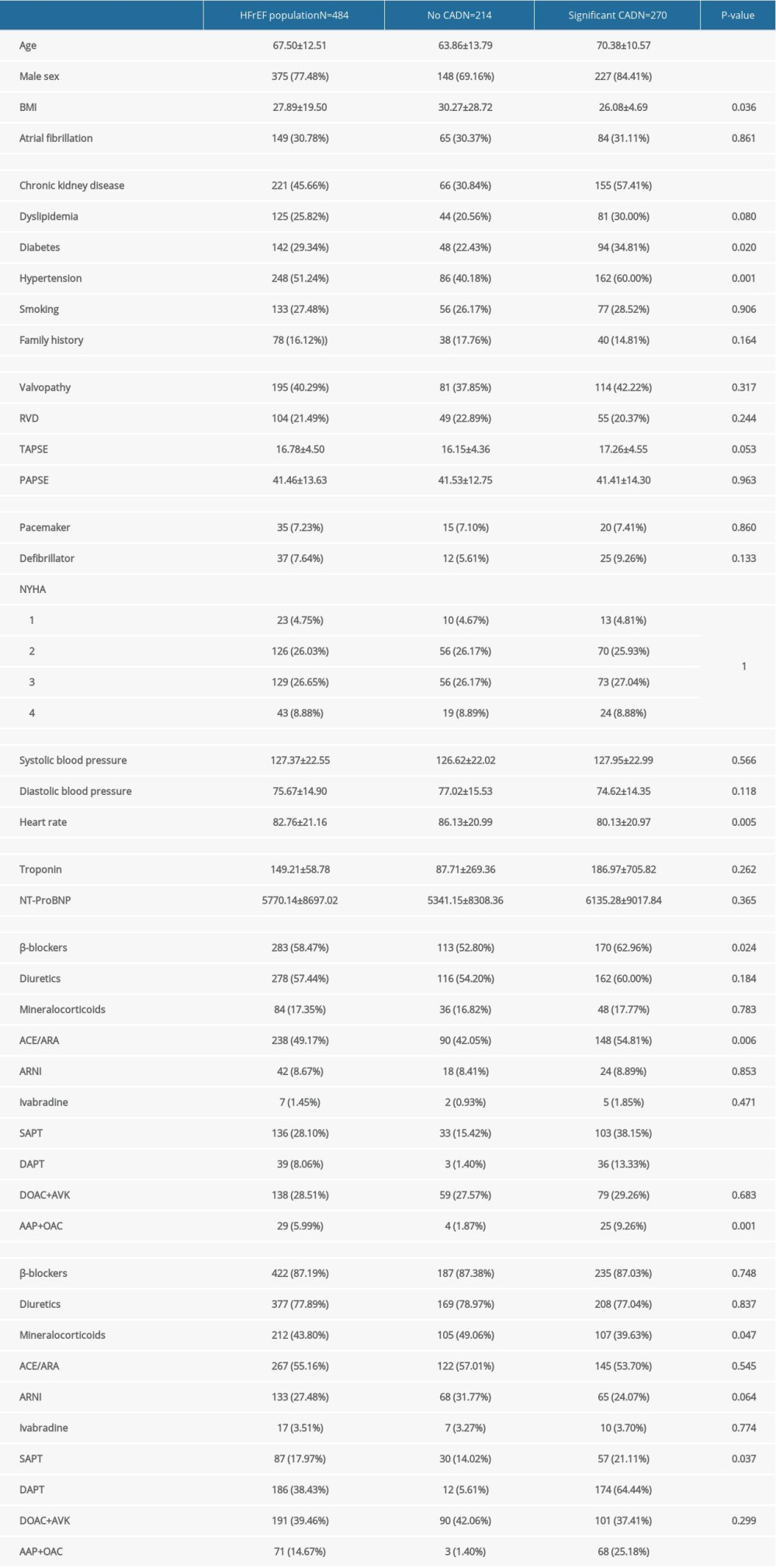 Table 5. Characteristics of the study population stratified by the presence of coronary artery disease.
Table 5. Characteristics of the study population stratified by the presence of coronary artery disease.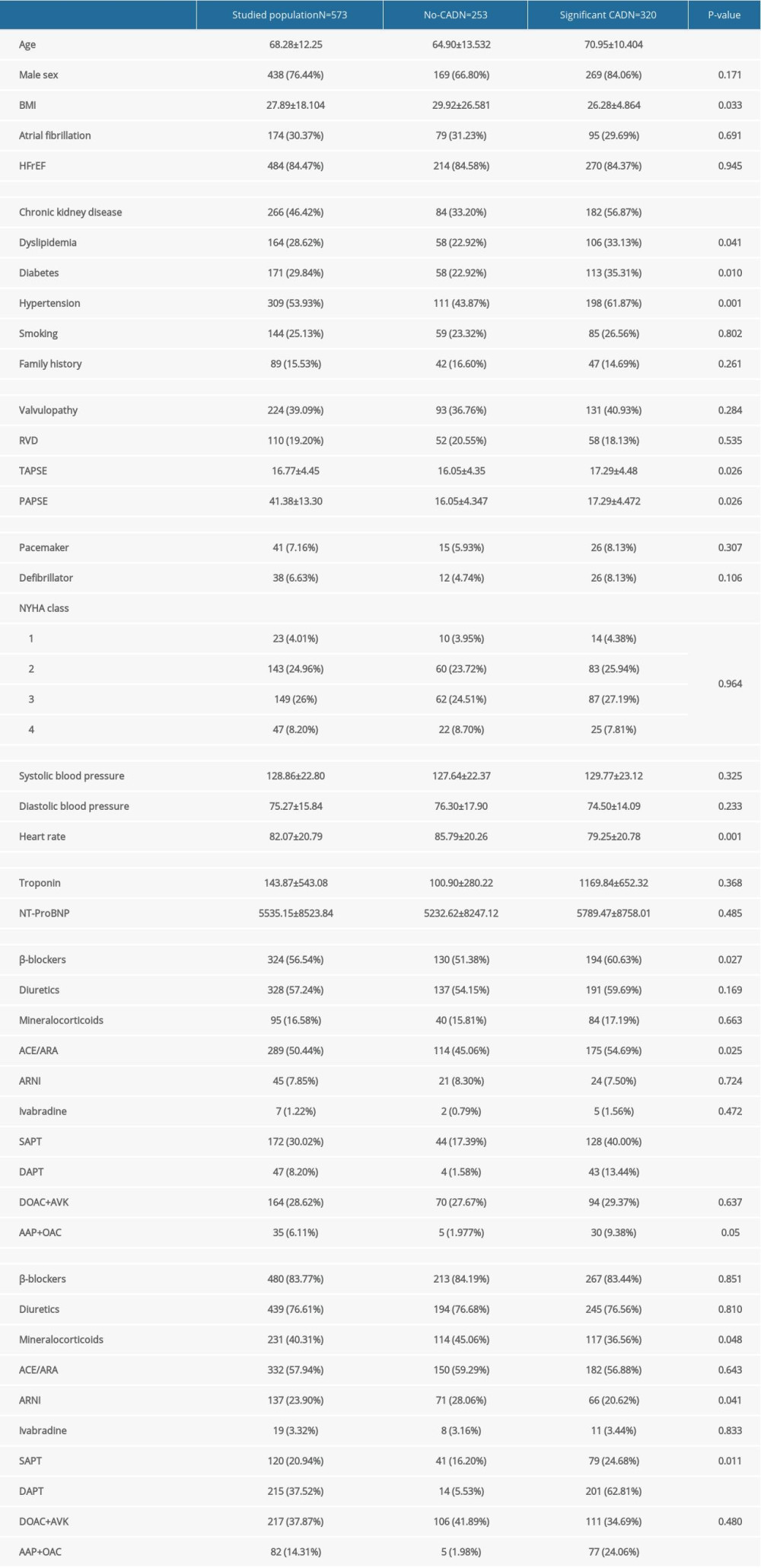 Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure.
Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure.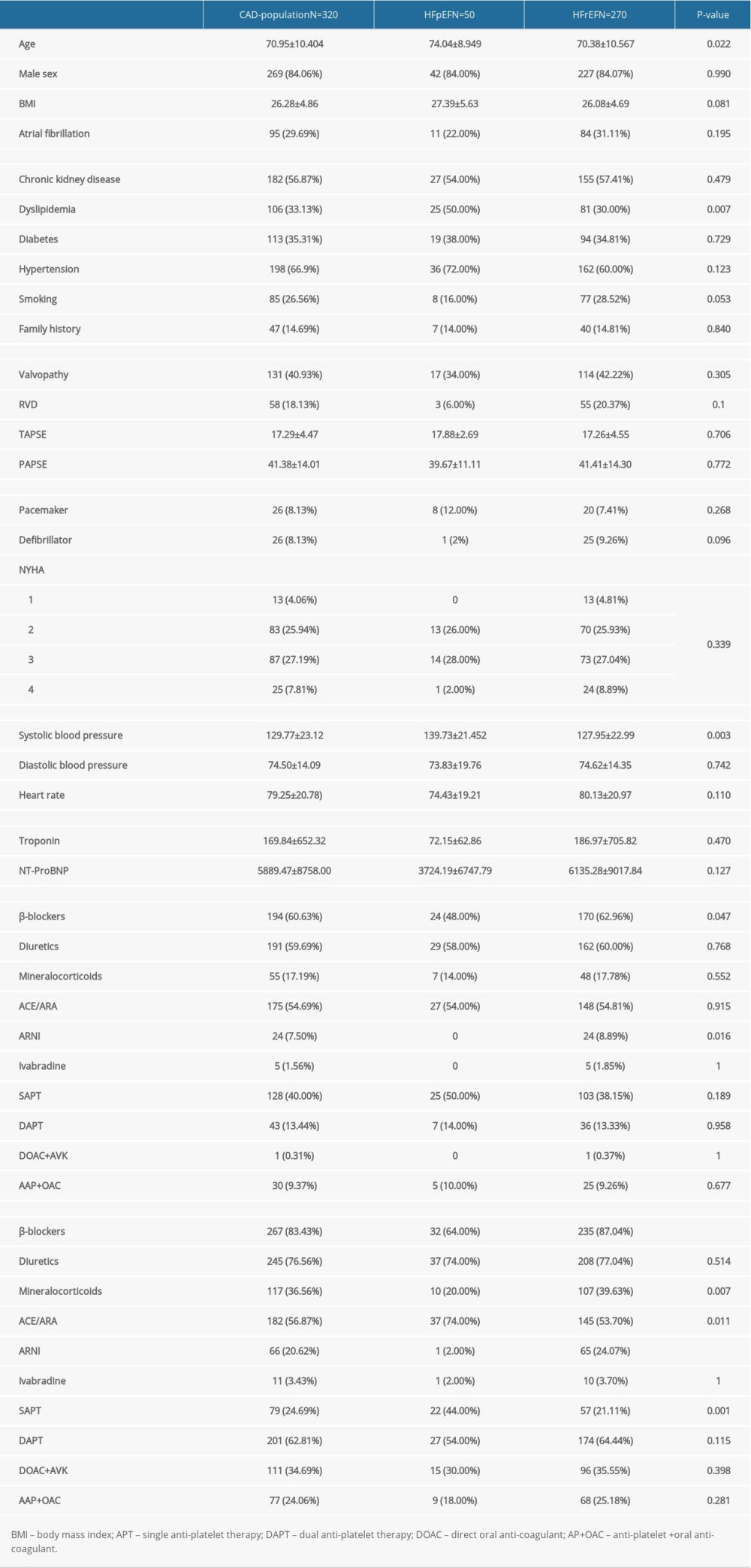
References
1. Savarese G, Lund LH, Global public health burden of heart failure: Card Fail Rev, 2017; 3; 7-11
2. Mentzer G, Hsich EM, Heart failure with reduced ejection fraction in women: Heart Fail Clin, 2019; 15; 19-27
3. Najafi F, Jamrozik K, Dobson AJ, Understanding the epidemic of heart failure: Eur J Heart Fail, 2009; 11; 472-79
4. Virani SS, Alonso A, Benjamin EJAmerican Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee, Heart disease and stroke statistics-2020 update: A report from the American Heart Association: Circulation, 2020; 141; e139-596
5. Ponikowski P, Voors AA, Anker SDESC Scientific Document Group, 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC: Eur Heart J, 2016; 37; 2129-200
6. Murphy SP, Ibrahim NE, Januzzi JL, Heart failure with reduced ejection fraction: A review: JAMA, 2020; 324; 488-504
7. Egbuche O, Hanna B, Onuorah I, Contemporary pharmacologic management of heart failure with reduced ejection fraction: A review: Curr Cardiol Rev, 2020; 16; 55-64
8. Lala A, Desai AS, The role of coronary artery disease in heart failure: Heart Failure Clin, 2014; 10; 353-65
9. Fox KF, Cowie MR, Wood DA, Coronary artery disease as the cause of incident heart failure in the population: Eur Heart J, 2001; 22; 228-36
10. Hwang SJ, Melenovsky V, Borlaug BA, Implications of coronary artery disease in heart failure with preserved ejection fraction: J Am Coll Cardiol, 2014; 63; 2817-27
11. Trevisan L, Cautela J, Resseguier N, Prevalence and characteristics of coronary artery disease in heart failure with preserved and mid-range ejection fractions: A systematic angiography approach: Archives of Cardiovascular Disease, 2018; 111; 109-18
12. Nader V, Matta A, Canitrot R, Evaluation of mitral and aortic valvular disease and left ventricular dysfunction in a Lebanese population: A single center experience: Med Sci Monit, 2021; 27; e928218
13. Azad N, Kathiravelu A, Minoosepeher S, Gender differences in the etiology of heart failure: A systematic review: J Geriatr Cardiol, 2011; 8(1); 15-23
14. Trevisan L, Cautela J, Resseguier N, Prevalence and characteristics of coronary artery disease in heart failure with preserved and mid-range ejection fractions: A systematic angiography approach: Arch Cardiovasc Dis, 2018; 111; 109-18
15. Fox KF, Cowie MR, Wood DA, Coronary artery disease as the cause of incident heart failure in the population: Eur Heart J, 2001; 22; 228-36
16. Rush CJ, Berry C, Oldroyd KJ, Prevalence of coronary artery disease and coronary microvascular dysfunction in patients with heart failure with preserved ejection fraction: JAMA Cardiol, 2021; 6; 1130-43
17. John JE, Claggett B, Skali H, CAD is a risk factor for heart failure with preserved ejection fraction: the ARIC study: J Cardiac Fail, 2019; 25; S93
18. Drissa M, Hilali S, Chebbi M, Drissa H, Ischemic heart failure versus non ischemic heart failure is there differences?: Arch Cardiovasc Dis, 2020; 12; 45
19. Lam CSP, Arnott C, Beale AL, Sex differences in heart failure: Eur Heart J, 2019; 40; 3859-68
20. Gheisari F, Emami M, Shahraki HR, The role of gender in the importance of risk factors for coronary artery disease: Cardiol Res Pract, 2020; 2020; 6527820
Figures
Tables
 Table 1. Characteristics of the studied population stratified by type of heart failure.
Table 1. Characteristics of the studied population stratified by type of heart failure. Table 2. The distribution of significant coronary artery disease.
Table 2. The distribution of significant coronary artery disease. Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease.
Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease. Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease.
Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease. Table 5. Characteristics of the study population stratified by the presence of coronary artery disease.
Table 5. Characteristics of the study population stratified by the presence of coronary artery disease. Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure.
Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure. Table 1. Characteristics of the studied population stratified by type of heart failure.
Table 1. Characteristics of the studied population stratified by type of heart failure. Table 2. The distribution of significant coronary artery disease.
Table 2. The distribution of significant coronary artery disease. Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease.
Table 3. Characteristics of heart failure with preserved ejection fraction (HFpEF) group stratified by the presence of coronary artery disease. Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease.
Table 4. Characteristics of heart failure with reduced ejection fraction (HFrEF) group stratified by the presence of coronary artery disease. Table 5. Characteristics of the study population stratified by the presence of coronary artery disease.
Table 5. Characteristics of the study population stratified by the presence of coronary artery disease. Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure.
Table 6. Characteristics of ischemic heart failure (IHF) group stratified by type of heart failure. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952