07 January 2022: Meta-Analysis
Efficacy and Safety of Light Therapy as a Home Treatment for Motor and Non-Motor Symptoms of Parkinson Disease: A Meta-Analysis
Wenjie Sun1AE, Junqiang Yan1AFG*, Jiannan Wu1BC, Hongxia Ma1BDDOI: 10.12659/MSM.935074
Med Sci Monit 2022; 28:e935074
Abstract
BACKGROUND: Non-visual effects of the retina have been increasingly confirmed in developing Parkinson disease (PD). Light therapy (LT) has been proven to be an effective non-pharmacotherapy for improving the prognosis of PD, but the pathway of action is unclear, and there is a lack of a unified and standardized LT regimen. We aimed to evaluate the efficacy and safety of various LT measures in improving motor and non-motor symptoms in patients with idiopathic PD via a meta-analysis.
MATERIAL AND METHODS: CENTRAL, EMBASE, CINAHL, PEDro, and PubMed were searched for randomized controlled trials (RCTs) investigating the efficacy of LT for PD. Cochrane’s Risk of bias tool and the GRADE approach were used to assess evidence quality. A meta-analysis and subgroup analyses evaluated the differences in efficacy produced by the different LT protocols. Trial sequential analysis (TSA) verified the analyses outcome and quantified the statistical relevance of the data.[color=#0e101a] [/color]
RESULTS: Patients receiving LT had significantly better scores for motor function (MD=-4.68, 95% Cl -8.25 to -1.12, P=0.01) compared with the control group exposed to dim-red light. In addition, in terms of non-motor symptoms, depression (SMD=-0.27, 95% Cl -0.52 to -0.02, P=0.04) and sleep disturbance-related scores (MD=3.45, 95% Cl 0.12 to 6.78, P=0.04) similarly showed significant optimization after receiving LT.
CONCLUSIONS: The results of this meta-analysis show strong evidence that LT has significant efficacy on motor and non-motor function in PD patients.
Keywords: Parkinson Disease, Phototherapy, Retinal Diseases, Humans, Symptom Assessment
Background
Parkinson disease (PD) is the second most common neurodegenerative disease of the central nervous system, affecting 2–3% of the global population over 65 years old [1]. PD is clinically characterized by motor and non-motor symptoms (NMS) that reduce the quality of life and hinder daytime functioning. Motor symptoms mainly manifest as resting tremor, rigidity, and bradykinesia, while non-motor symptoms manifest as depression, excessive daytime sleepiness, sleep disorders, fatigue, and anxiety. Long-term dopamine replacement therapy has been associated with motor complications, excessive daytime sleepiness, sleep problems, and psychiatric adverse effects [2]. Therefore, it is urgent to introduce non-drug therapy to prevent and manage complex symptoms of PD patients to reduce the clinical application of drug therapy.
Several studies have shown that the retina plays an essential role in the occurrence and development of some PD patients [3]. Notably, retinal thinning and retinal ganglion cell (RGC) loss may occur in the early stage of PD [4]. Retinal thickness is a potential imaging biomarker of cognitive decline in the early stage of PD [5,6]. The photosensitive properties of melanin secreted by intrinsically photosensitive RGCs (ipRGCs) play a crucial role in non-visual effects. These ipRGC are located at the starting point of the hypothalamic tract, and melanin is projected to many subcortical regions through them, stimulating the rhythmic production and secretion of melatonin in the pineal gland [7]. In conclusion, photosensitive melanin is the starting point for the retina to affect melatonin and circadian rhythm.
It is well known that the pathological feature of PD is the loss of dopaminergic neurons in the substantia nigra. There is abundant evidence that PD is an endocrine disease in which dopamine and melatonin work together [8,9]. Melatonin, produced by the pineal gland and retina, affects circadian and seasonal rhythms, most notably the sleep-wake cycle. The circadian rhythm can regulate dopamine rhythm. Melatonin can inhibit dopamine release [10]. Light can drive the acute suppression of melatonin and regulate the circadian rhythm by affecting the melanin system.
Studies have shown that dopamine production and release increase in light and decrease in darkness, while melatonin does the opposite [8,11]. LT before bedtime can delay the peak of melatonin secretion. Phototherapy performed during the day can decrease melatonin, which inhibits dopamine release, increasing dopamine release [12]. Additionally, dopamine levels in the retina likewise fluctuate with a circadian rhythm [13]. Studying the effect of light in the processes that are regulated by melatonin provides us with a sufficient theoretical basis (Figure 1).
LT is non-drug therapy using daily rhythmic exposure to high-intensity light with active properties which comes from sunlight, lightboxes, or other light-emitting devices. Light with active properties effectively stimulates melanocytes, such as short-wavelength monochromatic light or mixed polychromatic white light. It has been used as adjunctive therapy for many diseases for more than 30 years, including depression [14,15], bipolar disorder [16,17], seasonal affective disorder (SAD) [18], and circadian rhythm sleep disorders [19]. In recent years, several clinical studies have shown that light has great potential in treating motor and non-motor symptoms of PD [10,20]. Bright white (>3000 lx) and short-wavelength blue-green (450–500 nm) light are generally considered to have active properties, while dim-red light (<300 lx) is considered not to have active properties. The absorption peak of melanopsin is between 460 nm and 484 nm, corresponding to the short-wavelength blue-green light in the visible spectrum [21]. Blue light can activate melanin and rapidly inhibit melatonin [22]. Different wavelengths, intensities, and instruments were used for LT in the current study. However, no meta-analysis has examined the efficacy of different treatment strategies for PD by subgroup analysis.
Our study showed that both motor and non-motor functions were improved in patients with PD receiving light therapy. We analyzed the potential mechanisms of light therapy to improve motor and non-motor function. Moreover, we discuss the influence of the scheme design of light therapy on the treatment effect through subgroup analysis. The objective of this study was to evaluate the efficacy and safety of LT in improving motor and non-motor symptoms of PD patients and determine the best regimen of LT via a meta-analysis.
Material and Methods
SYSTEMATIC REVIEW STRATEGY:
We searched the Cochrane Central Register of Controlled Trials (CENTRAL), EMBASE, CINAHL, PubMed, the Physiotherapy Evidence Database (PEDro), and the Cochrane Movement Disorders Group trials register. We also searched clinical trials registries such as
INCLUSION AND EXCLUSION CRITERIA:
The review included all (RCTs) including different methods to compare LT with control treatment. The inclusion criteria were: (1) according to the accepted criteria, participants must be diagnosed with idiopathic PD by a neurologist (as defined by the UK Parkinson’s Disease Society brain bank criteria or other), accompanied by different degrees of excessive daytime sleepiness, sleep disorders, depression, and other related symptoms; (2) all patients in the included studies were treated with a stable PD regimen for at least 4 weeks before inclusion; (3) if participants take sedatives, hypnotics, or psychotropic drugs simultaneously, they should also take them consistently for at least 4 weeks to avoid interfering with the evaluation. Exclusion criteria were: (1) Eye diseases that interfere with vision; (2) Cognitive impairment; (3) Serious mental illness; (4) Severe dyskinesia.
We included various forms with LT as active interventions, such as bright light (polychromatic white light; 3000 lux–10 000 lux) and blue-green light. We did not limit LT in terms of duration, time of day, device, or wavelength. Acceptable control interventions were any form of light less than 300 lux.
We excluded studies that were duplicates, irrelevant, marked as ineligible by automation tools, did not meet the inclusion criteria, or were not retrieved. The excluded articles and detailed reasons for exclusion are listed in the “Characteristics of studies” file.
After integration, we selected 5 primary outcomes (motor function, depression, daytime sleepiness, sleep, and adverse events) and 3 secondary outcomes (fatigue, anxiety, and quality of life).
DATA EXTRACTION:
Two reviewers independently extracted data from the included studies using the data extraction form we designed for this review. We resolved differences by reaching a consensus or inviting other reviewers. We extracted the publication details (first author name, year of publication, manuscript title, cohort name, country of cohort), research design information (randomization, allocation sequence hiding, blind method, time frame, research environment), participant characteristics (age, gender, course of disease, severity, country, baseline scores for specific symptoms), interventions (equipment, intensity, wavelength, frequency and time, distance), and outcome measures (results and time points, definitions of related outcomes, and measurement units). One reviewer entered the data into Review Manager 5.4 [24], and another reviewer checked the accuracy of the data.
STATISTICAL ANALYSES:
We followed the guidelines in the Cochrane Handbook [25] for Systematic Reviews of Interventions for data analyses [26]. We carried out data analysis using Review Manager 5.4 and STATA (version 14.0, StataCorp, USA). We used a fixed-effects model for all studies because the value of heterogeneity was less than 50%. For continuous outcomes, the effect measure is the mean difference (MD) with 95% CI. If scales of different lengths are used, and if we judged that the outcome measured was similar enough, we used the standardized mean difference (SMD) with 95% CI. We presented results as risk ratios (RR) for dichotomous outcomes with 95% confidence intervals (CI). We contacted all authors of the included studies to obtain missing information. For continuous measures, where primary outcome data were not provided in the form of a mean and standard deviation, where possible, we obtained means or SDs from other measures, such as medians, quartiles, means change from baseline, and SDs change from baseline.
If indicated, the authors examined heterogeneity between trial results using a standard chi-squared test. For the heterogeneity adjustment of the diversity-adjusted required information size (DARIS) in the Trial Sequential Analysis, we used diversity (D2) because the I2 statistic used for this purpose might underestimate the required information size [27]. We formally assessed publication bias using funnel plots, performing Egger’s test [28].
ASSESSMENT OF RISK OF BIAS AND GRADE:
Two review authors independently assessed risk of bias of included randomized trials using the Cochrane ‘Risk of bias’ tool, as described in the Cochrane Handbook for Systematic Reviews of Interventions. We resolved discrepancies by reaching consensus or by involving another review author.
We assessed the certainty of the evidence using the GRADE system to present review results in ‘Summary of findings’ (SoF) tables, using GRADEPro 3.6 (http://ims.cochrane.org.ermg.femh.org.tw:81/revman/gradepro) [29]. The GRADE approach appraises the quality of a body of evidence-based on the extent to which one can be confident that an estimate of effect or association reflects the item being assessed [30]. Considerations are due to the within-trial risk of bias, directness of the evidence, data heterogeneity, precision of effect estimates, and risk of publication bias. In SoF tables, we included 5 Primary outcomes (motor functions, depression, daytime sleepiness, sleep, adverse events). The GRADE approach results in an assessment of the quality of a body of evidence in 1 of 4 grades (high certainty, moderate certainty, low certainty, very low certainty).
SUBGROUP ANALYSIS:
All the following groups were included by more than 3 trials. The MD was calculated of these subgroups.
TRIAL SEQUENTIAL ANALYSIS:
We performed Trial Sequential Analysis to preserve the risk of type 1 and type 2 errors at desired levels in the setting of sparse data and potential repeated testing [31]. For all meta-analyses performed, we used Trial Sequential Analysis to verify the power adequacy and adjust the 95% confidence intervals if the data were sparse [32,33]. Preserving a type 1 error risk of 5% and a type 2 error risk of 20%, we constructed monitoring boundaries using the pooled SD for continuous data and the unweighted mean of the control event rate for categorical data, and the diversity was calculated from the actual meta-analysis [34]. The diversity-adjusted required information size (RIS) on the incidence of adverse events was calculated based on incidence in the control group of 22.2%; risk ratio reduction of 30% in the experimental group; type I error of 5%; and type II error of 20% (80% power).
Result
RESULTS OF THE SEARCH:
We identified 2193 records through the database (2145 studies) and trials register (48 studies) searches. After merging the search results and discarding the duplicates, the 2 authors examined the cited titles and abstracts, respectively, to identify studies that might be included in the review. If the title or abstract appeared to meet our inclusion criteria, we evaluated the eligibility of the included trials based on the full text. From the 2193 titles and abstracts retrieved, we assessed 16 articles for eligibility. We excluded studies that did not meet the predefined inclusion criteria, such as non-randomized controlled trials. Following a thorough screening, we identified 15 articles. The articles by Sonja and colleagues were, in fact, a single study with 2 bibliographic references. Five trials were excluded [35–39]. As such, we included 10 trials in the qualitative analyses. Excluding 4 ongoing trials and 1 study with only abstracts, we included 5 studies in the quantitative analyses [40–44]. We documented the selection process in sufficient detail to complete the PRISMA flowchart (Figure 2) [32]. The excluded articles and the reasons for exclusion are listed in the “Characteristics of studies” file. The 2 authors resolved their differences through discussion or referred to another author if necessary. If there was a lack of relevant information, we contacted the study authors. We tracked all the results in the EndNote X9 database.
INCLUDED STUDIES:
The evaluation included 5 RCTs. A total of 231 participants with PD were included, of which 156 were male (59%) and 110 were female (41%). Reported mean ages ranged between 58.9 and 70.8 years old. The patients came from 5 countries (USA, Germany, The Netherlands, Belgium, Australia). Four trials described the severity of EDS, and participants with mean baseline ESS >12 accounted for 24.7%. The demographic characteristics of all studies are shown in Table 1.
Two studies used lightboxes (Sun Ray Sunbox SB-558; Brazil, Lumie, Cambridge UK). Three other studies used SpectraMax light therapy devices, portable head-mounted devices (Luminette®, Belgium), or fluorescent tubes (Apollo BL-6) phototherapy devices. The device was placed at a distance of 0.2 m to 1 m, at an angle or eye level. For therapy of seasonal affective disorder, the illuminance of 10 000 lux for 30 min daily had been proven to be most effective, while illuminance of less than 2500 lux is thought to be ineffective. Participants in the 3 studies were exposed to white or polychromatic light with various intensities (from 3000 to 10 000 lux). The other 2 were exposed to blue/green light (450–570 nm). In addition, light therapy was performed once or twice a day (in the morning or evening) with a duration of 30 min to 60 min. All studies used phototherapy devices with the same appearance as the active intervention group in the placebo control group. Placebo phototherapy was performed in various colors, such as red, orange, or polychromatic, and intensity was less than 300 lux. Almost all of the studies were conducted at home, and only 1 was conducted in a hospital [44]. The equipment and relevant parameters used in all studies are shown in Table 2. In addition, Table 3 shows all the clinical variables in each included study, SoF, and demonstrates all the methods of measuring primary and secondary outcomes.
RISK OF BIAS AND QUALITY ASSESSMENT:
Detailed reasons for assessing the risk of bias are recorded in the “Characteristics of studies”. In the ‘Risk of bias’ tables, we recorded this information and presented a summary’ Risk of bias’ figure (Figure 3). We considered 4 studies at low risk and Videnovic 2017 at unclear risk of bias due to insufficient information on how the random assignment was performed. All trials reported adequate blinding of the outcome assessor and participants. We did not find incomplete outcome data or selective reporting. The studies were found to be “low risk of bias” when these studies were subjected to the Cochrane Collaboration’s Tool for Quality Assessment of Randomized Controlled Trials.
EFFECTS OF INTERVENTIONS:
The GRADE evaluation results are displayed in Table 3.
MOTOR FUNCTION: Four trials with 209 participants recorded motor function measured by Unified Parkinson’s Disease Rating Scale subscale Part III (URPDS III) [40–42,44]. Pooled analysis showed a statistically significant difference between bright light therapy and placebo (MD=−4.68; 95% CI −8.25 to −1.12; P=0.01) (Figure 4A), with low heterogeneity (I2=0%, P=0.63), proving that the light treatment was better than a placebo. On the whole, the movement symptom was obviously improved. The homogeneity test indicated no significant heterogeneity (I2=0%) (Figure 4A).
DEPRESSION: Five trials with 234 participants measured improvement of depression after light treatment by using the Beck Depression Inventory (BDI) and Hamilton Depression Rating Scale (HDRS). There was a significant improvement in depression measured by BDI and HDRS in the light treatment group compared with the control group (SMD=−0.27; 95% CI −0.52 to −0.02; P=0.04; I2=0%; high quality of evidence) (Figure 4B). No statistical heterogeneity was present.
DAYTIME SLEEPINESS: Four studies used the Epworth Sleepiness Scale (ESS) [40,42–44], and 1 study used the SCOPA-daytime sleepiness (higher scores mean worse daytime sleepiness) involving 241 participants with PD assessing the effects of bright light treatment compared to the dim light treatment group on daytime sleepiness [42]. There was no statistically significant improvement in the bright light treatment group for daytime sleepiness. Assessment of statistical heterogeneity among trials indicated I2=0%, indicating no apparent heterogeneity (Figure 4C).
SLEEP: Two studies evaluated the effectiveness of LT in sleep involving 122 participants [40,42]. Data showed a statistically significant difference in sleep measured by the Parkinson’s Disease Sleep Scale (PDSS) (MD=3.45, 95% Cl 0.12 to 6.78, P=0.04, n=122). The heterogeneity test indicated no significant heterogeneity between the LT and placebo groups (I2=0%; high quality of evidence) (Figure 4D).
ADVERSE EVENTS: The adverse events in the LT group were significantly higher than those in the placebo group (RR=2.65,95% Cl 1.38 to 5.09, P=0.003, n=231) (Figure 4E). However, adverse events were described in all studies as mild and transient ocular symptoms and headache, and no serious adverse events were reported.
SECONDARY OUTCOMES (FATIGUE; ANXIETY; AUALITY OF LIFE): Three trials involving 81 participants with PD assessed fatigue utilizing the Brief Bronowski Scale (BBS), the Fatigue Impact Scale (FIS), and Fatigue Severity Scale (FSS). This study demonstrated no significant improvement in the LT group (SMD=−0.13, 95% Cl −0.57 to 0.30, P=0.55, n=81) (Figure 5A). Three trials involving 141 participants with PD assessed anxiety by Beck Anxiety Inventory (BAI) and Hamilton Depression Rating Scale (HDRS). This study demonstrated no significant improvement in the LT group (MD=−0.01, 95% Cl −0.34 to 0.33, P=0.98, n=141) (Figure 5B). Four studies involving 209 participants with PD investigated the effects of LT on quality of life, using the PDQ-39 as an outcome measure of the quality of life. This study showed no significant difference between LT and active control interventions (MD=0.71, 95% Cl −4.12 to 5.55, P=0.77, n=209), whereby LT was found not to be superior (Figure 5C).
SUBGROUP ANALYSIS:
We performed a subgroup analysis to compare the effects of different treatment courses, duration, time, wavelength, and devices on motor function, depression, and daytime sleepiness.
COURSE OF TREATMENT: Interestingly, when we deleted 3 studies (158/209 participants) for short-term treatment [40,44], those with long-term treatment showed more significant changes for motor functions (MD=−5.33; 95% CI −9.28 to −1.37; P=0.008) [41,42]. However, we did not find such changes in different treatment courses for other non-motor functions. Short-term treatment could reduce the adverse reactions (RR=1.88, 95% Cl 0.19 to 18.60, P=0.59, n=51).
DAILY PERIODS: In terms of the effect on motor symptoms, the 2 studies providing treatment both in the morning and at night showed a more significant improvement than those treating only at night (MD=−5.39; 95% CI −9.21 to −1.58; P=0.006) [40,41].
DAILY DURATION: Continuous light treatment for 30 min (MD=−5.33; 95% CI −9.28 to −1.37; P=0.008) [41] was associated with significantly better motor functions as compared to other trials using continuous LT for 45–60 min. However, depression improved more significantly in the 60-min subgroup of LT (MD=−0.32; 95% CI −0.62 to −0.02; P=0.04).
OPTICAL WAVELENGTH: Three studies with 118/209 participants used polychromatic white light in the active treatment group [40,41,44]. We found a more significant change in motor functions in the polychromatic white light treatment subgroup (MD=−5.06; 95% CI −8.82 to −1.30; P=0.008) [42,43].
DEVICES: Two studies [40,41] with 98 participants using lightbox treatment were compared with 2 studies [42,44] with 111 participants using other devices, showing statistically significant improvement in the lightbox subgroups on motor functions (MD=−5.39; 95% CI −9.21 to −1.58; P=0.006). There was no difference between the 2 subgroups in other non-motor measures, including depression and daytime sleepiness.
TRIAL SEQUENTIAL ANALYSIS (TSA) AND PUBLICATION BIAS:
We performed TSA on the primary outcome. In TSA for motor function, the cumulative Z-curve (blue line) crossed the trial sequential monitoring boundaries (red inward sloping lines), indicating the conclusion was sufficient and no more trials were needed. The cumulative Z-score (blue line) did not cross the RIS (red vertical line) for depression, daytime sleepiness, and sleep. The cumulative Z-curve (blue line) did not cross the trial sequential monitoring boundaries (red inward sloping lines), suggesting that further trials are required (Figure 6A–6E). All the P values for the 5 primary outcomes were over 0.05 using Egger’s test. Funnel plots and Egger’s test, as 2 methods used to evaluate publication bias for selected outcomes of LT for Parkinson disease, did not exhibit significant publication bias (Figure 7).
Discussion
EFFECT OF LIGHT THERAPY ON MOTOR FUNCTION:
According to the UPDRS III scoring system, 4 studies reported that light therapy could significantly improve motor function. Paus et al reported improvement of tremor, bradykinesia, and rigidity, but the control light was too strong to form an effective contrast [37]. It has been reported that patients without tremors secrete less melatonin than patients with severe tremors [45]. We speculate that the improvement of motor function is due to the regulation of circadian rhythm, inhibiting melatonin and promote dopamine secretion [11]. The biological clock of the retina may indirectly affect the dopamine rhythm by adjusting the melatonin rhythm, or it may directly affect the biological clock of dopamine secretion by dopamine neurons [46]. Secondly, light can cause retinal dopamine neurons to secrete dopamine [11,46]. studies have shown that infrared light, but not red light, can protect damaged dopamine neurons [47]. Another possible explanation is that LT can protect dopamine neurons and increase dopamine secretion.
Our study shows that in studies of less than 6-month duration, motor symptoms seem to be more effective with increased treatment time. The treatment effect was the best at 6 months, and the UPDRS III score improved by 5.33. Paradoxically, the quality of life has not been improved. At present, there is no study lasting longer than 6 months. It is impossible to determine whether the motor symptoms improve to a greater extent with the longer treatment and whether the quality of life will also improve.
EFFECT OF LIGHT THERAPY ON DEPRESSION:
The depressive symptoms of patients with PD exposed to bright light were significantly improved. Previous studies have shown that phototherapy can effectively improve expression and seasonal affective disorder (SAD). However, the placebo effect is also considered to have the ability to improve depression, nor does it rule out the possibility that the sleep-wake cycle affects depressive symptoms [48]. Further study on LT’s mechanism will help clarify the potential mechanism by which LT improves depression.
EFFECTS OF LIGHT THERAPY ON SLEEP AND EXCESSIVE DAYTIME SLEEPINESS:
Multiple reports suggest that LT has beneficial effects on the sleep-wake cycle in patients with PD, enhancing sleep and daytime alertness. According to Willis et al, sleep parameters decreased by 50% in the 2-week trial, illustrating that sleep symptoms improved early in light therapy [35]. Studies have shown that blue/green light can effectively stimulate melatonin and improve alertness [49]. Other studies showed that the sensitivity of the blue-yellow pathway is higher in the early stage of PD [50]. Exposure to blue/green light before bedtime can delay the peak of melatonin secretion, while waking up in the morning can increase daytime arousal [51]. Fabin Lin et al also published a systematic review and meta-analysis of RCT regarding the effect of bright light therapy (BLT) on depression and sleep disturbances in patients with PD, and they demonstrated that BLT was an effective intervention for improving depressive symptoms and sleep disturbances in patients with PD [52]. Their results were similar to ours, but they only focused on BLT using 3000–10 000 lux.
EFFECT OF LIGHT THERAPY ON OTHER NON-MOTOR SYMPTOMS:
In addition to depression, sleep disturbance, and excessive daytime sleepiness, our study demonstrates that fatigue and anxiety are also significantly improved by LT in patients with PD. The study by Paus et al showed that LT significantly improved mood in patients with PD [37]. In winter, the spectrum changes, and the blue light component decreases. Blue light has been shown to effectively promote the brain’s processing of emotions [53]. ipRGCs in the suprachiasmatic nucleus (SCN) and the perihabenular nucleus (PHb) are the 2 retina–brain pathways that regulate light’s effects on emotion and cognition [54].
ADVERSE EVENTS:
There was no mention of significant adverse effects in the reviewed studies, only minor eye discomfort, and headache. With a longer duration of LT at home, it is likely to result in better efficacy while potentially increasing adverse effects. Once transient mild adverse effects appeared, we considered that adjusting the frequency and 1–2 weeks washout period could resolve these adverse effects.
LIGHT THERAPY SCHEMES:
The studies reviewed mostly assessed LT administered in the patients’ homes. Light therapy using lightboxes is easy to operate, allowing patients to listen to music, news, or audiobooks during treatment. The long course of treatment may achieve better results, but it may also increase the probability of eye discomfort and headache [55]. Current treatment regimens do not unify daily time points, and we believe that being performed simultaneously at bedtime and after getting started early is more beneficial for symptom improvement. As melatonin secretion peaks at between 22: 00–23: 00, LT at bedtime helps regulate circadian rhythms [43]. In contrast, LT in the early morning suppressed melatonin secretion, while cortisol peaked [41]. Treatment duration in the current study was controlled at 30–60 min, and there were no regimens beyond 60 min, making it impossible to determine whether 60 min or less is the optimal treatment duration for PD. In the treatment of depression, a time length of less than 60 min is considered to have a better therapeutic effect. Polychromatic white light may be more effective in improving motor symptoms, and blue-green light may be better for depression [56]. Previous studies have shown that blue light, which enhances melanin expression and light responses in the retina, is critical in treating depression [57,58]. Both bright white and blue light appears to have significant efficacy, but the effects of high-sensitivity blue light on adverse effects are not well-defined. Head-mounted equipment is now available in addition to the lightbox, which affects the pupil’s angle and distance of the light source. However, the effect of both near (≤50 cm) and far (>50 cm) LT on efficacy remains uncertain and awaits further study.
CLINICAL SIGNIFICANCE OF LIGHT THERAPY:
LT as a noninvasive and convenient treatment have been proved to be effective not only in PD but also for other conditions [10,14–20]. Motor symptoms and non-motor symptoms including depression, sleep, sleep disturbance, excessive daytime sleepiness, fatigue, and anxiety can affect quality of life of patients. It will be of clinical significance to use LT to treat PD patients to improve motor and non-motor symptoms and improve their quality of life. However, the overall change in UPDRS III score was about 4 to 5, while the quality-of-life score did not change. We speculate that the possible reason for this result was that the quality-of-life score may reflect the quality of life of the previous month, which cannot reflect the effect of LT in time. Longer follow-up is needed to determine the impact of LT on quality of life. Further research may help to verify this hypothesis.
LIMITATIONS:
A limitation of this meta-analysis is that there were only 5 RCTs suitable for quantitative analysis, so their publication bias could not be effectively assessed. Moreover, the current sample size is not enough to support us in comparing the efficacy of different treatment schemes, which can be used as the goal of future research. Secondly, most of the included studies were small-sample studies with less than 50 subjects. In addition, as a family treatment, long-term observation is required. The current research schemes are guided by staff to use phototherapy equipment, and long-term treatment compliance cannot be evaluated.
Conclusions
The overall evidence shows that LT is a noninvasive, non-pharmacological, and well-tolerated intervention. This review shows significant improvements in motor function, depression, and sleep disorders after LT treatment. Even if we believe that LT can improve excessive daytime sleepiness, the existing evidence is insufficient to form firm conclusions, and more experiments are needed. We argue that LT may reduce the time to onset of dopamine replacement medications, or can be used as a levodopa-sparing strategy or adjuvant therapy. Considering its economic benefits and good compliance, we think it would be popular with families. Further research is needed to verify the benefits of LT for excessive daytime sleepiness, fatigue, and anxiety. Further research is needed to determine the best course of treatment, daily time, dose, light intensity, and wavelength of LT for each symptom.
Figures
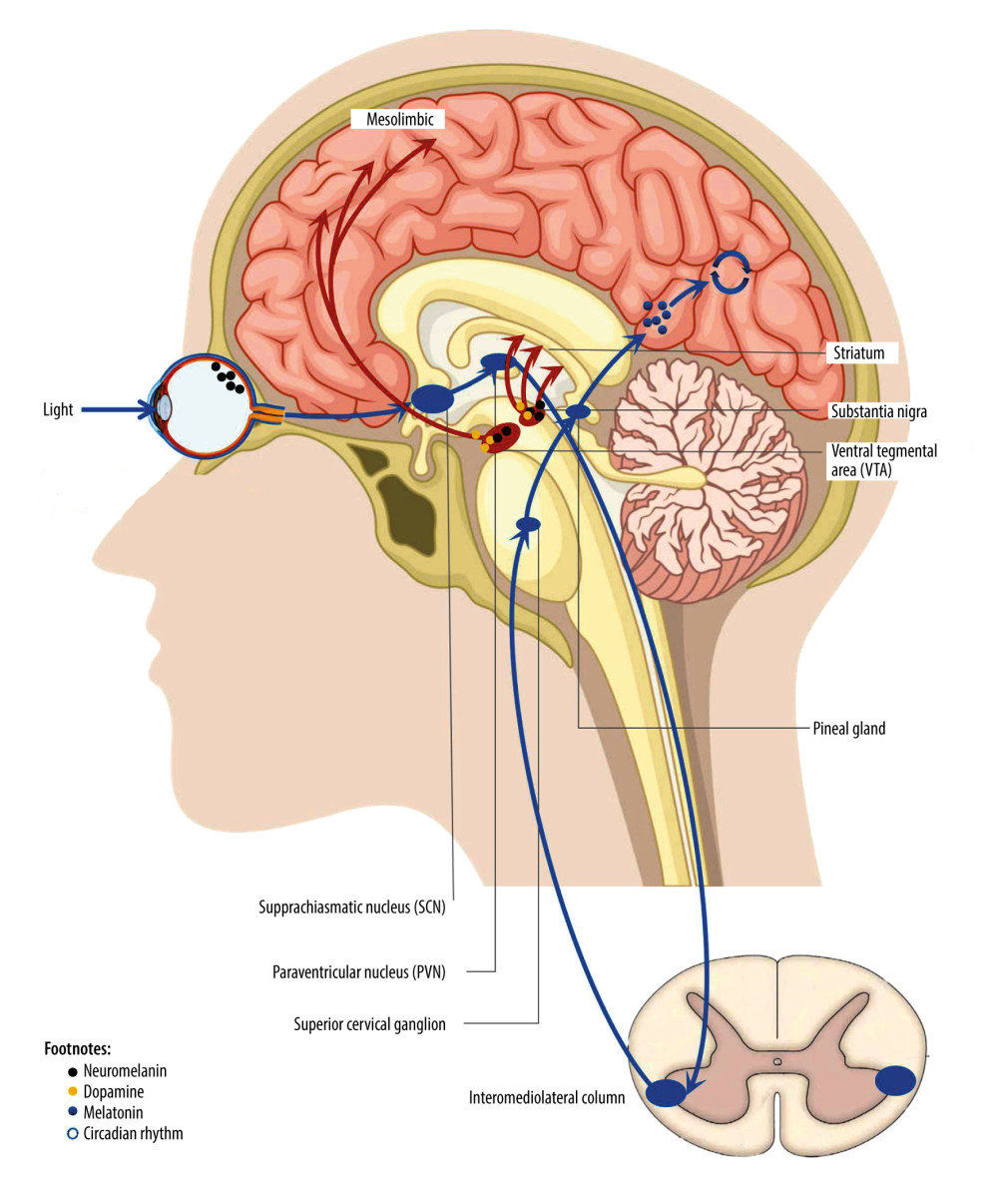 Figure 1. Non-visual effect pathway of retina.
Figure 1. Non-visual effect pathway of retina. 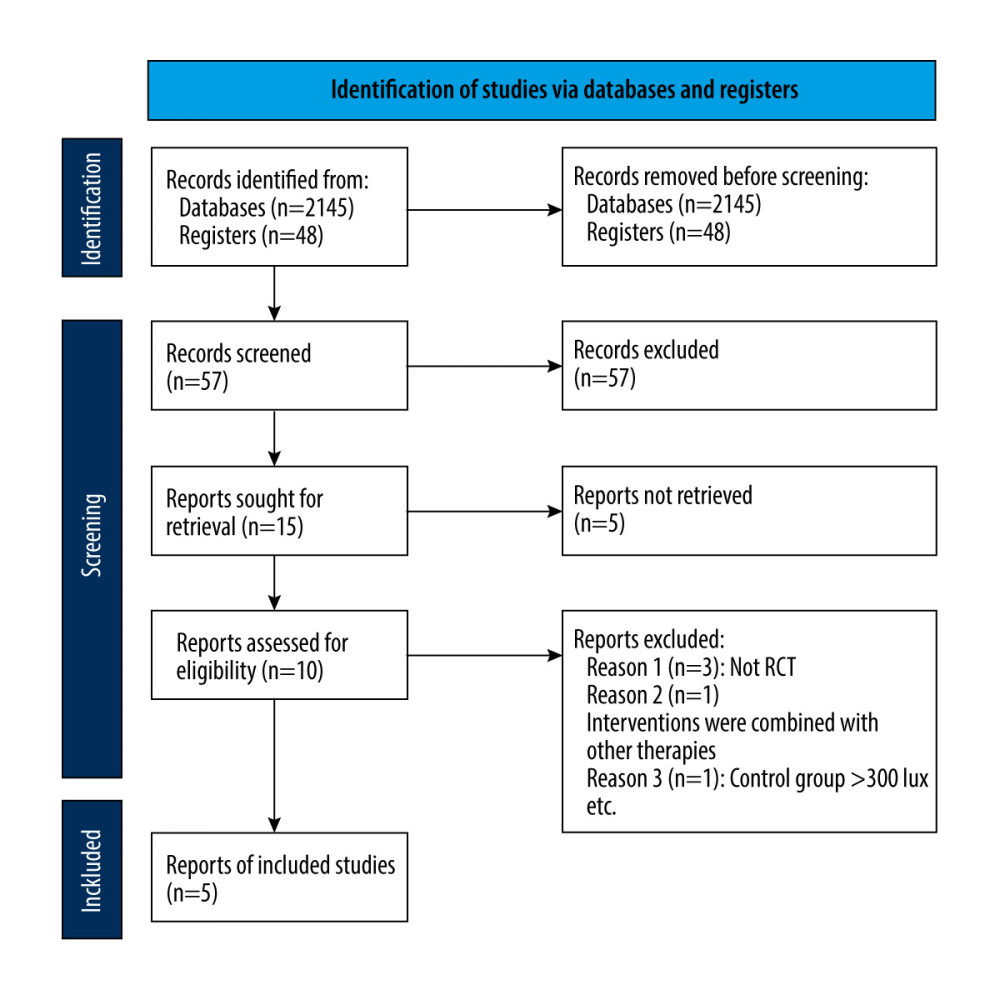 Figure 2. PRISMA flow diagram.
Figure 2. PRISMA flow diagram. 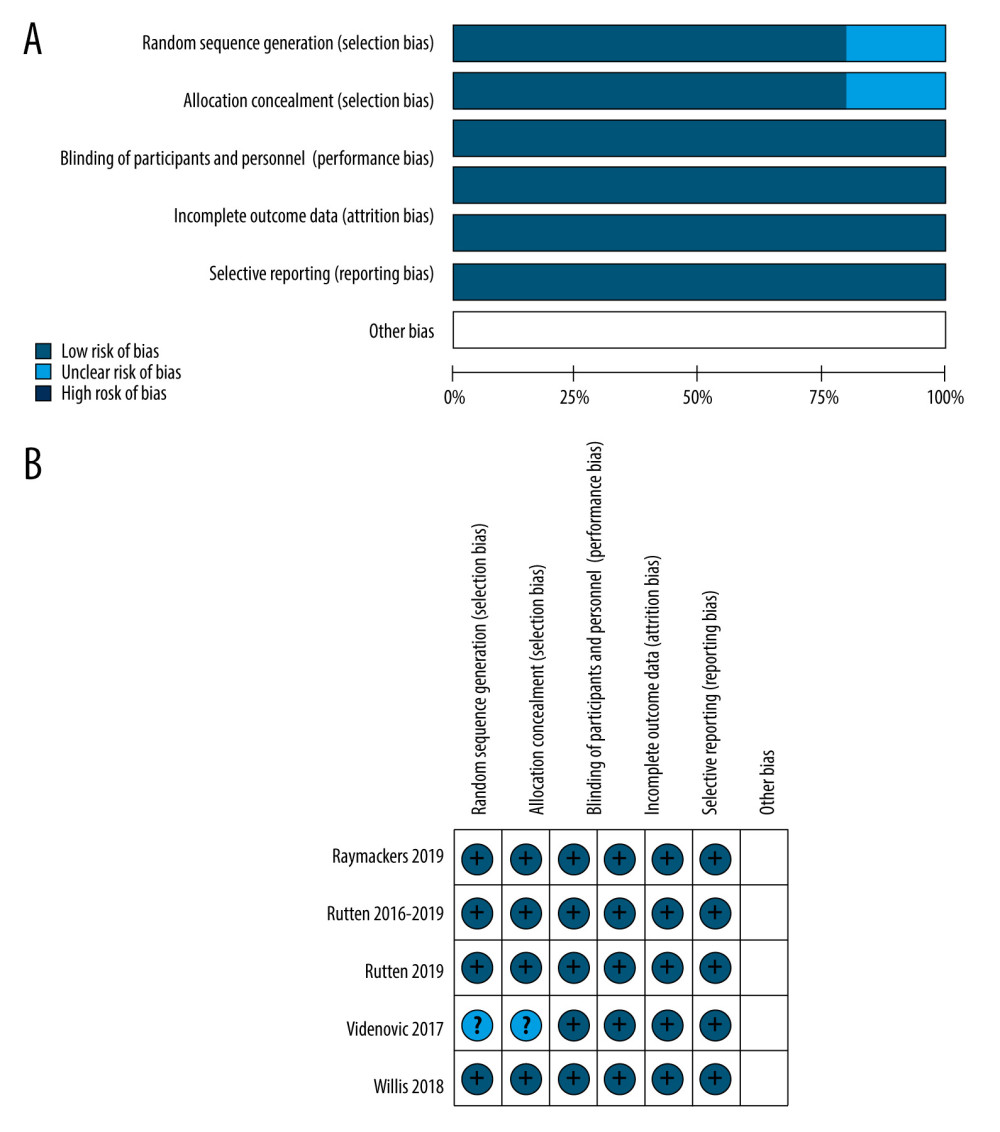 Figure 3. Risk of bias assessment of each included study. (A) Risk of bias graph. (B) Risk of bias summary. Review Manager 5.4 was used to create the pictures.
Figure 3. Risk of bias assessment of each included study. (A) Risk of bias graph. (B) Risk of bias summary. Review Manager 5.4 was used to create the pictures. 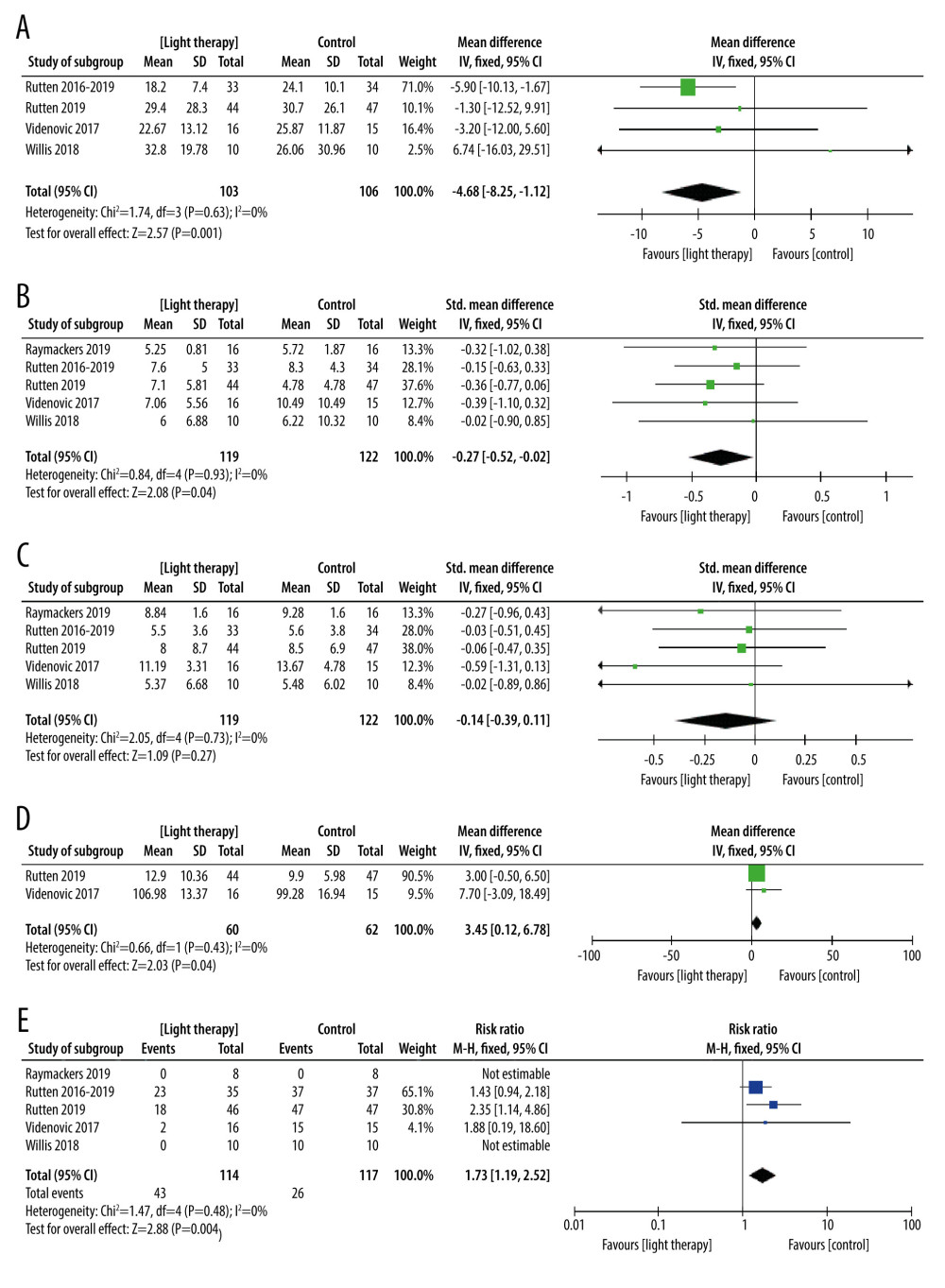 Figure 4. The forest plot of the meta-analysis of randomized clinical trials compared motor function (A), depression status (B), excessive daytime sleepiness (C), sleep disorders (D), and adverse events (E) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures.
Figure 4. The forest plot of the meta-analysis of randomized clinical trials compared motor function (A), depression status (B), excessive daytime sleepiness (C), sleep disorders (D), and adverse events (E) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures. 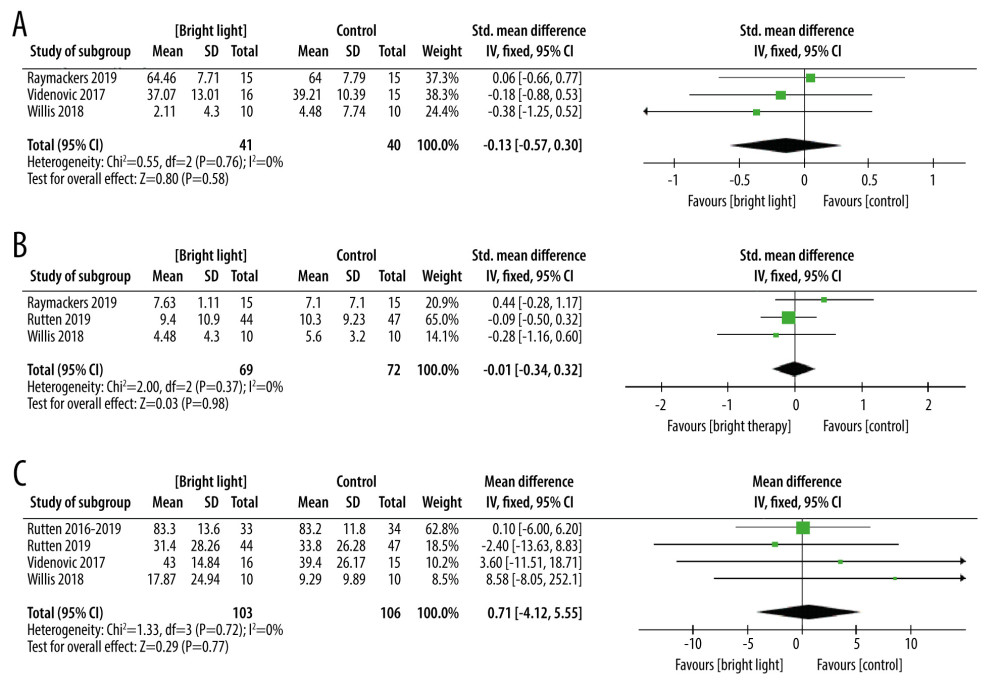 Figure 5. The forest plot of the meta-analysis of randomized clinical trials compared fatigue (A), anxiety (B) and quality of life (C) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures.
Figure 5. The forest plot of the meta-analysis of randomized clinical trials compared fatigue (A), anxiety (B) and quality of life (C) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures. 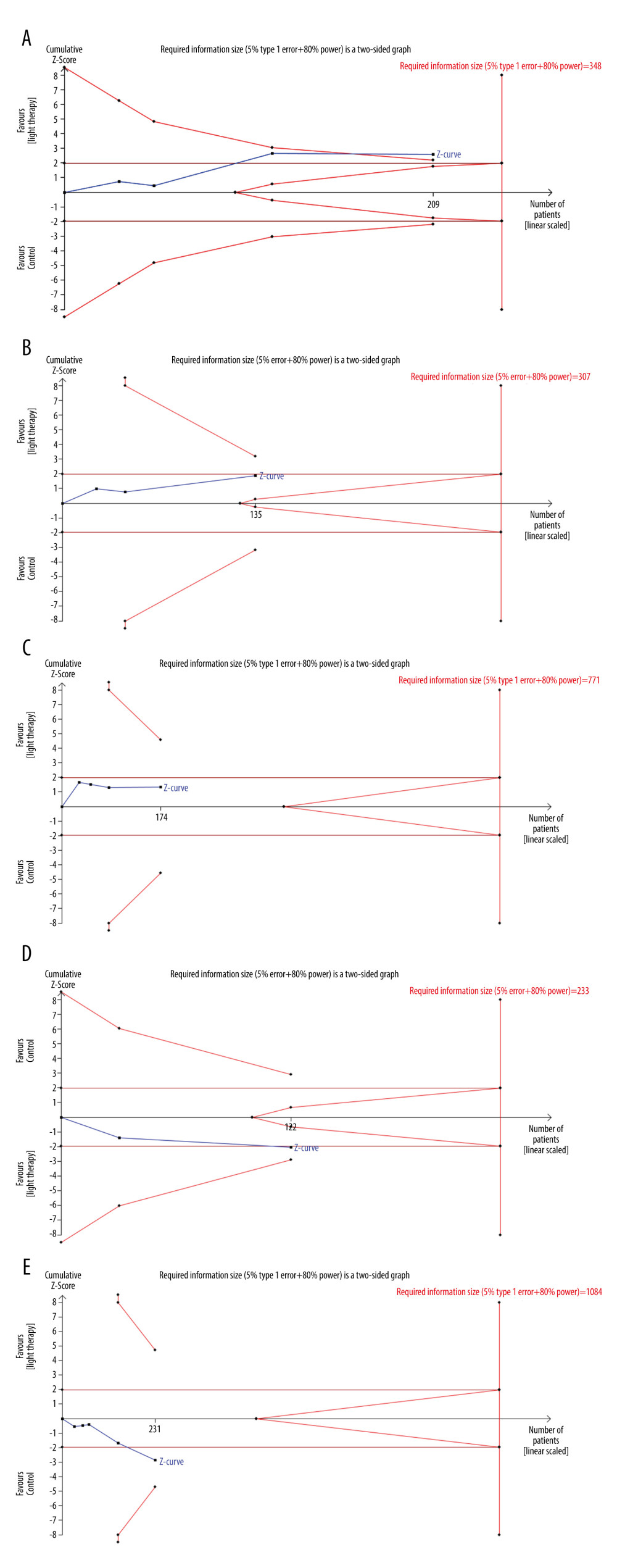 Figure 6. Trial sequential analysis on the effect of light therapy for improved motor function (A), depressive status (B), excessive daytime sleepiness (C), sleep disturbance (D), and adverse effects (E). Blue lines – the cumulative Z-score; Red vertical lines – the required information size (RIS); Red inward sloping lines – the trial sequential monitoring boundaries; Brown lines – the conventional statistical boundaries (P=0.05). TSA (Version 0.9.5.10) was used to create the figures.
Figure 6. Trial sequential analysis on the effect of light therapy for improved motor function (A), depressive status (B), excessive daytime sleepiness (C), sleep disturbance (D), and adverse effects (E). Blue lines – the cumulative Z-score; Red vertical lines – the required information size (RIS); Red inward sloping lines – the trial sequential monitoring boundaries; Brown lines – the conventional statistical boundaries (P=0.05). TSA (Version 0.9.5.10) was used to create the figures. 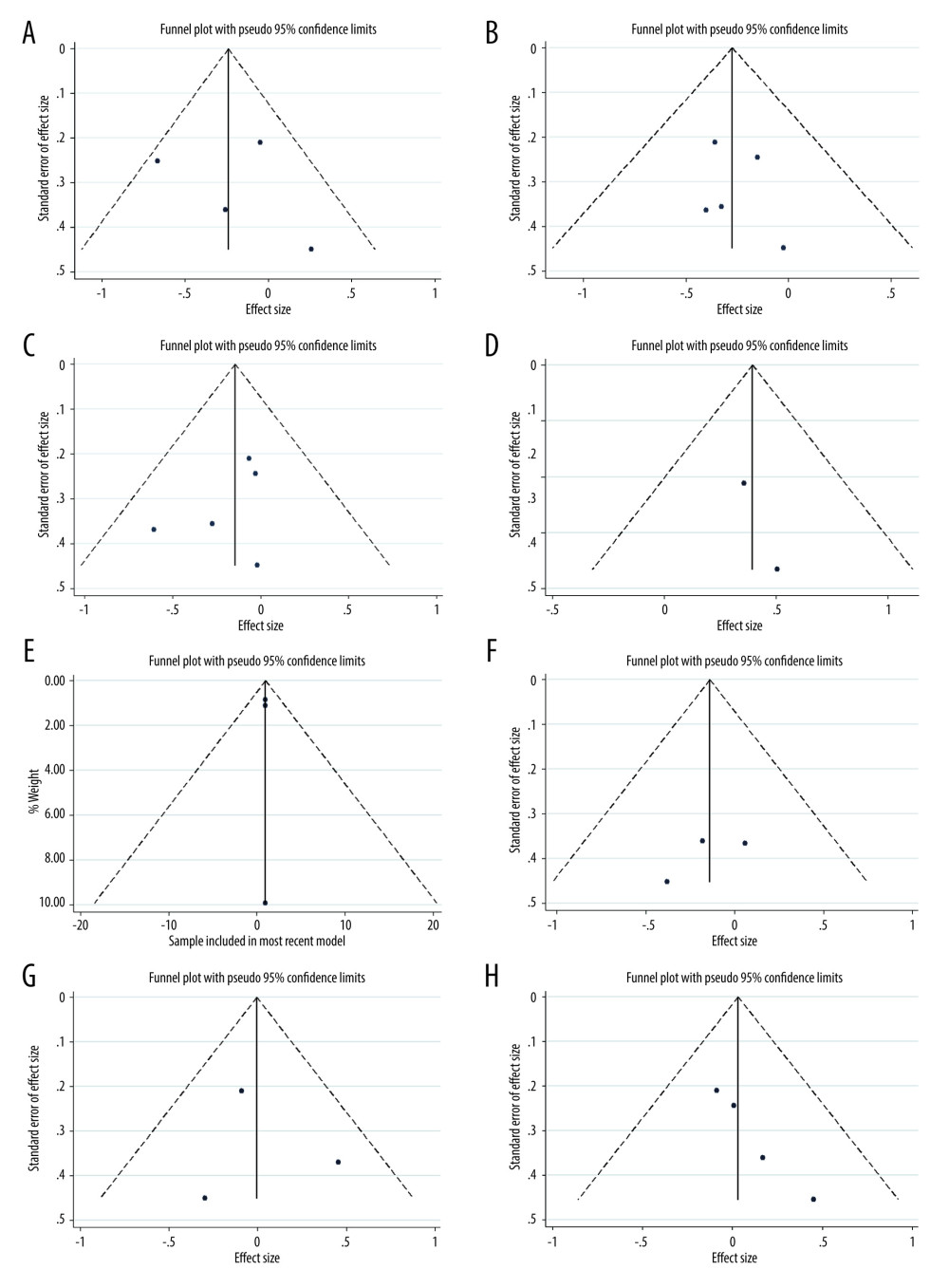 Figure 7. Funnel plot of the estimated publication bias of primary and secondary outcomes. (A) motor function, (B) depression, (C) daytime sleepiness, (D) sleep, (E) adverse events, (F) fatigue, (G) anxiety, and (H) quality of life. Stata (Version 14.0) was used to create the figures.
Figure 7. Funnel plot of the estimated publication bias of primary and secondary outcomes. (A) motor function, (B) depression, (C) daytime sleepiness, (D) sleep, (E) adverse events, (F) fatigue, (G) anxiety, and (H) quality of life. Stata (Version 14.0) was used to create the figures. References
1. Poewe W, Seppi K, Tanner CM, Parkinson disease: Nat Rev Dis Primers, 2017; 3; 17013
2. Gonzalez-Latapi P, Bhowmick SS, Saranza G, Fox SH, Non-dopaminergic treatments for motor control in Parkinson’s disease: An update: CNS Drugs, 2020; 34(10); 1025-44
3. Vandewalle G, Maquet P, Dijk DJ, Light as a modulator of cognitive brain function: Trends Cogn Sci, 2009; 13(10); 429-438
4. Ahn J, Lee JY, Kim TW, Retinal thinning associates with nigral dopaminergic loss in de novo Parkinson disease: Neurology, 2018; 91(11); e1003-e12
5. Murueta-Goyena A, Del Pino R, Galdos M, Retinal thickness predicts the risk of cognitive decline in Parkinson disease: Ann Neurol, 2021; 89(1); 165-76
6. Murueta-Goyena A, Gabilondo I, Reply to: Clinical and cognitive correlates of selective regional retina thinning in dementia with lewy bodies: Mov Disord, 2019; 34(10); 1584
7. Fernandez DC, Fogerson PM, Lazzerini Ospri L, Light affects mood and learning through distinct retina–brain pathways: Cell, 2018; 175(1); 71-84.e18
8. Willis GL, Parkinson’s disease as a neuroendocrine disorder of circadian function: Dopamine-melatonin imbalance and the visual system in the genesis and progression of the degenerative process: Rev Neurosci, 2008; 19(4–5); 234-316
9. Shen Y, Guo X, Han C, The implication of neuronimmunoendocrine (NIE) modulatory network in the pathophysiologic process of Parkinson’s disease: Cell Mol Life Sci, 2017; 74(20); 3741-68
10. Schmoll C, Lascaratos G, Dhillon B, The role of retinal regulation of sleep in health and disease: Sleep Med Rev, 2011; 15(2); 107-13
11. Li Z, Tian T, Light therapy promoting dopamine release by stimulating retina in Parkinson disease: JAMA Neurol, 2017; 74(10); 1267-68
12. Zhang Z, Silveyra E, Jin N, Ribelayga CP, A congenic line of the C57BL/6J mouse strain that is proficient in melatonin synthesis: J Pineal Res, 2018; 65(3); e12509
13. Jacoby J, Nath A, Jessen ZF, Schwartz GW, A self-regulating gap junction network of amacrine cells controls nitric oxide release in the retina: Neuron, 2018; 100(5); 1149-1162.e1145
14. Rutten S, Vriend C, Smit JH, A double-blind randomized controlled trial to assess the effect of bright light therapy on depression in patients with Parkinson’s disease: BMC Psychiatry, 2016; 16(1); 355
15. Perera S, Eisen R, Bhatt M, Light therapy for non-seasonal depression: Systematic review and meta-analysis: BJ Psych Open, 2016; 2(2); 116-26
16. Sit DK, McGowan J, Wiltrout C, Adjunctive bright light therapy for bipolar depression: A randomized double-blind placebo-controlled trial: Am J Psychiatry, 2018; 175(2); 131-39
17. Gold AK, Kinrys G, Treating circadian rhythm disruption in bipolar disorder: Curr Psychiatry Rep, 2019; 21(3); 14
18. Pjrek E, Friedrich ME, Cambioli L, The efficacy of light therapy in the treatment of seasonal affective disorder: A meta-analysis of randomized controlled trials: Psychother Psychosom, 2020; 89(1); 17-24
19. van Maanen A, Meijer AM, van der Heijden KB, Oort FJ, The effects of light therapy on sleep problems: A systematic review and meta-analysis: Sleep Med Rev, 2016; 29; 52-62
20. Fifel K, Videnovic A, Light therapy in Parkinson’s disease: Towards mechanism-based protocols: Trends Neurosci, 2018; 41(5); 252-54
21. Do MTH, Melanopsin and the intrinsically photosensitive retinal ganglion cells: Biophysics to behavior: Neuron, 2019; 104(2); 205-26
22. Wahl S, Engelhardt M, Schaupp P, The inner clock-Blue light sets the human rhythm: J Biophotonics, 2019; 12(12); e201900102
23. Page MJ, McKenzie JE, Bossuyt PM, The PRISMA 2020 statement: An updated guideline for reporting systematic reviews: PLoS Med, 2021; 18(3); e1003583
24. Nordic Cochrane Centre: The Cochrane CollaborationReview Manager 5 (RevMan 5). Version 5.4, 2019, Copenhagen, Nordic Cochrane Centre, The Cochrane Collaboration
25. Higgins JP, Altman DG, Sterne JA: Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011)
26. Deeks JJ, Higgins JPT, Altman DGon behalf of the Cochrane Statistical Methods Group, Chapter 9: Analysing data and undertaking meta-analyses: Cochrane, 2017
27. Brok J, Thorlund K, Gluud C, Wetterslev J, Trial sequential analysis reveals insufficient information size and potentially false positive results in many meta-analyses: J Clin Epidemiol, 2008; 61(8); 763-69
28. Egger M, Davey Smith G, Bias in meta-analysis detected by a simple, graphical test: BMJ (Clinical Research Ed), 1997; 315(7109); 629-34
29. McMaster University(developed by Evidence Prime): GRADEpro GDT. Version, 2015, Hamilton (ON), McMaster University (developed by Evidence Prime)
30. GRADE Working Group: GRADE handbook for grading quality of evidence and strength of recommendations, 2013 guidelinedevelopment.org/handbook
31. Wetterslev J, Thorlund K, Brok J, Gluud C, Estimating required information size by quantifying diversity in random-effects model meta-analyses: BMC Med Res Methodol, 2009; 9; 86
32. Copenhagen Trial Unit: TSA – Trial Sequential Analysis. Version 0.9.5.10 Beta, 2011, Copenhagen, Copenhagen Trial Unit
33. Imberger G, Thorlund K, Gluud C, Wetterslev J, False-positive findings in Cochrane meta-analyses with and without application of trial sequential analysis: An empirical review: BMJ Open, 2016; 6(8); e011890
34. Thorlund K, Engstrøm J, Wetterslev J: User manual for Trial Sequential Analysis (TSA), 2011 ctu.dk/tsa/files/tsa_manual.pdf
35. Willis GL, Turner DEJ, Primary and secondary features of Parkinson’s disease improve with strategic exposure to bright light: A case series study: Chronobiol Int, 2007; 24(3); 521-37
36. Martino JK, Freelance CB, Willis GL, The effect of light exposure on insomnia and nocturnal movement in Parkinson’s disease: An open label, retrospective, longitudinal study: Sleep Med, 2018; 44; 24-31
37. Paus S, Schmitz-Hubsch T, Wullner U, Bright light therapy in Parkinson’s disease: A pilot study: Mov Disord, 2007; 22(10); 1495-98
38. Rios Romenets S, Creti L, Fichten C, Doxepin and cognitive behavioural therapy for insomnia in patients with Parkinson’s disease – a randomized study: Parkinsonism Relat Disord, 2013; 19(7); 670-75
39. Endo T, Matsumura R, Tokuda IT, Bright light improves sleep in patients with Parkinson’s disease: Possible role of circadian restoration: Sci Rep, 2020; 10(1); 7982
40. Videnovic A, Klerman EB, Wang W, Timed light therapy for sleep and daytime sleepiness associated with Parkinson disease: A randomized clinical trial: JAMA Neurol, 2017; 74(4); 411-18
41. Rutten S, Vriend C, Smit JH, Bright light therapy for depression in Parkinson disease: A randomized controlled trial: Neurology, 2019; 92(11); e1145-56
42. Rutten S, Vriend C, Smit JH, A double-blind randomized controlled trial to assess the effect of bright light therapy on depression in patients with Parkinson’s disease: BMC Psychiatry, 2016; 16(1); 355
43. Raymackers JM, Andrade M, Baey E, Bright light therapy with a head-mounted device for anxiety, depression, sleepiness and fatigue in patients with Parkinson’s disease: Acta Neurol Belg, 2019; 119(4); 607-13
44. Willis GL, Boda J, Freelance CB, Polychromatic light exposure as a therapeutic in the treatment and management of Parkinson’s disease: A controlled exploratory trial: Front Neurol, 2018; 9; 741
45. Videnovic A, Noble C, Reid KJ, Circadian melatonin rhythm and excessive daytime sleepiness in Parkinson disease: JAMA Neurol, 2014; 71(4); 463-69
46. Chaffiol A, Ishii M, Cao Y, Mangel SC, Dopamine regulation of GABAA receptors contributes to light/dark modulation of the ON-Cone bipolar cell receptive field surround in the retina: Curr Biol, 2017; 27(17); 2600-2609.e4
47. Moro C, El Massri N, Darlot F, Effects of a higher dose of near-infrared light on clinical signs and neuroprotection in a monkey model of Parkinson’s disease: Brain Res, 2016; 1648(Pt A); 19-26
48. Tao L, Jiang R, Zhang K, Light therapy in non-seasonal depression: An update meta-analysis: Psychiatry Res, 2020; 291; 113247
49. Walsh CM, Prendergast RL, Sheridan JT, Murphy BA, Blue light from light-emitting diodes directed at a single eye elicits a dose-dependent suppression of melatonin in horses: Vet J, 2013; 196(2); 231-35
50. Sartucci F, Porciatti V, Visual-evoked potentials to onset of chromatic red-green and blue-yellow gratings in Parkinson’s disease never treated with L-Dopa: J Clin Neurophysiol, 2006; 23(5); 431-35
51. Smilowska K, van Wamelen DJ, Schoutens AMC, Blue light therapy glasses in Parkinson’s disease: Patients’ experience: Parkinsons Dis, 2019; 2019; 1906271
52. Lin F, Su Y, Weng Y, The effects of bright light therapy on depression and sleep disturbances in patients with Parkinson’s disease: A systematic review and meta-analysis of randomized controlled trials: Sleep Med, 2021; 83; 280-89
53. Vandewalle G, Hebert M, Beaulieu C, Abnormal hypothalamic response to light in seasonal affective disorder: Biol Psychiatry, 2011; 70(10); 954-61
54. Fernandez DC, Fogerson PM, Lazzerini Ospri L, Light affects mood and learning through distinct retina-brain pathways: Cell, 2018; 175(1); 71-84.e18
55. Meesters Y, Winthorst WH, Duijzer WB, Hommes V, The effects of low-intensity narrow-band blue-light treatment compared to bright white-light treatment in sub-syndromal seasonal affective disorder: BMC Psychiatry, 2016; 16; 27
56. Emens JS, Burgess HJ, Effect of light and melatonin and other melatonin receptor agonists on human circadian physiology: Sleep Med Clin, 2015; 10(4); 435-53
57. Meng Q, Jiang J, Hou X, Antidepressant effect of blue light on depressive phenotype in light-deprived male rats: J Neuropathol Exp Neurol, 2020; 79(12); 1344-53
58. Bilu C, Einat H, Tal-Krivisky K, Red white and blue-bright light effects in a diurnal rodent model for seasonal affective disorder: Chronobiol Int, 2019; 36(7); 919-26
Figures
 Figure 1. Non-visual effect pathway of retina.
Figure 1. Non-visual effect pathway of retina. Figure 2. PRISMA flow diagram.
Figure 2. PRISMA flow diagram. Figure 3. Risk of bias assessment of each included study. (A) Risk of bias graph. (B) Risk of bias summary. Review Manager 5.4 was used to create the pictures.
Figure 3. Risk of bias assessment of each included study. (A) Risk of bias graph. (B) Risk of bias summary. Review Manager 5.4 was used to create the pictures. Figure 4. The forest plot of the meta-analysis of randomized clinical trials compared motor function (A), depression status (B), excessive daytime sleepiness (C), sleep disorders (D), and adverse events (E) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures.
Figure 4. The forest plot of the meta-analysis of randomized clinical trials compared motor function (A), depression status (B), excessive daytime sleepiness (C), sleep disorders (D), and adverse events (E) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures. Figure 5. The forest plot of the meta-analysis of randomized clinical trials compared fatigue (A), anxiety (B) and quality of life (C) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures.
Figure 5. The forest plot of the meta-analysis of randomized clinical trials compared fatigue (A), anxiety (B) and quality of life (C) in patients with Parkinson disease treated with light therapy and placebo. Review Manager 5.4 was used to create the figures. Figure 6. Trial sequential analysis on the effect of light therapy for improved motor function (A), depressive status (B), excessive daytime sleepiness (C), sleep disturbance (D), and adverse effects (E). Blue lines – the cumulative Z-score; Red vertical lines – the required information size (RIS); Red inward sloping lines – the trial sequential monitoring boundaries; Brown lines – the conventional statistical boundaries (P=0.05). TSA (Version 0.9.5.10) was used to create the figures.
Figure 6. Trial sequential analysis on the effect of light therapy for improved motor function (A), depressive status (B), excessive daytime sleepiness (C), sleep disturbance (D), and adverse effects (E). Blue lines – the cumulative Z-score; Red vertical lines – the required information size (RIS); Red inward sloping lines – the trial sequential monitoring boundaries; Brown lines – the conventional statistical boundaries (P=0.05). TSA (Version 0.9.5.10) was used to create the figures. Figure 7. Funnel plot of the estimated publication bias of primary and secondary outcomes. (A) motor function, (B) depression, (C) daytime sleepiness, (D) sleep, (E) adverse events, (F) fatigue, (G) anxiety, and (H) quality of life. Stata (Version 14.0) was used to create the figures.
Figure 7. Funnel plot of the estimated publication bias of primary and secondary outcomes. (A) motor function, (B) depression, (C) daytime sleepiness, (D) sleep, (E) adverse events, (F) fatigue, (G) anxiety, and (H) quality of life. Stata (Version 14.0) was used to create the figures. Tables
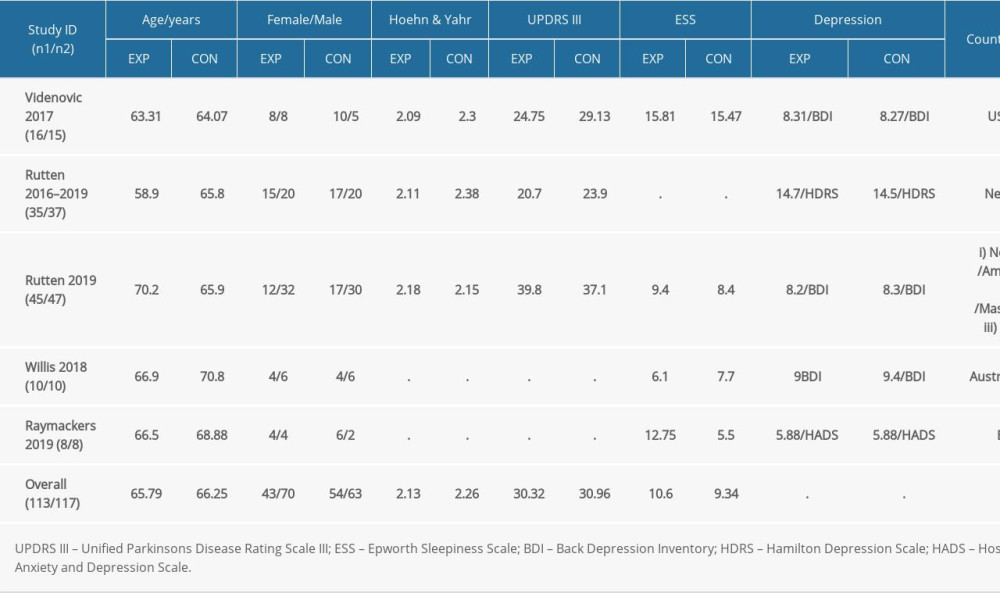 Table 1. Demographic and clinical data of the included study.
Table 1. Demographic and clinical data of the included study.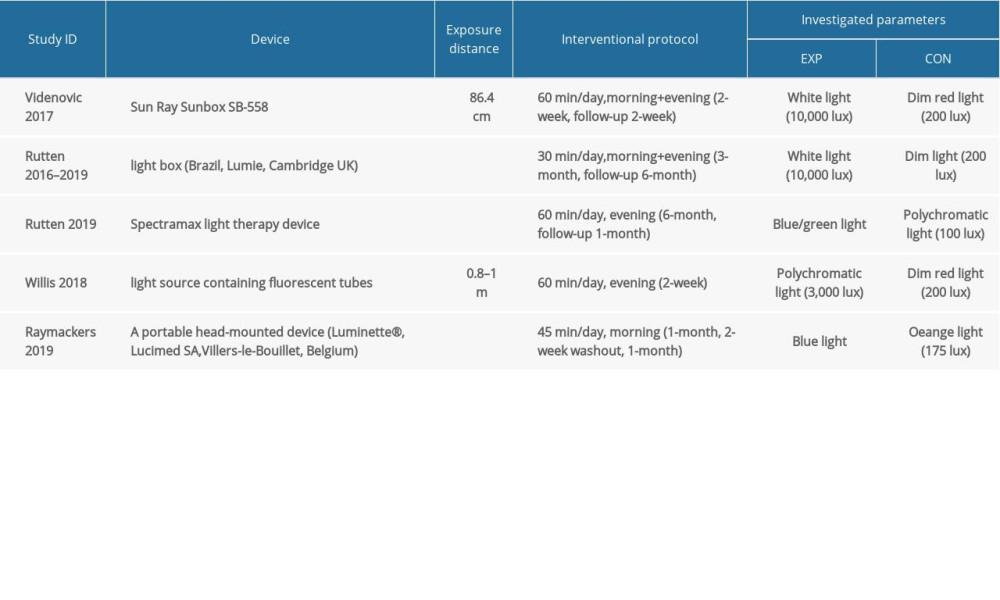 Table 2. Equipment and relevant parameters of all studies used in LT.
Table 2. Equipment and relevant parameters of all studies used in LT.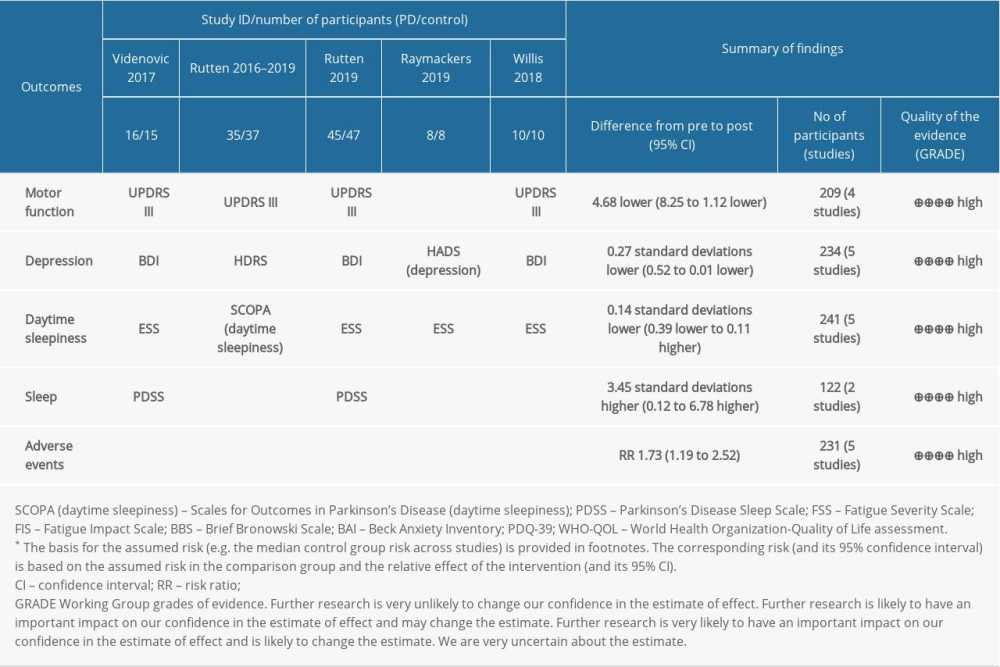 Table 3. Methods of measuring primary outcomes and summary of finding (SoF). GRADEprofiler was used to create the table.
Table 3. Methods of measuring primary outcomes and summary of finding (SoF). GRADEprofiler was used to create the table. Table 1. Demographic and clinical data of the included study.
Table 1. Demographic and clinical data of the included study. Table 2. Equipment and relevant parameters of all studies used in LT.
Table 2. Equipment and relevant parameters of all studies used in LT. Table 3. Methods of measuring primary outcomes and summary of finding (SoF). GRADEprofiler was used to create the table.
Table 3. Methods of measuring primary outcomes and summary of finding (SoF). GRADEprofiler was used to create the table. In Press
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








