22 August 2020: Clinical Research
Effect of the Combination of Ketorolac and Bupivacaine on Transversus Abdominis Plane Block for Postoperative Analgesia After Gynecological Laparoscopic Surgery
Qi Jiang1ABCDEF, Shao-Qiang Huang1ACEF*, Jing Jiao1BCD, Xiao-Min Zhou1BCDDOI: 10.12659/MSM.925006
Med Sci Monit 2020; 26:e925006
Abstract
BACKGROUND: This study assessed the additional benefits of bupivacaine when combined with ketorolac for transversus abdominis plane (TAP) block after gynecological laparoscopic surgery.
MATERIAL AND METHODS: This randomized, observer-blind trial recruited 153 patients who underwent gynecological laparoscopic surgery. Patients were randomly assigned to receive bupivacaine combined with ketorolac 15 mg/side for TAP block (TK group), bupivacaine for TAP block and 30 mg postoperative intravenous ketorolac (T group), or 30 mg postoperative intravenous ketorolac alone (C group). The primary endpoints included consumption of sufentanil for 24 h postoperatively, actual press times of the patient-controlled analgesia (PCA) pump, and effective press times of the PCA pump, whereas the secondary endpoints included numerical rating scale (NRS) pain scores at rest and during activity, satisfaction with analgesia, episodes of nausea and vomiting and length of hospital stay.
RESULTS: Sufentanil consumption, actual press times of the PCA pump, and effective press times of the PCA pump were lower in the TK and T groups than in the C group. NRS scores at rest and during activity at 1, 2, 4, 6, and 24 hours were significantly lower in the TK and T groups than in the C group. The TK and T groups showed greater satisfaction with analgesia than the C group, while the TK group showed greater overall satisfaction than the C group. Lengths of stay, rates of nausea and vomiting, and venting times did not differ significantly among the three groups.
CONCLUSIONS: Combined ketorolac and bupivacaine as TAP block improved the effectiveness of analgesia without increasing adverse events. Trial registration number: ChiCTR1900022577.
Keywords: Bupivacaine, Gynecologic Surgical Procedures, Ketorolac, Abdominal Muscles, Adolescent, Analgesia, Patient-Controlled, Analgesics, Opioid, Anesthetics, Intravenous, Anesthetics, Local, Drug Therapy, Combination, Laparoscopy, Morphine, Nerve Block, pain management, Pain Measurement, Pain, Postoperative, Patient Satisfaction, Single-Blind Method, Sufentanil, young adult
Background
Because it is minimally invasive, laparoscopic surgery has several advantages over open surgery, including reduced trauma and postoperative pain, and shorter hospital stay. Advances in laparoscopic surgical instruments and techniques have enabled laparoscopic surgery to be performed in most surgical fields, including gynecology [1]. Although the incidence of postoperative discomfort was found to be lower in patients who underwent laparoscopic than traditional gynecologic surgery, 35–80% of patients who underwent laparoscopic surgery experienced postoperative pain, with the pain lasting up to 72 hours [2,3]. Effective analgesic strategies are therefore needed to reduce discomfort after gynecological laparoscopic surgery.
The delivery of local anesthetic to the transversus abdominis plane (TAP) has shown promise in reducing pain, with ultrasound-guided TAP block associated with better pain control after lower abdominal surgery [4–6]. Moreover, TAP block could reduce postoperative nausea and vomiting induced by intravenous analgesics [7]. The mixture of ketorolac with lidocaine or bupivacaine has been associated with longer local anesthetic time and reduced postoperative pain [8]. However, it is unclear whether TAP block with the mixture of bupivacaine plus ketorolac is more effective than keterolac alone in controlling postoperative pain after gynecologic laparoscopic surgery. This randomized, observer-blinded trial therefore assessed the efficacy and safety of combined bupivacaine and ketorolac as TAP block for patients after gynecologic laparoscopic surgery.
Material and Methods
TRIAL DESIGN AND OVERSIGHT:
This study was a randomized, observer-blinded, 3-armed, single center clinical trial. The protocol and amendments were approved by the ethics committee of our hospital, and the sponsor was responsible for data collection. The first draft of the manuscript was completed by the first author, with the coauthors contributing to revisions of the manuscript. Submission of the manuscript for publication and responsibility for the accuracy and completeness of the data and analyses were per protocol and at the sponsor’s discretion.
TRIAL POPULATION:
The study included women aged 18–60.0 years, classified as ASA I–III who underwent elective gynecologic laparoscopic surgery and were indicated for postoperative intravenous analgesia. Enrolled patients were instructed on use of the patient-controlled analgesia (PCA) device, and all patients provided written informed consent. Patients were excluded if they (1) were users of narcotic analgesics or sedatives or long-term users of non-steroidal anti-inflammatory drugs; (2) experienced allergic reactions to the study drugs; (3) had a history of disorders of the neuromuscular, cardiovascular, or endocrine system or allergic or mental illnesses; (4) had a body mass index >30 kg/m2 or <18 kg/m2; (5) had a history of peptic ulcer or bleeding; (6) had a blood-clotting disorder; (7) were pregnant or lactating; (8) were addicted to drugs of abuse or alcohol; and (9) were deemed unsuitable for the study by the researcher.
RANDOMIZATION AND STUDY AGENTS:
Eligible patients were randomly assigned in a 1: 1: 1 ratio to receive bupivacaine combined with ketorolac (Sichuan Medcalo Pharmaceutical Co., Ltd) 15 mg/side for TAP block (TK group), bupivacaine for TAP blocks and 30 mg postoperative intravenous ketorolac (T group), or 30 mg postoperative intravenous ketorolac alone (C group). Randomization was performed out in an observer-blinded manner using a central computerized system and random number table.
ANESTHESIA:
None of the patients was administered drugs preoperatively. After the patient entered the operating room, her right upper limb vein was opened with an 18 G trocar, followed by inhalation of 5 L/min oxygen through the anesthesia mask. Baseline non-invasive blood pressure, electrocardiogram, heart rate and pulse oxygen saturation were recorded by a multi-function monitor. Anesthesia was induced by intravenous injection of a mixture of sufentanil (5 μg/kg), propofol (2 mg/kg), and succinylcholine (1.5 mg/kg), and was maintained at a BIS of 40–60 by administration of remifentanil (0.2 μg/kg.min) and desflurane. Sufentanil (5 μg/kg) was added if a patient’s heart rate or blood pressure increased by at least 20%, with 4 mg ondansetron administered intravenously to prevent postoperative nausea and vomiting.
POSTOPERATIVE ANALGESIA:
Patients in the T and C groups were administered 30 mg ketorolac postoperatively. Patients in the T and TK groups were administered ultrasound-guided bilateral TAP block at the time of postoperative wound coverage. Briefly, patients were placed in the supine position and covered with a sterile towel that had undergone iodine-volt disinfection. Ultrasonic images were obtained with an ultrasonic instrument using a high-frequency probe of 6~13 Hz wrapped in a sterile glove, and the nerve block operation was performed under ultrasound guidance. After the completion on one side, TAP block was performed on the opposite side in the same manner.
ENDPOINTS:
The primary endpoints included consumption of sufentanil for 24 hours postoperatively, actual press times of the PCA pump, and effective press times of the PCA pump. Secondary endpoints included numerical rating scale (NRS) pain scores at rest and during activity, satisfaction with analgesia, length of hospital stay, episodes of nausea and vomiting, and venting times. These outcomes were collected and analyzed by investigators who were blinded to the study group assignments.
STATISTICAL ANALYSIS:
Continuous data are reported as the mean (standard deviation) and median (quartile), depending on the normality of data distribution. Categorical data are reported as the number (percent) of patients and compared among the three groups by CMH or Fisher’s exact test. Numerical outcomes in the three study groups were compared by ANOVA, with pairwise comparisons assessed by LSD methods. Satisfaction with analgesia is reported as ranked data, with the Kruskal-Wallis method used for comparisons among the three groups. All analyses were performed on an intention-to-treat basis, with missing data not imputed. The sample size was based on the consumption of sufentanil after laparoscopic surgery of about 22±13 g, an analgesic efficacy ratio of sufentanil to morphine [9], alpha=0.05 and beta=0.20. All reported P-values are two-sided, with P-values <0.05 considered statistically significant. All statistical analyses were performed using the IBM Statistical Package for the Social Sciences software for Windows, version 19.0.
Results
PATIENTS:
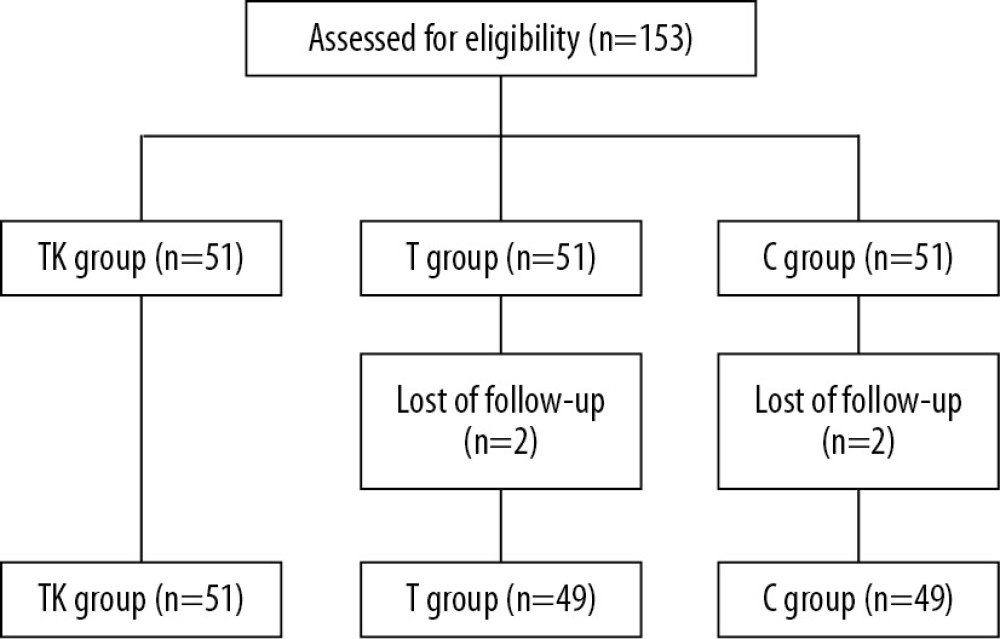
From July 2018 to November 2018, 153 patients were randomized, 51 each to the TK, T, and C groups (Figure 1). Two patients in the T group discontinued, resulting in the removal of two patients in the C group (Supplementary Table 1). Table 1 summarizes the baseline characteristics of the patients in the three groups. Their mean age was 40.28 years, their mean body mass index (BMI) was 22.29 kg/m2, and the mean duration of the operation was 56.21 minutes. The patient population included 34 with a medical history of diseases, and seven with other diseases and drug use. There were no significant differences among the three groups in mean age, height, weight, BMI, duration of operation, medical history, combination of disease and medication, diastolic blood pressure, heart rate and SpO2. However, systolic blood pressure was significantly lower in the T group than in the TK and C groups (P=0.0377).
PRIMARY EFFICACY OUTCOMES:
Table 2 shows the primary efficacy outcomes in the TK, T, and C groups. Significant differences were observed among the three groups in the consumption of sufentanil over 24 hours, actual press times of the PCA pump, and effective press times of the PCA pump (P<0.001 each). Sufentanil consumption was lower in the TK (10.49±5.36 ml) and T (13.42±6.39 ml) groups than in the C group (25.26±11.19 ml); whereas there was no significant difference between the TK and T groups. The actual press times of the PCA pump were also significantly lower in the TK (4.39±2.81 min) and T (5.53±2.78 min) groups than in the C group (10.41±4.72 min), but did not differ significantly in the TK and T groups. In additin, the effective press times of the PCA pump were significantly lower in the TK (4.20±2.14 min) and T (5.37±2.56 min) groups than in the C group (10.10±4.48 min), with no significant difference between the TK and T groups.
SECONDARY OUTCOMES:
Table 3 shows NRS pain scores in the three groups at rest and during activity measured 1, 2, 4, 6, and 24 hours after surgery. Mean NRS pain scores at rest and during activity were lower in the TK and T groups than in the C group at 1 and 2 hours. Although there were no significant differences between the TK and T groups at 1 hour, NRS pain scores at rest were lower in the TK group than in the T group (1.75±0.89) at 2 hours. At 4 hours, NRS pain scores at rest and during activity were lower in the TK group than in the T and C groups, and NRS pain scores during activity, but not at rest, were lower in the T group than in the C group. At both 6 and 24 hours, NRS pain scores at rest and during activity were lower in the TK group than in the T and C groups, with no significant differences between the T and C groups.
There were no significant differences among the three groups in the incidence of nausea and vomiting at all time points (Supplementary Table 2), mean venting times (Supplementary Table 3), and duration of hospital stay (Table 4). Satisfaction with analgesia (P=0.0006) and overall satisfaction (P=0.0031) differed significantly among the three groups (Table 4), with pairwise comparisons showing higher levels of satisfaction with analgesia and overall satisfaction in the TK group than in the C group, and higher levels of satisfaction with analgesia in the T group than in the C group (Supplementary Table 4).
Discussion
Although laparoscopic surgery can shorten recovery time, controlling pain caused by port-site wounds remains a challenge [10]. The use of bupivacaine as a TAP block after laparoscopic surgery was found to significantly reduce pain score at discharge [11,12]. The combination of TAP block and ketorolac 30 mg was associated with less variable dynamic pain than TAP block or ketorolac 30 mg alone in patients undergoing total abdominal hysterectomy, whereas pain at rest, opioid consumption, and rates of nausea, vomiting, and rescue antiemetics did not differ significantly among these three groups [13]. However, it was not clear whether TAP block with combined ketorolac and bupivacaine could provide additional benefits. The present study found that sufentanil consumption and actual and effective press times of the PCA pump were significantly lower in the TK and T groups than in the C group. Moreover, NRS pain scores at rest and during activity were lower in the TK than in the C group at various postoperative timepoints. Furthermore, satisfaction with analgesia dnd overall satisfaction were greater in the TK group. In contrast, the incidence of nausea and vomiting at various timepoints, along with venting time and duration of hospital stay, did not differ significantly in the three groups of patients.
This study found TAP blocks could reduce sufentanil consumption and actual and effective press times of the PCA pump, regardless of whether TK or T was used as TAP blocks. This finding could explained by the reduced vascular supply at the application site and the residual effects of local anesthetic in the neural sheaths [14,15]. Moreover, our finding, that the TK group achieved better pain control at rest and during activity than the T and C groups at 6 and 24 hours, suggests that analgesic effects were longer in patients administered ketorolac and bupivacaine for TAP block than in those administered bupivacaine alone and those without TAP blocks. Ketorolac inhibits the biosynthesis of prostaglandins, suggesting that the combination of ketorolac and opioids could reduce the consumption of opioids and provide greater analgesic effects. Ketorolac delivered at the subarachnoid level could act directly on the spine, blocking hyperalgesia caused by activation of spinal glutamate and substance P receptors, suggesting that delivery of ketorolac to a more proximal site of the nerve would have greater effect than its delivery to the nerve terminal [16–18].
Our study found that patients in the TK group experienced greater satisfaction with analgesia and greater overall satisfaction than patients without TAP blocks, and that patients in the T group achieved greater satisfaction with analgesia than patients in the C group, findings that may be due to significant improvements in the primary endpoints and pain control provided by TK. However, there were no differences among these groups in the incidence of nausea and vomiting at various timepoints, venting time, and duration of hospital stay. However, the incidence of nausea and vomiting in these groups was low, resulting in insufficient power to detect potential differences among groups [19–21]. In addition, the severity of pain gradually decreased in the three groups, reducing the differences among groups in venting times and duration of hospital stay.
This study has three main strengths. First, this study was designed as a randomized, observer-blinded, 3-armed clinical trial, with the baseline characteristics being generally balanced among the three patient groups, thereby eliminating potential selection and confounder biases. Second, this study was the first to use TK as a TAP block in patients who underwent gynecological laparoscopic surgery. Third, outcomes evaluating the effectiveness of treatment were thoroughly investigated. Fourth, pain scores and rates of nausea and vomiting were evaluated at various timepoints, enabling a determination of treatment effectiveness over time.
Nevertheless, this study also had several limitations. First, this was a single-center study, which may reduce the ability to generalize from our findings. Second, detailed medical histories and combinations of diseases and medications were not recorded, preventing investigation of the potential impact of these factors. Third analyses of primary efficacy endpoints were not stratified. Fourth, the statistical power of this study was insufficient to detect differences among groups of the incidence of nausea and vomiting.
Conclusions
Combined ketorolac and bupivacaine, or bupivacaine alone as a TAP block reduced the consumption of sufentanil and the actual and effective press times of PCA pumps when compared with ketorolac alone in women undergoing laparoscopic gynecologic surgery. Moreover, combined ketorolac and bupivacaine as a TAP block resulted in better pain control, especially with regard to prolonged analgesic effects, without increasing the incidence of nausea and vomiting. Future studies showed compare the effects of various TAP block strategies.
Tables
Table 1. Characteristics of the patients at baseline.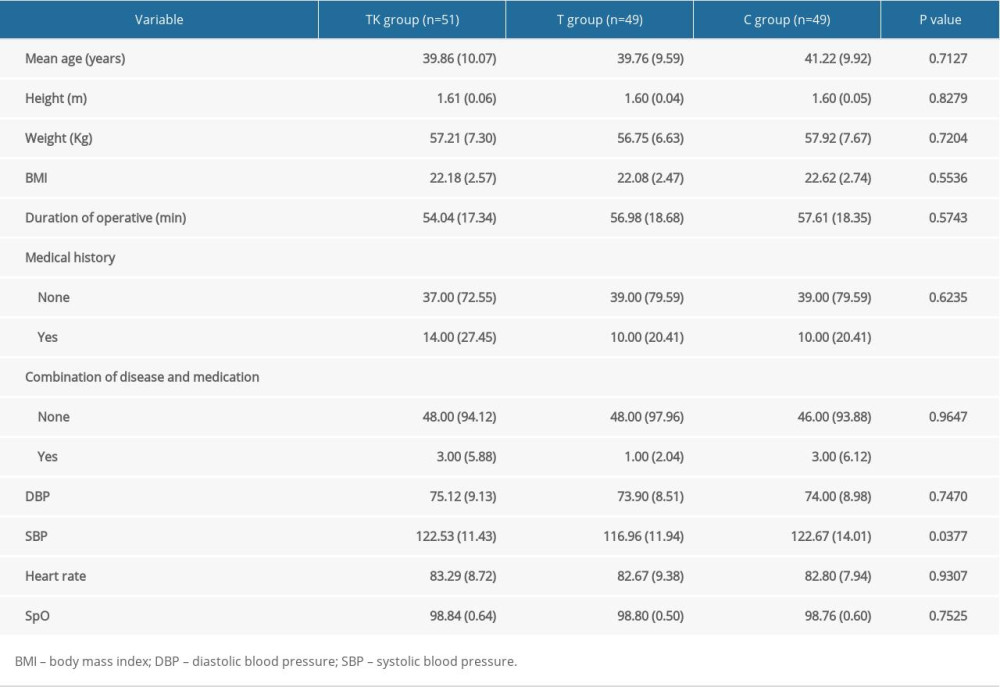 Table 2. Primary end points.
Table 2. Primary end points.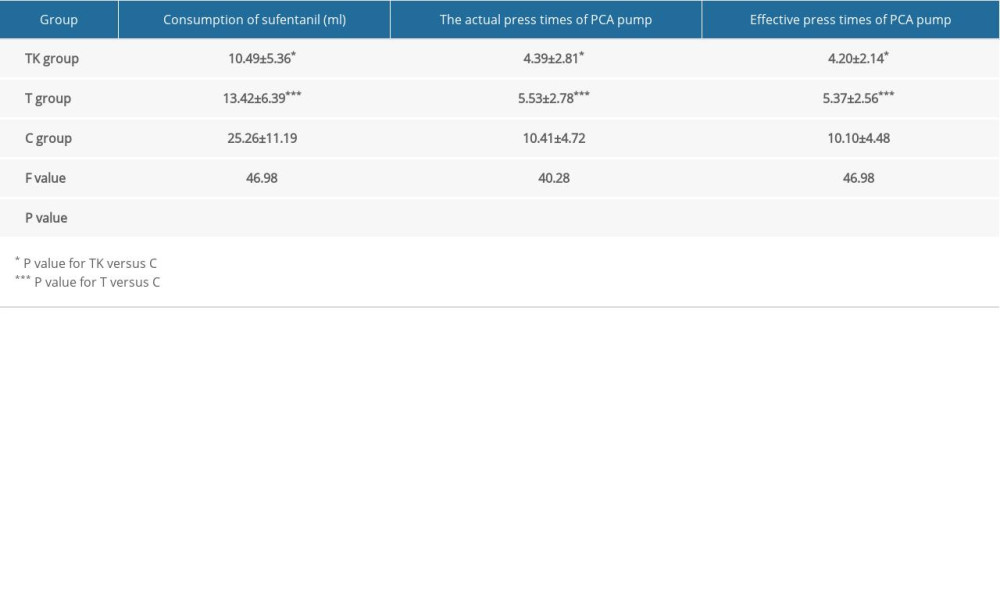 Table 3. NRS scores at rest and activity after surgery.
Table 3. NRS scores at rest and activity after surgery.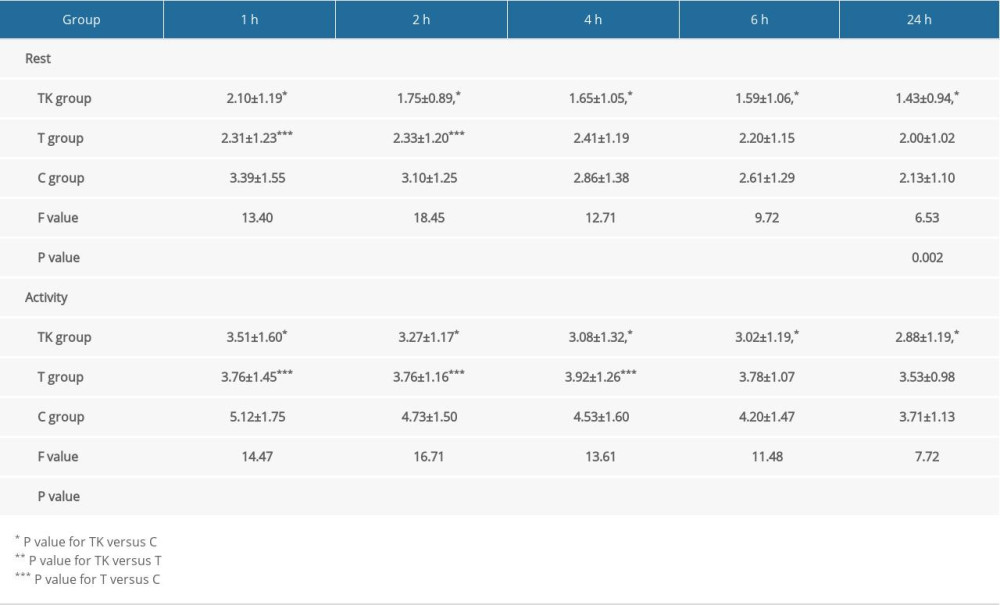 Table 4. Analgesic satisfaction and length of stay.
Table 4. Analgesic satisfaction and length of stay.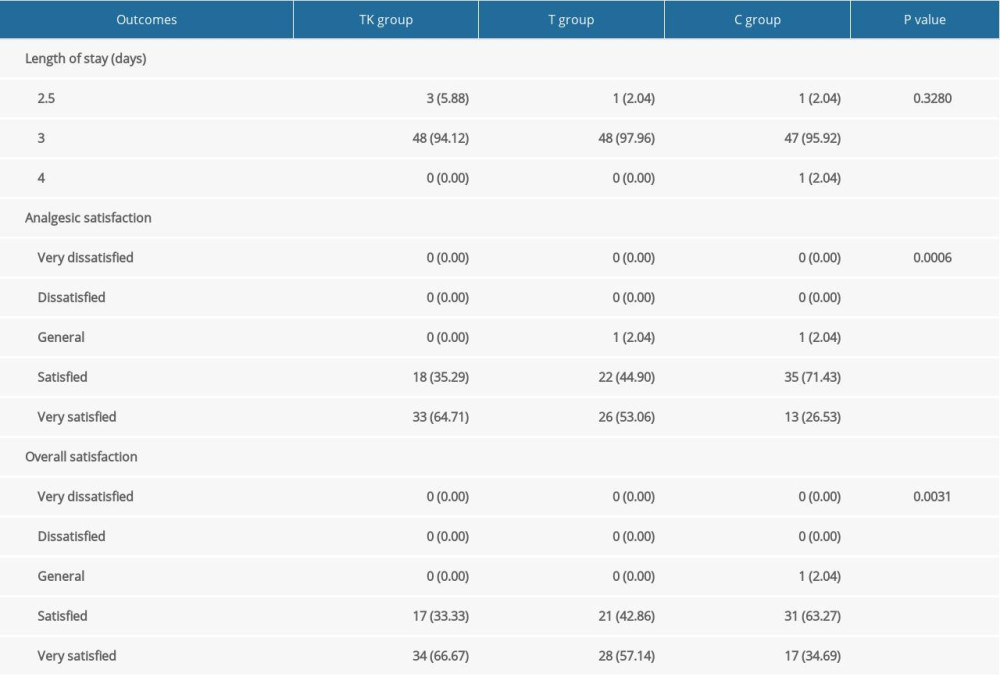 Supplemenatry Table 1. Details regarding the non-inclusion of recruited patients.
Supplemenatry Table 1. Details regarding the non-inclusion of recruited patients.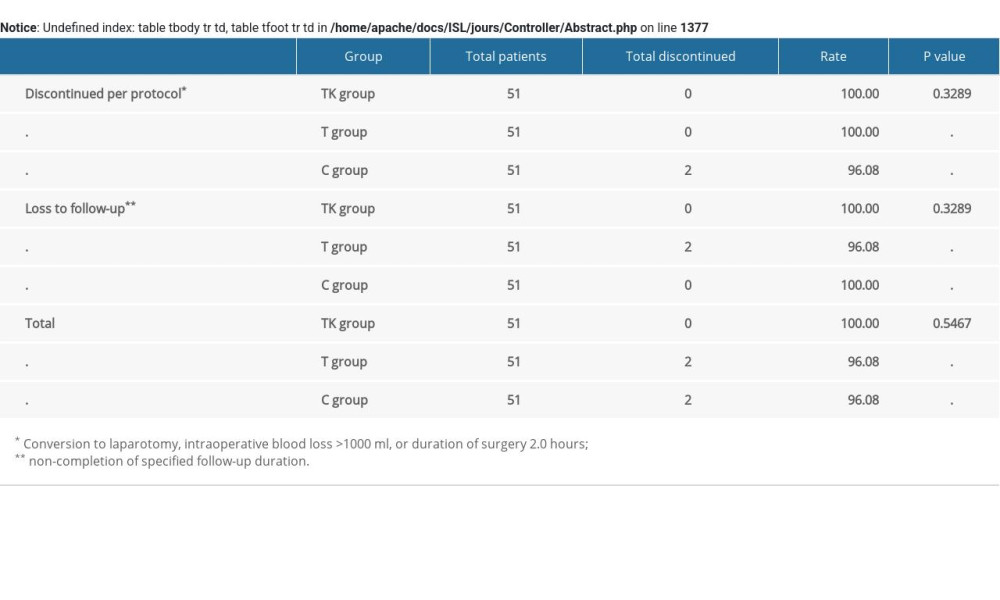 Supplemenatry Table 2. Incidence of nausea and vomiting over time among patients in the three groups.
Supplemenatry Table 2. Incidence of nausea and vomiting over time among patients in the three groups.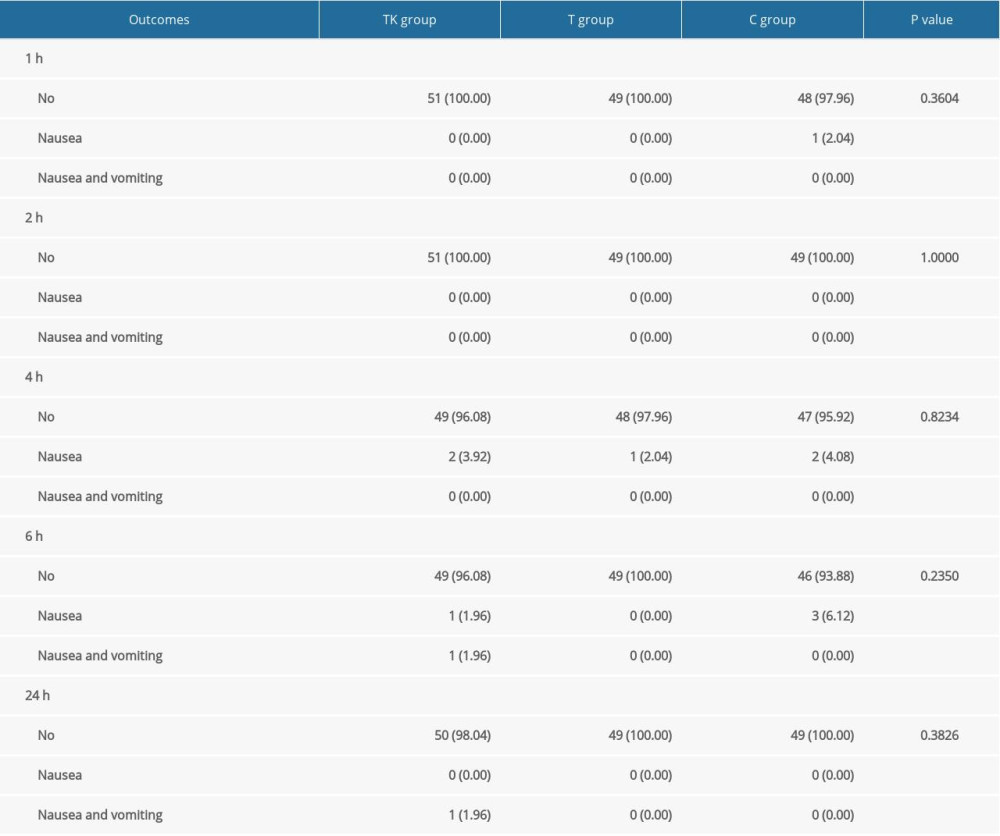 Supplemenatry Table 3. Mean venting time among patients in the three groups.
Supplemenatry Table 3. Mean venting time among patients in the three groups.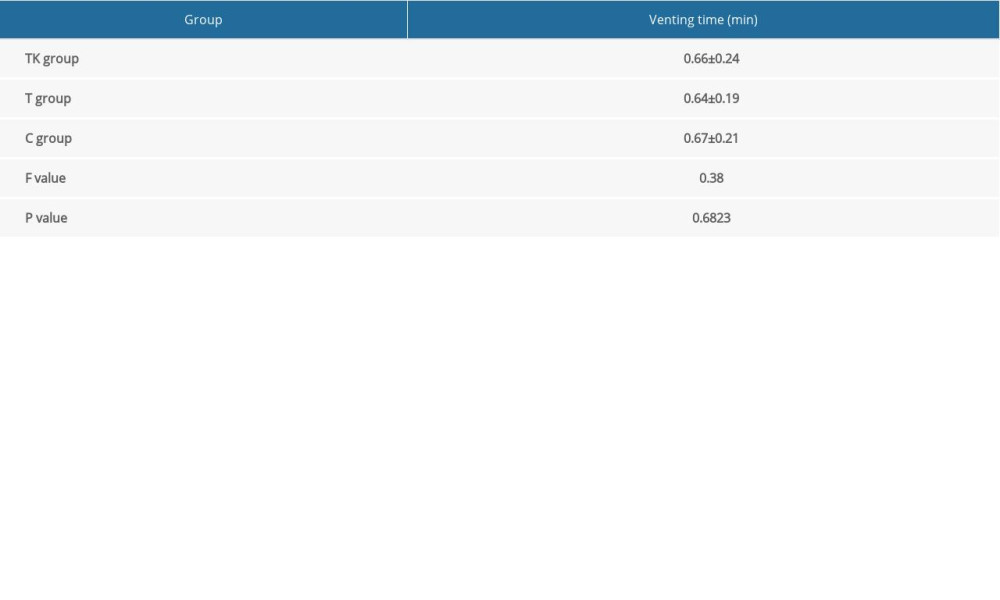 Supplemenatry Table 4. Results of pairwise comparisons of satisfaction with analgesia and overall satisfaction.
Supplemenatry Table 4. Results of pairwise comparisons of satisfaction with analgesia and overall satisfaction.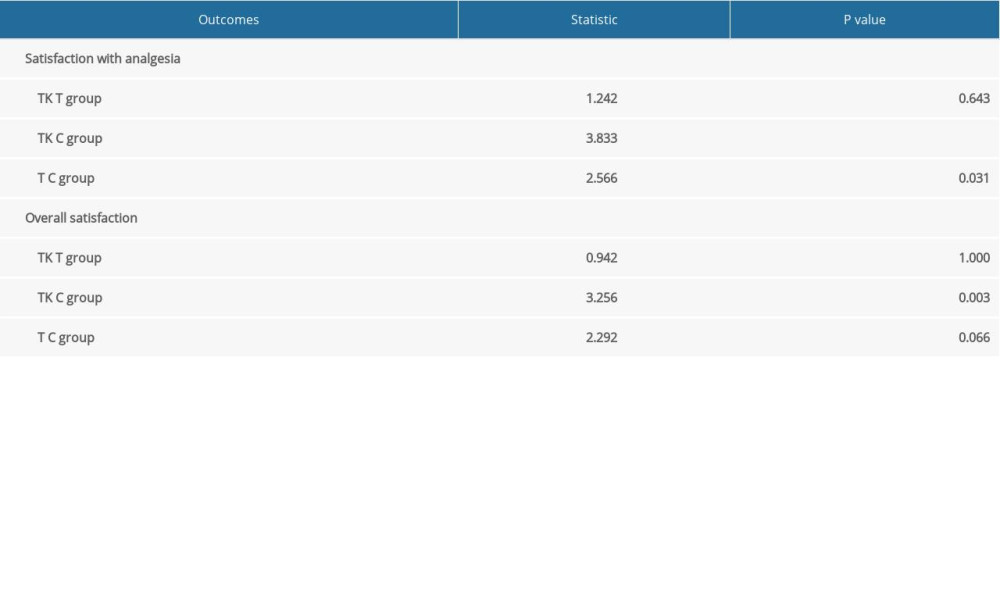
References
1. Dargent D, Martin X, Sacchetoni A, Mathevet P, Laparoscopic vaginal radical trachelectomy a treatment to preserve the fertility of cervical carcinoma patients: Cancer, 2000; 88; 1877-82
2. Dixon JB, Reuben Y, Halket C, O’Brien PE, Shoulder pain is a common problem following laparoscopic adjustable gastric band surgery: Obes Surg, 2005; 15; 1111-17
3. Jabbour-Khoury SI, Dabbous AS, Gerges FJ, Intraperitoneal and intravenous routes for pain relief in laparoscopic cholecystectomy: JSLS, 2005; 9; 316-21
4. McDonnell JG, O’Donnell BD, Tuite D, The regional abdominal field infiltration (R.A.F.I.) technique: Computerised tomographic and anatomical identification of a novel approach to the transversus abdominis neurovascular fascial plane: Anesthesiology, 2004; 101; A899 (abstract)
5. Hebbard P, Fujiwara Y, Shibata Y, Royse C, Ultrasound guided transversus abdominis plane (TAP) block: Anaesth Intensive Care, 2007; 35; 616-17
6. Yu N, Long X, Lujan-Hernandez JR, Transversus abdominis-plane block versus local anesthetic wound infiltration in lower abdominal surgery: A systematic review and meta-analysis of randomized controlled trials: BMC Anesthesiol, 2014; 14; 121
7. Hutchins J, Delaney D, Vogel RI, Ultrasound guided subcostal transversus abdominis plane (TAP) infiltration with liposomal bupivacaine for patients undergoing robotic assisted hysterectomy: A prospective randomized controlled study: Gynecol Oncol, 2015; 138; 609-13
8. Reinhart DJ, Stagg KS, Walker KG, Postoperative analgesia after peripheral nerve block for podiatric surgery: Clinical efficacy and chemical stability of lidocaine alone versus lidocaine plus ketorolac: Reg Anesth Pain Med, 2000; 25; 506-13
9. Lenz H, Sandvik L, Qvigstad E, A comparison of intravenous oxycodone and intravenous morphine in patient-controlled postoperative analgesia after laparoscopic hysterectomy: Anesth Analg, 2009; 109; 1279-83
10. Wu CL, Rowlingson AJ, Partin AW, Correlation of postoperative pain to quality of recovery in the immediate postoperative period: Reg Anesth Pain Med, 2005; 30; 516-22
11. Calle GA, López CC, Sánchez E, Transversus abdominis plane block after ambulatory total laparoscopic hysterectomy: Randomized controlled trial: Acta Obstet Gynecol Scand, 2014; 93; 345-50
12. De Oliveira GS, Fitzgerald PC, Marcus RJ, A dose-ranging study of the effect of transversus abdominis block on postoperative quality of recovery and analgesia after outpatient laparoscopy: Anesth Analg, 2011; 113; 1218-25
13. Gasanova I, Grant E, Way M, Ultrasound-guided transversus abdominal plane block with multimodal analgesia for pain management after total abdominal hysterectomy: Arch Gynecol Obstet, 2013; 288; 105-11
14. Johns N, O’Neill S, Ventham NT, Clinical effectiveness of transversus abdominis plane (TAP) block in abdominal surgery: A systematic review and meta-analysis: Colorectal Dis, 2012; 14; e635-42
15. Ma N, Duncan JK, Scarfe AJ, Clinical safety and effectiveness of transversus abdominis plane (TAP) block in post-operative analgesia: A systematic review and meta-analysis: J Anesth, 2017; 31; 432-52
16. Malmberg AB, Yaksh TL, Pharmacology of the spinal action of ketorolac, morphine, ST-91, U50488H, and L-PIA on the formalin test and isobolographic analysis of the NSAID interaction: Anesthesiology, 1993; 79; 270-81
17. Malmberg AB, Yaksh TL, Antinociceptive actions of spinal nonsteroidal anti-inflammatory agents on the formalin test in the rat: J Pharmacol Exp Ther, 1992; 263; 136-46
18. Jurna I, Brune K, Central effect of the non-steroidal anti-inflammatory agents indomethacin, ibuprofen, and diclofenac, determined in C fibre-evoked activity in single neurons of the rat thalamus: Pain, 1990; 41; 71-80
19. De Oliveira GS, Castro-Alves LJ, Ahmad S, Dexamethasone to prevent postoperative nausea and vomiting: An updated meta-analysis of randomized controlled trials: Anesth Analg, 2013; 116; 58-74
20. De Oliveira GS, Castro-Alves LJ, Chang R, Systemic metoclopramide to prevent postoperative nausea and vomiting: A meta-analysis without Fujii’s studies: Br J Anaesth, 2012; 109; 688-97
21. Habib AS, Gan TJ, Postoperative nausea and vomiting: Then & now: Anesth Analg, 2012; 115; 493-95
Tables
 Table 1. Characteristics of the patients at baseline.
Table 1. Characteristics of the patients at baseline. Table 2. Primary end points.
Table 2. Primary end points. Table 3. NRS scores at rest and activity after surgery.
Table 3. NRS scores at rest and activity after surgery. Table 4. Analgesic satisfaction and length of stay.
Table 4. Analgesic satisfaction and length of stay. Table 1. Characteristics of the patients at baseline.
Table 1. Characteristics of the patients at baseline. Table 2. Primary end points.
Table 2. Primary end points. Table 3. NRS scores at rest and activity after surgery.
Table 3. NRS scores at rest and activity after surgery. Table 4. Analgesic satisfaction and length of stay.
Table 4. Analgesic satisfaction and length of stay. Supplemenatry Table 1. Details regarding the non-inclusion of recruited patients.
Supplemenatry Table 1. Details regarding the non-inclusion of recruited patients. Supplemenatry Table 2. Incidence of nausea and vomiting over time among patients in the three groups.
Supplemenatry Table 2. Incidence of nausea and vomiting over time among patients in the three groups. Supplemenatry Table 3. Mean venting time among patients in the three groups.
Supplemenatry Table 3. Mean venting time among patients in the three groups. Supplemenatry Table 4. Results of pairwise comparisons of satisfaction with analgesia and overall satisfaction.
Supplemenatry Table 4. Results of pairwise comparisons of satisfaction with analgesia and overall satisfaction. In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952