26 June 2020: Clinical Research
Regulation of Keap-1/Nrf2 Signaling Pathway Is Activated by Oxidative Stress in Patients with Premature Rupture of Membranes
Wenfeng Zhang1ABCE, Meng Li1BC, Nana Li1BF, Zonghua Liu1AFG*DOI: 10.12659/MSM.921757
Med Sci Monit 2020; 26:e921757
Abstract
BACKGROUND: The potential mechanisms underlying premature rupture of membrane (PROM) is still unknown. The aim of this study was to determine the role of Keap-1/Nrf2 signaling pathway activation by oxidative stress in patients with preterm premature rupture of membranes.
MATERIAL AND METHODS: Placental tissues from preterm premature rupture of membranes (PPROM) (n=20), full-term premature rupture of membranes (FPROM) (n=20), and normal-term births (n=20) were collected and amniotic tissues were separated from the placental tissues from pregnant women at Shandong Provincial Qianfoshan Hospital. RT-PCR and Western blot were used to detect the levels of factors in the Keap-1/Nrf2 signaling pathway. To investigate the roles of Nrf2, we downregulated Nrf2 expression using siRNA in primary human amniotic epithelial (HAE) cells.
RESULTS: Among the control group, FPROM group, and PPROM group, the reactive oxygen species (ROS) levels were significantly increased in the FPROM and PPROM groups. The differences indicated higher levels of oxidative stress in amniotic tissues with FPROM and PPROM after downregulation of si-Nrf2 in HAE cells. Antioxidants were lower in amniotic tissues with the FPROM group and PPROM group than in the control group. The antioxidant enzymes catalase (CAT), glutathione (GSH), glutathione peroxidase (GSHPx), and superoxide dismutases (SOD1 and SOD2) were examined in amniotic tissues. We found that the ROS levels were significantly increased after downregulation of si-Nrf2 compared with the control group. We found that the expression of Heme Oxygenase-1 (HO-1) and Glycogen Synthase Kinase-3β (GSK-3β), which is critical in the Keap-1/Nrf2 signaling pathway, increased significantly after downregulation of si-Nrf2 in HAE cells.
CONCLUSIONS: We found that increased ROS levels and decreased antioxidant enzymes in the PPROM and FPROM patients compared with the control group.
Keywords: Fetal Membranes, Premature Rupture, Fetal Research, Amnion, Blotting, Western, Case-Control Studies, Extraembryonic Membranes, Gestational Age, glycogen synthase kinase 3 beta, Heme Oxygenase-1, Kelch-Like ECH-Associated Protein 1, NF-E2-Related Factor 2, Pregnancy, RNA, Messenger, RNA, Small Interfering, Real-Time Polymerase Chain Reaction, Reverse Transcriptase Polymerase Chain Reaction, superoxide dismutase-1, young adult
Background
Premature rupture of membranes (PROM) is associated with neonatal morbidity and mortality all over the world. It is divided into full-term premature rupture of membranes (FPROM) and preterm premature rupture of membranes (PPROM) [1,2]. PPROM frequently occurs and is closely associated with all perinatal deaths [3,4]. Previous reports showed that several factors interact between preterm birth (PTB) and PPROM, including high and low body mass indexes, behavioral factors, different modes of delivery, obstetrical complications, several environmental factors, and genetic predisposition [5–7].
Oxidative stress is a pathophysiologic mechanism related with PPROM and was observed in previous studies [8–10]. Excessive production of reactive nitrogen species (RNS) and reactive oxygen species (ROS) occurs by removing electrons from other molecules or adding oxygen as an oxidant to other molecules, thereby exceeding the degree of oxidation [11,12]. PPROM frequently occurs and is associated with endogenous ROS, which has an important role in maintaining homeostasis [13].
Transcription factor nuclear factor-erythroid 2 (NF-E2) p45-related factor 2 (Nrf2, gene name NFE2L2) regulates the expression of large gene networks encoding inducible cytoprotective proteins that allow mammalian cells and organisms to adapt and survive under conditions of oxidative stress. Nrf2, together with the major negative regulator Kelch-like ECH-related protein 1 (Keap1), forms a molecular effector and sensor system that can respond strongly to disturbances of cellular redox balance and orchestrate a comprehensive defense program to restore homeostasis [14,15]. Nrf2 status affects ROS production through mitochondria and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, which are 2 major ROS production systems. The proteins in the Nrf2 transcription target play a key role in the production and utilization of reducing equivalents such as NADPH and reduced glutathione (GSH), as well as thioredoxin, thioredoxin reductase, peroxidase, and thioredoxins, which together perform partitioned redox sensing of ROS to maintain redox balance under steady-state conditions [16,17].
Based on data on oxidative stress, we hypothesized that the anti-oxidative capacity of fetal-maternal tissues, oxidative stress-induced cell damage, and activated oxygen-activated signaling pathways indicate that PPROM and FPROM are clinically different. We also hypothesized that the critical effect of the Keap-1/Nrf2 signaling pathway is involved in the mechanism of PPROM and FPROM. We investigated the gene expression of factors in the Keap-1/Nrf2 signaling pathway at mRNA levels and protein levels by RT-PCR and Western blot. We also sought to identify the biochemical and histological markers in HAE cells and amniotic tissues of patients.
Material and Methods
PATIENTS AND STUDY DESIGN:
From December 1, 2014 to July 30, 2018, a case-control study was performed on pregnant women at 24 to 33+6 weeks of pregnancy, admitted to the Department of Obstetrics and Gynecology, The First Affiliated Hospital of Shandong First Medical University. In the case group, all individuals were women with premature rupture of membranes. After joining the group, PPROM and FPROM were divided into PPROM/FPROM at a ratio of 1/1. The dates of all eligible cases are obtained from the medical record system. To avoid data loss due to incomplete medical records, we conducted telephone interviews within 6 months after diagnosis. A control group matched for gestational age was selected from pregnant women of normal gestational age. For the case group, 52 patients were eligible. Complete information was collected from 40 patients. As a control, a total of 48 patients were eligible, including complete information on 40 patients. This study enrolled a total of 20 eligible PPROM and FPROM cases and 20 controls, with a patient/control ratio of 1/1. The Ethics Committee approval number was S326. The required sample size was calculated according to previous reports [18,19] using Power and Sample Size (PASS) v15 software. Sixty patients were eligible and were enrolled after calculation. Among them, 20 patients with PPROM, 20 patients with FPROM, and 20 patients with normal-term births were enrolled.
TISSUES COLLECTION AND AMNION TISSUES ISOLATION:
Placental tissues were derived from a normal delivery or artificially ruptured maternal membranes (38 weeks with no prior history of PTB or PPROM), FPRPOM (38 weeks), or PPROM. The diagnosis of rupture of membranes was established by a positive Nitrazine test result and/or positive pooling on speculum examination and oligohydramnios on ultrasound examination. Several aliquots of tissue were randomly collected from the maternal side of the placenta. We separated amnion tissues from human placental tissues and washed the blood stains. The amnion tissue was cut into pieces, and the first tissue digestive juice was added for digestion. We removed the first tissue digestive fluid after digestion, washed the amnion tissue and shredded it, added a second tissue digestive fluid for digestion, and added trypsin solution for digestion.
OXIDATION OF DCFH:
A 100-μL aliquot of DCFH (40 μmolL-1) was added to 100 μL PBS containing Cu2+ or Cu+, and the development of fluorescence was monitored in an automatic plate reader at 37°C. The excitation wavelength was set at 485 nm and the emission spectrum was recorded at 535 nm. At the specified timepoints, 20 μL of copper chelator or PBS (for control) was added to obtain the final concentration.
RNA EXTRACTION AND REAL-TIME REVERSE TRANSCRIPTASE-PCR:
Total RNA of tissue samples was isolated using TRIzol (Life Technologies, Inc., Rockville, MD) according to the manufacturer’s instructions. cDNA was generated from 1 ug of each RNA sample and a reverse-transcribed using a transcription kit (Takara, Kyoto, Japan). Real-time quantitative reverse transcriptase-PCR (RT-PCR) was done in the 7300 Real-Time PCR System (Applied Biosystems).
WESTERN BLOT ANALYSIS:
Denatured protein samples were resolved on SDS-PAGE and transferred to PVDF membranes (Millipore, Billerica, MA. After blocking with non-fat milk, membranes were incubated overnight at 4°C with Keap-1, Nrf2, HO-1, GSK-3β, and GAPDH antibodies (Abcam, USA) followed by incubation with anti-rabbit HRP-conjugated secondary antibodies (Santa Cruz, Billerica, MA). Chemiluminescence detection was performed using ECL Advance Western blotting detection reagents (GE Healthcare, Little Chalfont, Buckinghamshire, UK). The relative expression was quantified using ImageJ software.
STATISTICAL ANALYSIS:
The chi-square test or Fisher’s exact test were used to compare qualitative variables, while continuous variables were compared using the
Results
PARTICIPANTS AND PREGNANCY OUTCOMES:
The general characteristics of all 60 patients are shown in Table 1. We assessed 60 tissue samples in the control, FPROM, and PPROM groups. Maternal age, gestational age, marital status, mode of delivery, and BMI were recorded. Gestational ages were significantly different among the 3 groups but were similar between the control group and FPROM group.
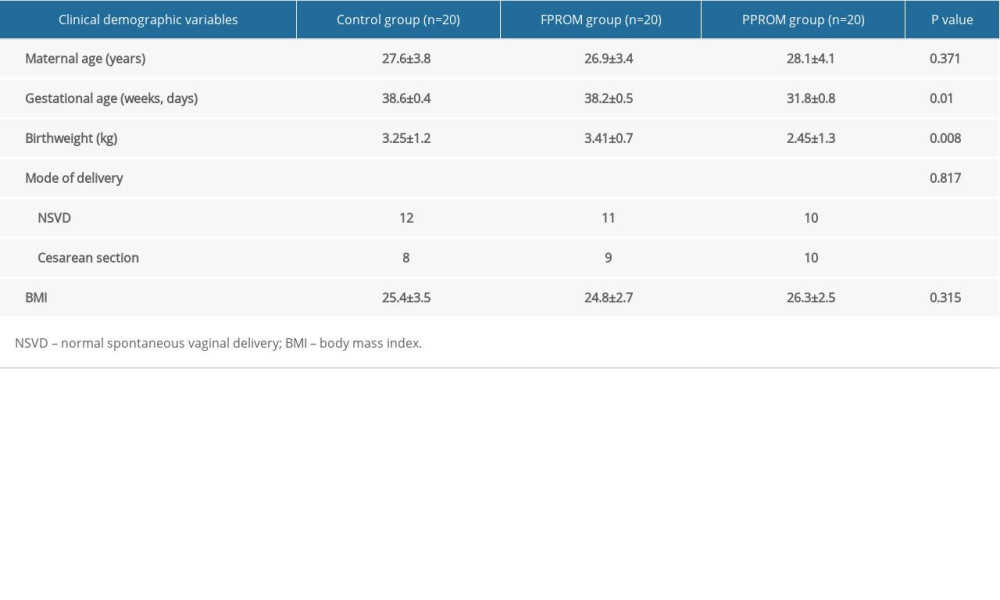
COMPARISON OF ROS LEVELS AMONG DIFFERENT GROUPS:
ROS levels were detected by dichlorofluorescein (DCFH). Compared with the control group, the ROS levels were significantly enhanced in FPROM and PPROM (P<0.01, Figure 1). The differences indicated that higher levels of oxidative stress in amniotic tissues with FPROM and PPROM.
COMPARISON OF ANTIOXIDANT ENZYMES LEVELS AMONG DIFFERENT GROUPS:
The FPROM and PPROM membranes had lower antioxidants than the control group. The membranes were checked for the antioxidant enzymes listed below. CAT in the control group was significantly higher than in the FPROM and PPROM groups (Figure 2A). GSH and GSH-Px were significantly higher in the control group than in the FPROM and PPROM groups (Figure 2B, 2C), and GSH and GSH-Px were significantly higher in the FPROM group than in the PPROM group. SOD1 and SOD2 were significantly higher in the control group than in the FPROM and PPROM groups (Figure 2D, 2E), and SOD1 and SOD2 were significantly higher in the FPROM group than in the PPROM group. PRX was significantly higher in the control group than in the FPROM and PPROM groups (Figure 2F). There were no significant differences in TRX between the FPROM and PPROM groups.
EXPRESSION OF KEAP-1 AND NRF 2 IN HUMAN AMNIOTIC TISSUES:
To further confirm whether the Keap-1/Nrf2 was associated with PPROM mediated by oxidative stress, we assessed changes in levels of Keap-1 and Nrf2 at mRNA levels and protein levels. Real-time PCR and Western blot analysis were used to examine the expression of Keap-1 and Nrf2 in different groups. As shown in Figure 2G and 2I, the expression of Keap-1 was significantly upregulated the control group compared with the PPROM and FPROM groups. Conversely, the expression of Nrf2 were significantly downregulated in the control group compared with the PPROM and FPROM groups (Figure 3A–3C).
KNOCKDOWN OF EXPRESSION OF NRF2 IS INVOLVED IN KEAP-1/NRF2 SIGNALING PATHWAY IN HAE CELLS:
To examine the effect of Nrf2 on regulation the oxidative stress and the targeted molecular factors, we knocked down the expression of Nrf2 HAE cells with RNAi. We found that the ROS levels were significantly increased after si-Nrf2 knockdown compared with the control group, as shown in Figure 3D and 3E. Moreover, knockdown of the expression of si-Nrf2 significantly decreased the expression of HO-1 and GSK-3β, but increased the expression of Keap-1 at mRNA and protein levels (Figure 4).
Discussion
The purpose of this study was to understand the changes/damages of membrane cells associated with oxidative stress, leading to different molecular signaling pathways that can be differentiated into PTB and PPROM as clinical phenotypes. The exact biological signals and mechanisms of premature membrane rupture (PPROM) are still poorly understood [20]. There are likely multiple and redundant pathways that converge on the phenotype of preterm and term parturition. The core hypothesis is the oxidative stress induces a range of protective proteins to alleviate damage to cells [8,21]. Endogenous conditions and some exogenous factors may cause oxidative stress, which in part leads to the promotion of the above regulations and gene activation [22]. The degree of oxide removal and the imbalance between the oxidation system and the antioxidant system result in tissue damage [9]. Studies measuring ROS or RNS or measuring oxidative stress by-products (i.e., biomarkers) generally report higher levels in PTB specimens compared to TB specimens, while studies measuring antioxidants report that they are lower in PTB specimens than in TB specimens [23,24]. Findings suggest that an imbalance in oxidants and antioxidants exists in PTB; thus, a dysregulated redox environment may be associated with the pathophysiology of PTB [25]. Although some studies support the relationship between oxidative stress and PTB, other studies are inconsistent and contradictory. The various measures used further limit the interpretation of the potential of PTB-associated oxidative stress mechanisms.
In the first phase of oxidative stress, Nrf2 is activated by dissociation from a repressor protein in the cytoplasm of Keap-1 cells, which contains cysteine residues. Keap-1 reacts with oxidizing and electrophilic groups, resulting in a conformational change and release of Nrf2 [26,27]. Subsequently, Nrf2 is transported to the nucleus and combines with the antioxidant response element (ARE), resulting in the transcription of defense genes [28]. The activation of transcription involves Nrf2 recognizing its own promoter and establishing effective interaction with it, and newly formed and accumulated Nrf2 in the nucleus binds to promoters of other specific genes [29].
Confounding factors seriously interfered with our estimation of risk, so we must find ways to control these variables. However, because the confounding factors we need to consider are not measurable, to be able to analyze the real results we can use surrogate confounders to control the confounding factors. In the present study, we found the antioxidant enzymes CAT, GSH, GSH-Px, SOD1, SOD2, and TRX were significantly higher in the control group than in the FPROM and PPROM groups, which suggests the activation of oxidative stress. These factors could be used as surrogate confounders to control the confounding bias. The Chinese population cohort selected in the present study minimized confounding factors introduced by interventions. Moreover, in patient selection, the demographics and clinical characteristics of patients in the control group, FPROM group, and PPROM group were similar and the potential selection bias was controlled.
This study also has some limitations. Retrospective sampling after delivery is the only way to obtain tissue in the uterus; although some of the indicators of oxidative stress we tested were specific, they were not enough to confirm Keap-1/Nrf2 as a pathway involved in PPROM. Prospective studies of sample sets with better clinical and phenotypic characteristics are needed.
Conclusions
We found that increased ROS levels and decreased antioxidant enzymes in the PPROM and FPROM patients compared with the control group. The levels of several antioxidant enzymes in the FPROM group and PPROM group also were significantly different. In an
Figures
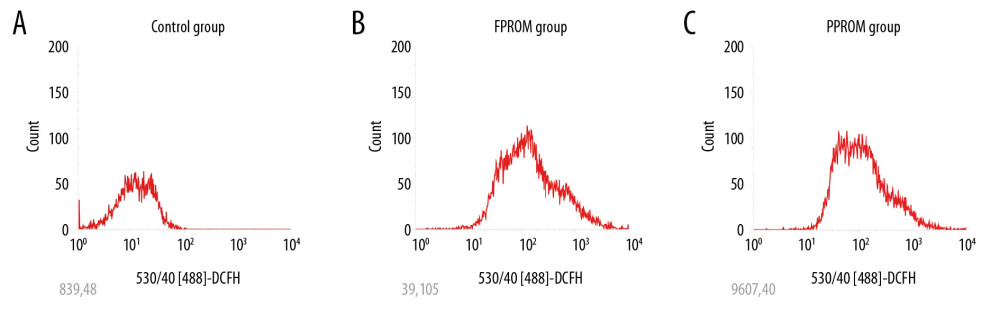 Figure 1. The ROS levels were detected and compared in the control group (A), FPROM group (B), and PPROM group (C) (P<0.01).
Figure 1. The ROS levels were detected and compared in the control group (A), FPROM group (B), and PPROM group (C) (P<0.01). 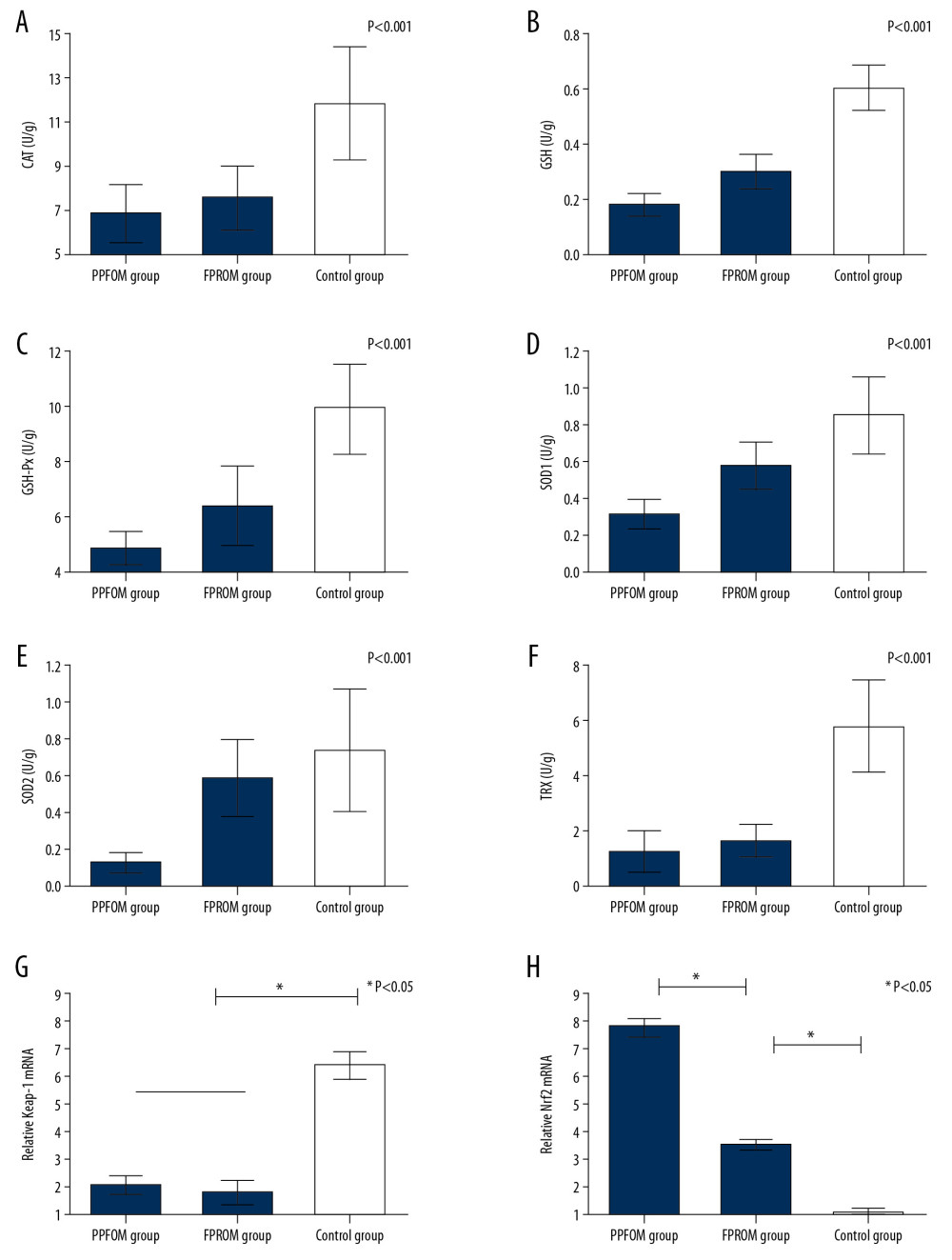 Figure 2. (A–F) Antioxidants were detected among the control group, FPROM, and PPROM group; (G) Keap-1 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05); (H) Nrf2 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05).
Figure 2. (A–F) Antioxidants were detected among the control group, FPROM, and PPROM group; (G) Keap-1 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05); (H) Nrf2 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05). 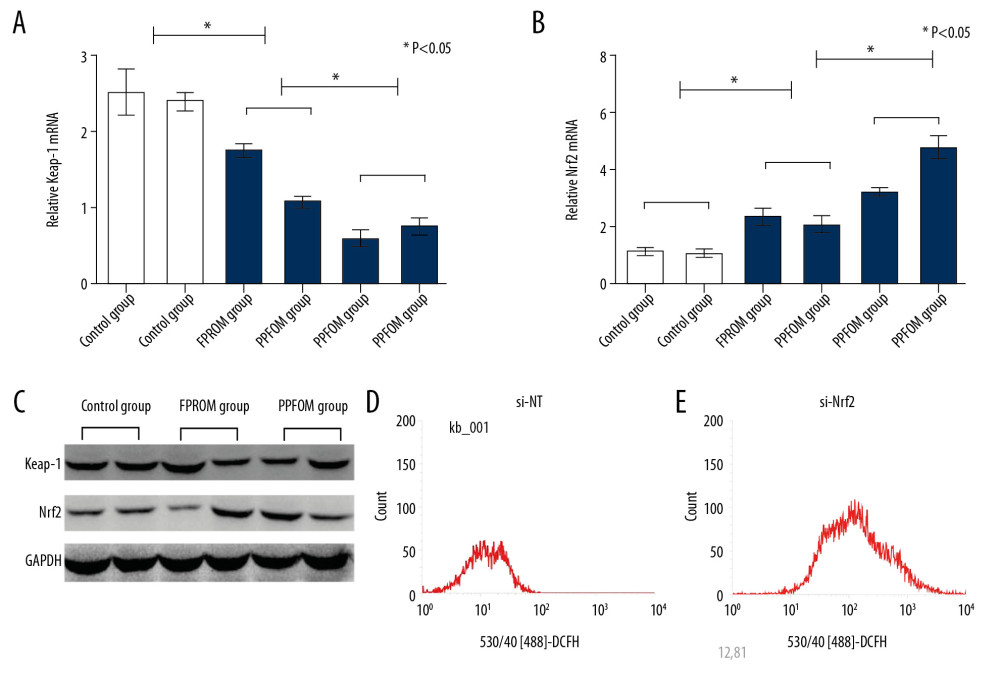 Figure 3. (A–C) Expressions of Keap-1 and Nrf2 were significantly different in the control group, FPROM group, and PPROM group as assessed by Western blot (P<0.05); The ROS levels were detected and compared in the control group (D) and si-Nrf2 group (E) (P<0.01).
Figure 3. (A–C) Expressions of Keap-1 and Nrf2 were significantly different in the control group, FPROM group, and PPROM group as assessed by Western blot (P<0.05); The ROS levels were detected and compared in the control group (D) and si-Nrf2 group (E) (P<0.01). 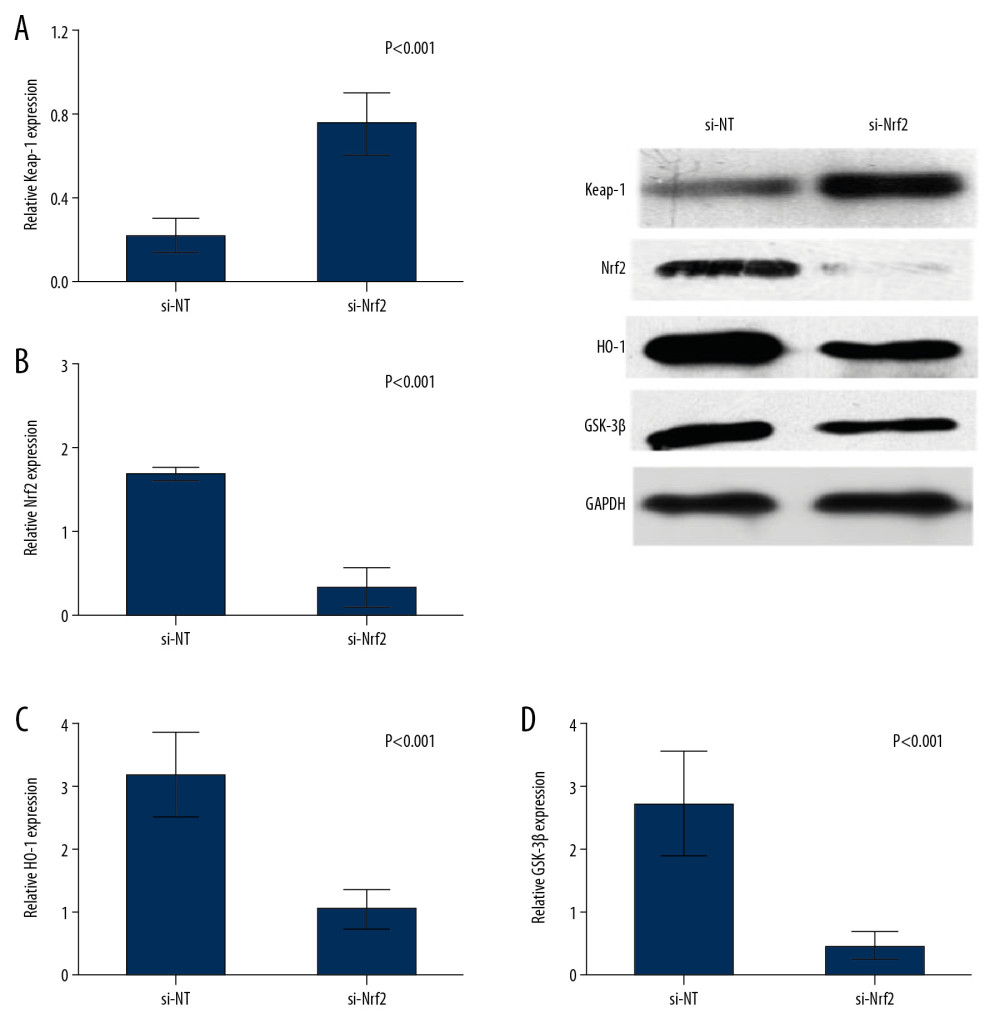 Figure 4. Knockdown of the expression of si-Nrf2 significantly increased the expression of Keap-1 (A) and decreased the expression of Nrf2 (B), HO-1 (C), and GSK-3β (D) at both mRNA and protein levels.
Figure 4. Knockdown of the expression of si-Nrf2 significantly increased the expression of Keap-1 (A) and decreased the expression of Nrf2 (B), HO-1 (C), and GSK-3β (D) at both mRNA and protein levels. References
1. Schmitz T, Sentilhes L, Lorthe E, Preterm premature rupture of the membranes: Guidelines for clinical practice from the French College of Gynaecologists and Obstetricians (CNGOF): Eur J Obstet Gynecol Reprod Biol, 2019; 236; 1-6
2. Weiner E, Barrett J, Zaltz A, Amniotic fluid volume at presentation with early preterm premature rupture of the membranes and the association with severe neonatal respiratory morbidity: Ultrasound Obstet Gynecol, 2019; 54(6); 767-73
3. Dagklis T, Petousis S, Margioula-Siarkou C, Parameters affecting latency period in PPROM cases: A 10-year experience of a single institution: J Matern Fetal Neonatal Med, 2013; 26; 1455-58
4. Polettini J, Dutta EH, Behnia F, Aging of intrauterine tissues in spontaneous preterm birth and preterm premature rupture of the membranes: A systematic review of the literature: Placenta, 2015; 36; 969-73
5. Auger N, Le TU, Park AL, Luo ZC, Association between maternal comorbidity and preterm birth by severity and clinical subtype: Retrospective cohort study: BMC Pregnancy Childbirth, 2011; 11; 67
6. Lynch AM, Hart JE, Agwu OC, Association of extremes of prepregnancy BMI with the clinical presentations of preterm birth: Am J Obstet Gynecol, 2014; 210; 428.e1-9
7. Shree R, Caughey AB, Chandrasekaran S, Short interpregnancy interval increases the risk of preterm premature rupture of membranes and early delivery: J Matern Fetal Neonatal Med, 2018; 31(22); 3014-20
8. Menon R, Oxidative stress damage as a detrimental factor in preterm birth pathology: Front Immunol, 2014; 5; 567
9. Dutta EH, Behnia F, Boldogh I, Oxidative stress damage-associated molecular signaling pathways differentiate spontaneous preterm birth and preterm premature rupture of the membranes: Mol Hum Reprod, 2016; 22; 143-57
10. Martin LF, Moco NP, de Lima MD, Histologic chorioamnionitis does not modulate the oxidative stress and antioxidant status in pregnancies complicated by spontaneous preterm delivery: BMC Pregnancy Childbirth, 2017; 17; 376
11. Biswal S, Rizwan H, Pal S, Oxidative stress, antioxidant capacity, biomolecule damage, and inflammation symptoms of sickle cell disease in children: Hematology, 2019; 24; 1-9
12. Zou D, Li J, Fan Q, Reactive oxygen and nitrogen species induce cell apoptosis via a mitochondria-dependent pathway in hyperoxia lung injury: J Cell Biochem, 2019; 120; 4837-50
13. Longini M, Perrone S, Vezzosi P, Association between oxidative stress in pregnancy and preterm premature rupture of membranes: Clin Biochem, 2007; 40; 793-97
14. Muller SG, Jardim NS, Quines CB, Nogueira CW, Diphenyl diselenide regulates Nrf2/Keap-1 signaling pathway and counteracts hepatic oxidative stress induced by bisphenol A in male mice: Environ Res, 2018; 164; 280-87
15. Wang J, Jiang C, Zhang K, Melatonin receptor activation provides cerebral protection after traumatic brain injury by mitigating oxidative stress and inflammation via the Nrf2 signaling pathway: Free Radic Biol Med, 2019; 131; 345-55
16. Ci X, Lv H, Wang L, The antioxidative potential of farrerol occurs via the activation of Nrf2 mediated HO-1 signaling in RAW 264.7 cells: Chemico Biol Interact, 2015; 239; 192-99
17. Zhu C, Dong Y, Liu H: Biomed Pharmacother, 2017; 88; 124-33
18. Rodriguez Del Aguila M, Gonzalez-Ramirez A, Sample size calculation: Allergol Immunopathol (Madr), 2014; 42; 485-92
19. Rohrig B, du Prel JB, Wachtlin D, Sample size calculation in clinical trials: part 13 of a series on evaluation of scientific publications: Dtsch Arztebl Int, 2010; 107(31–32); 552-56
20. Makieva S, Dubicke A, Rinaldi SF, The preterm cervix reveals a transcriptomic signature in the presence of premature prelabor rupture of membranes: Am J Obstet Gynecol, 2017; 216; 602.e1-602.21
21. Pasquier JC, Doret MFetal membranes: embryological development, structure and the physiopathology of the preterm premature rupture of membranes: J Gynecol Obstet Biol Reprod (Paris), 2008; 37(6); 579-88 [in French]
22. El Kamouni S, El Kebbaj R, Andreoletti P, Protective effect of argan and olive oils against LPS-induced oxidative stress and inflammation in mice livers: Int J Mol Sci, 2017; 18 pii: E2181
23. Polettini J, Silva MG, Kacerovsky M, Expression profiles of fetal membrane nicotinamide adenine dinucleotide phosphate oxidases (NOX) 2 and 3 differentiates spontaneous preterm birth and pPROM pathophysiologies: Placenta, 2014; 35; 188-94
24. Hadi T, Bardou M, Mace G, Glutathione prevents preterm parturition and fetal death by targeting macrophage-induced reactive oxygen species production in the myometrium: FASEB J, 2015; 29; 2653-66
25. Yang X, Dong WB, Lei XP: J Matern Fetal Neonatal Med, 2018; 31; 1142-50
26. Yu H, Zhang J, Ji Q, Melatonin alleviates aluminium chloride-induced immunotoxicity by inhibiting oxidative stress and apoptosis associated with the activation of Nrf2 signaling pathway: Ecotoxicol Environ Saf, 2019; 173; 131-41
27. Zhang F, Munoz FM, Sun L, Cell-specific regulation of Nrf2 during ROS-Dependent cell death caused by 2,3,5-tris(glutathion-S-yl)hydroquinone (TGHQ): Chem Biol Interact, 2019; 302; 1-10
28. Lu MC, Ji JA, Jiang ZY, You QD, The Keap1-Nrf2-ARE pathway as a potential preventive and therapeutic target: An update: Med Res Rev, 2016; 36; 924-63
29. Fan J, Lv H, Li J, Roles of Nrf2/HO-1 and HIF-1alpha/VEGF in lung tissue injury and repair following cerebral ischemia/reperfusion injury: J Cell Physiol, 2019; 234; 7695-707
Figures
 Figure 1. The ROS levels were detected and compared in the control group (A), FPROM group (B), and PPROM group (C) (P<0.01).
Figure 1. The ROS levels were detected and compared in the control group (A), FPROM group (B), and PPROM group (C) (P<0.01). Figure 2. (A–F) Antioxidants were detected among the control group, FPROM, and PPROM group; (G) Keap-1 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05); (H) Nrf2 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05).
Figure 2. (A–F) Antioxidants were detected among the control group, FPROM, and PPROM group; (G) Keap-1 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05); (H) Nrf2 mRNA levels were significantly different in the control group, FPROM group, and PPROM group (P<0.05). Figure 3. (A–C) Expressions of Keap-1 and Nrf2 were significantly different in the control group, FPROM group, and PPROM group as assessed by Western blot (P<0.05); The ROS levels were detected and compared in the control group (D) and si-Nrf2 group (E) (P<0.01).
Figure 3. (A–C) Expressions of Keap-1 and Nrf2 were significantly different in the control group, FPROM group, and PPROM group as assessed by Western blot (P<0.05); The ROS levels were detected and compared in the control group (D) and si-Nrf2 group (E) (P<0.01). Figure 4. Knockdown of the expression of si-Nrf2 significantly increased the expression of Keap-1 (A) and decreased the expression of Nrf2 (B), HO-1 (C), and GSK-3β (D) at both mRNA and protein levels.
Figure 4. Knockdown of the expression of si-Nrf2 significantly increased the expression of Keap-1 (A) and decreased the expression of Nrf2 (B), HO-1 (C), and GSK-3β (D) at both mRNA and protein levels. In Press
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
11 Mar 2024 : Clinical Research
Comparison of Effects of Sugammadex and Neostigmine on Postoperative Neuromuscular Blockade Recovery in Pat...Med Sci Monit In Press; DOI: 10.12659/MSM.942773
12 Mar 2024 : Clinical Research
Comparing Neuromuscular Blockade Measurement Between Upper Arm (TOF Cuff®) and Eyelid (TOF Scan®) Using Miv...Med Sci Monit In Press; DOI: 10.12659/MSM.943630
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952