13 July 2020: Laboratory Research
Effects of Adenovirus-Mediated Overexpression of on Chronic Inflammation: An and Study
Fanping Meng1ABCEFG, Po Hao2ABCEFG*, Hongxin Du1C, Zheng Zhou1DDOI: 10.12659/MSMBR.924124
Med Sci Monit Basic Res 2020; 26:e924124
Abstract
BACKGROUND: Insulin sensitivity and inflammation can be affected by juxtaposition with another zinc finger gene 1 (JAZF1), but its precise role in chronic inflammation is unclear. In this study, JAZF1-overexpression adenovirus plasmids were transfected into macrophages, CD4⁺ T cells, and C57BL/6J mice to assess the role of JAZF1 in chronic inflammation.
MATERIAL AND METHODS: JAZF1 was cloned into an adenovirus skeleton plasmid and transfected in HEK293 cells to package and enrich the virus particles. In vitro, the JAZF1 overexpression adenovirus vector (PAD-JAZF1) was cultured with peritoneal macrophages and peripheral blood CD4⁺ T cells of C57BL/6J mice, and samples were evaluated using flow cytometry. In vivo, PAD-JAZF1 was introduced into C57BL/6J mice, and livers were collected to evaluate factors related to inflammation by hematoxylin & eosin and immunohistochemical staining.
RESULTS: In vitro, PAD-JAZF1 decreased total macrophages, CD11c⁺ macrophages, and the secretion of proinflammatory cytokines, but increased CD206⁺ macrophages. It also decreased total CD4⁺T cells, active T cells, memory T cells, and the secretion of IL-6, IL-10, and IFN-γ, but increased Treg cells and restrictive T cells. In vivo, compared to those in the control group transfected with the adenovirus skeleton vector, mice transfected with the PAD-JAZF1 recombinant adenovirus had fewer CD11c⁺ ATMs and CD4⁺ T cells, lower levels of TNF-α and IL-6, and higher IL-10 concentrations in the liver.
CONCLUSIONS: These findings indicate that JAZF1 limits chronic inflammation by reducing macrophage and CD4⁺T cell populations, altering subtype differentiation, and regulating the secretion of immune-related factors.
Keywords: Adenoviridae, Genes, vif, Macrophages, Natural Killer T-Cells, CD11 Antigens, CD4-Positive T-Lymphocytes, Chronic Disease, Co-Repressor Proteins, Cytopathogenic Effect, Viral, Genetic Vectors, HEK293 Cells, Inflammation Mediators, Macrophages, Peritoneal, Organ Specificity, RNA, Messenger, Recombination, Genetic, Restriction Mapping
Background
The potential roles of insulin resistance and chronic inflammation in the pathogenesis of type 2 diabetes (T2D) have already demonstrated [1]. Inflammatory factors affect adipose tissues, oxidative stress, and the immune system, and then cause insulin resistance and β cell structure and function disorders, which play an important role in the progression of T2D. Preclinical evidence suggests that pancreatic β cell dysfunction is associated with the systemic inflammatory state, including C-reactive protein and its upstream regulator interleukin-6 (IL-6) [2]. A study conducted by Redwine et al. found that C-reactive protein and IL-6 are significantly correlated with both insulin sensitivity and β cell function [3]. Further, inflammation activates the internal insulin immune response. Chronic local inflammation of visceral adipose tissue (VAT) and subcutaneous adipose tissue (SAT) can result in the simultaneous secretion of a variety of fat-specific inflammatory factors involved in glycolipid metabolism [4]. Adipose tissue contains numerous immune cells, including T cells, B cells, and macrophages. CD4+ T cells are involved in the initial stage of adipose tissue inflammation during the progression of impaired glucose tolerance and insulin resistance. This suggests that in adipose tissues of patients with T2D, CD4+ T cells play important roles in the onset and maintenance of chronic inflammation [5].
Our preliminary studies showed that
Material and Methods
ETHICS STATEMENT:
All animal experiments were conducted according to the guidelines of the Ethics Committee of the Army Military Medical University.
FLOW CYTOMETRY AND REAGENTS:
For the
CONSTRUCTION OF THE ADENOVIRUS SHUTTLE PLASMID:
A plasmid with JAZF1, pIRES2-JAZF1, was provided by our task group. The recombinant adenovirus used in this study was prepared using the AdEasy-1XL Adenoviral Vector System (Stratagene, La Jolla, CA, USA). JAZF1 was digested with XhoI and EcoRI from the plasmid pIRES2-JAZF1, ligated with Pshuttle-CMV, amplified in DH-5a, selected, purified using the Plasmid Maxiprep Kit (OMEGA, Irving, TX, USA), and identified by XhoI and EcoRI digestion and DNA sequencing; the resulting plasmid was named Pshuttle-JAZF1.
HOMOLOGOUS RECOMBINATION OF THE ADENOVIRUS SKELETON PLASMID (PADEASY) AND PSHUTTLE-JAZF1:
A shuttle plasmid carrying the target gene fragment
PROPAGATION, PURIFICATION, TITER DETERMINATION, AND IDENTIFICATION OF THE PAD-JAZF1 ADENOVIRUS:
The recombinant adenovirus was propagated in human embryonic kidney 293 cells (HEK293) cultured in DMEM supplemented with 10% fetal bovine serum, 100 units/mL penicillin, and 100 mg/mL streptomycin at 37°C with 5% CO2. Twenty-four hours before transfection, 5 × 105 cells were seeded on a 6-well plate until 80% confluence was reached. The recombinant adenovirus pAD-JAZF1 from correct clones was linearized with PacI and transfected into 293 packaging cells using liposomes (Hanbio). Due to the loss of the early gene E1 in the adenovirus vector genome, 293 cells with E1 were used as packaging cells. After the transfected cells were incubated continuously for 5–7 days, the cytopathic effect (CPE) was observed. Then, viruses were harvested and purified on CsCl gradients and titers were determined. They were subsequently stored at −80°C in 4% sucrose buffer.
DETERMINATION OF RECOMBINANT ADENOVIRUS TITRATION:
The titer of recombinant adenoviral plasmids was measured by a plaque formation assay. AD293 cells were seeded (1×106 cells per well) in 6-well plates until they reached 50–70% confluence, which was followed by the addition of serial dilutions of viral samples. The cells were incubated in a 5% CO2 incubator at 37°C for 10 days. Cell monolayers were fixed with 25% formaldehyde. Then, plaques were counted by staining with neutral red according to the following formula: number of plaques/dilution coefficient×volume of the viral solution.
PREPARATION OF C57BL/6J MOUSE PERITONEAL MACROPHAGES:
Wild-type C57BL/6J mice at 8 weeks of age (22–28 g, normal chow diet) were used. DMEM with 5 mL of 75% serum was injected into the mouse abdomens. After 30 min, the liquid was recovered. Then, the mice were euthanized by cervical dislocation, placed in 75% alcohol for 3 min, injected with 5 mL of pre-cooled serum-free DMEM into the abdominal cavity, and washed, which was followed by collection of the liquid. After centrifugation at 200×
CO-CULTURE OF PERITONEAL MACROPHAGES WITH RECOMBINANT ADENOVIRUS PAD-JAZF1:
Two groups were established, a PAD-JAZF1 group and a pAdEasy blank virus (control) group. The C57BL/6J mouse peritoneal macrophages prepared as described previously herein were added to 48-well plates for each group and 200 μL of preheated DMEM-10% serum medium was added for culture at 37°C with 5% CO2. After 2 h, the recombinant adenovirus (5×107 PFU) and 50 μL of LPS (1 μg/mL) were added to each well. After 3 or 6 h, the cellular extract was collected and the macrophage subtypes (CD11c+ and CD206+) and inflammatory factors IL-1β, TNF-α (3 h), and IL-6 (6 h) were detected by flow cytometry.
:
Peripheral blood mononuclear cells in C57BL/6J mice were isolated by the immunomagnetic bead method, and PE-CD4+ T cells were separated by flow cytometry. The cell culture solution was IMDM containing 2 M glutamine, 100 U/mL penicillin, 100 μg/mL streptomycin, 50 μM β-mercaptoethanol, and 10% FBS. CD4+ T cells were cultured in a cell culture dish containing anti-CD3 (5 μg/mL) and anti-CD28 (1 μg/mL) antibodies in a 37°C, 5% CO2 culture box following the same process described previously, which was followed by the addition of 100 nM recombinant IL-2 to induce CD4+ T cell differentiation.
:
Following the procedure described previously herein, the cellular extract was collected and the CD4+ T cell subtype (active, memory, restrictive T cells, and Tregs) and inflammatory factors IL-4, IL-10, IFN-γ (3 h), and IL-6 (6 h) were assessed by flow cytometry.
:
A high-fat diet (HFD) mouse model was employed to assess the role of
JAZF1 EXPRESSION MEASURED BY WESTERN BLOTTING AND RT-PCR:
Total liver, muscle, and adipose tissue proteins were extracted and used for SDS-PAGE at 80 V for 30 min and 120 V for 90 min. After transfer to a membrane (wet), a primary antibody (rabbit anti-mouse
For RT-PCR, mRNA extraction and reverse transcription were performed using the TAKARA mRNA Extraction and Reverse Transcription Kit. The reaction conditions were as follows: 37°C for 15 min, 85°C for 5 s, and 4°C for 20 min. PCR was performed immediately using a PCR instrument (Hangzhou Anjes) with the following primers for
HE AND IHC STAINING TO DETECT THE EFFECT OF JAZF1 ON CHRONIC INFLAMMATION:
Fresh liver tissues were obtained from mice for HE and IHC (using reagents provided with a kit from China Biyuntian Biotechnology Co., Shanghai, China). The effects of
STATISTICAL ANALYSIS:
Data are presented as means±SEM and the differences between groups were calculated using independent-samples
Results
CONSTRUCTION OF AN ADENOVIRUS VECTOR TO OVEREXPRESS JAZF1 (PAD-JAZF1):
The Pshuttle-JAZF1 was digested by XhoI and EcoRI double-enzyme digestion and analyzed on agarose gels to identify these fragments (Figure 1A). Two fragments of 6.6 kb and 540 bp were detected, corresponding to pShuttle and the insertion fragment, and the sequences were verified. Using PacI, PAD-JAZF1 was digested into 2 fragments of 30 kb (adenovirus skeleton plasmid) and 3.0 kb (PAD-JAZF1) (Figure 1B), and the inserted fragments were confirmed by sequencing.
CPE assays were performed to determine whether increased viral protein expression led to enhanced cell killing. The infected AD293 cells exhibited an irregular shape consistent with CPE (Figure 1C). A plaque formation assay showed that the virus titers were 7.0×108 and 8.0×108 pfu/mL for PAD-JAZF1 and pAdEasy, respectively.
:
Mouse peritoneal macrophages were cultured with the recombinant adenovirus PAD-JAZF1 and airborne adenovirus pAdEasy. After adding LPS (3 or 6 h), inflammatory factors IL-1β, TNF-α (3 h), and IL-6 (6 h), as well as macrophage subtypes (CD11c+ and CD206+), were detected by flow cytometry. The CD11c+M population and secretion of inflammatory factors were significantly lower and the CD206+ M population was larger in the PAD-JAZF1 group than in the control group. These results indicated that JAZF1 inhibited macrophage differentiation into the CD11c+M lineage, as well as the secretion of inflammatory factors, in vitro (Figure 2).
:
Mouse CD4+ T cells were cultured with the recombinant adenovirus PAD-JAZF1 and airborne adenovirus pAdEasy. After adding recombinant IL-2 stimulus (3 or 6 h), CD4+ T cell subtypes (active, memory, restrictive T cells, and Tregs) and inflammatory factors (IL-4, IL-10, and IFN-γ (3 h) and IL-6 (6 h)), were detected by flow cytometry. The populations of total CD4+ T cells, active T cells, and memory T cells and the secretion of IL-6 and IFN-γ were suppressed in the PAD-JAZF1 group, but Tregs, restrictive T cells, IL-10, and IL-4 were elevated in the PAD-JAZF1 group (Figure 3).
EFFECTS OF THE RECOMBINANT ADENOVIRUS (PAD-JAZF1) ON JAZF1 MRNA AND PROTEIN EXPRESSION IN MOUSE ADIPOSE, LIVER, AND MUSCLE TISSUES:
After mouse tail vein injection of recombinant adenovirus (pad-JAZF1, pAdEasy) for 3 days, mouse adipose, liver, and muscle tissues were collected to evaluate the expression of JAZF1 at the mRNA and protein levels. By RT-PCR and Western blotting, JAZF1 mRNA and protein levels in the PAD-JAZF1 group were significantly higher than those in the pAdEasy infection group (Figure 4), demonstrating that PAD-JAZF1 can increase JAZF1 transcription and translation in each tissue type.
:
JAZF1 inhibited the number of and secretion by peritoneal macrophages in vitro. To determine whether JAZF1 has similar effects in vivo, we transfected PAD-JAZF1 into HFD-fed mice. Liver IHC results showed that CD11c+ macrophages and TNF-α and IL-1β levels in the PAD-JAZF1 group were lower than those in the control group, suggesting that JAZF1 can downregulate CD11c+M and suppress secretion in vivo. To demonstrate the chronic inflammation status in HFD-fed mice, we performed liver HE staining (using RD mice as a control group). Liver tissue inflammation was significantly greater in HFD mice than in RD mice (Figure 5).
:
JAZF1 inhibited the number of and secretion by CD4+ T cells in vitro. We further evaluated the effects of JAZF1 in vivo by transfecting PAD-JAZF1 into HFD-fed mice. Liver IHC results showed that the CD4+ T cell population and the secretion of IL-6 in the PAD-JAZF1 group were lower than those in the pAdEasy control group, but that IL-10 levels were higher. These results indicate that JAZF1 can regulate the liver CD4+ T cell populations and secretion in vivo (Figure 6).
Discussion
Gluconeogenesis, lipid metabolism, insulin sensitivity, islet β cell function, and inflammatory reactions are regulated by
Adipose tissue can secrete a variety of fat-specific inflammatory factors and is involved in glycolipid metabolism [2,4], which mediates macrophage activation, promotes inflammation, and leads to insulin resistance. Inflammatory factors such as TNF-α in the innate immune system released by macrophages phagocytosing necrotic adipose cells hinder the function of the remaining adipose cells. Free fatty acids released from dysfunctional adipose cells can then activate macrophages [14]. This suggests that the interaction between fat cells and macrophages can trigger a cycle of inflammatory reactions. The M1 lineage leads to inflammation and insulin resistance [15–18], but CD206+ macrophages are predominantly anti-inflammatory [19,20]. In tissues with local inflammation, M2 (expressing CD206+) macrophages tend to transform into the M1 type (expressing CD11c+) [21]. In this study, the populations of total macrophages and CD11c+ macrophages and the secretion of proinflammatory cytokines were reduced in the PAD-JAZF1 group. The results of our
Low-grade chronic inflammation contributes to systemic metabolic diseases such as T2D and is associated with immune disorders. The proinflammatory response is further associated with an imbalance in T cell subtypes, and particularly CD4+ T cell subtypes [22]. CD4+ T cells are important for obesity-associated diseases, and adipocytes, as antigen-presenting cells, regulate CD4+ T cell activity [22]. In immunodeficient Rag1−/− and H2A−/− mice, CD4+ T cells mediate obesity memory and promote weight regain, and the depletion of CD4+ T cells leads to obesity memory ablation [22]. In adipose tissues of obese individuals, the Th1-type immune environment dominates the Th2/Treg-type due to the overproduction of proinflammatory cytokines (IFN-γ, IL-6, and TNF-α) and a deficiency in Th2-type processes and interleukins (IL-4, IL-5, IL-10, and IL-13). ILCs2 secrete mostly IL-4, IL-5, IL-9, and IL-13, which are responsible for the accumulation of eosinophils and the polarization of alternatively-activated macrophages, creating a beneficial anti-inflammatory and metabolic regulatory environment in the adipose tissue [23]. These findings indicate that the differentiation of CD4+ T cell subtypes is closely related to lipid metabolism. The expression of genes related to lipid metabolism and the inhibition of lipid accumulation in adipocytes can be regulated by
Tregs, a subtype of CD4+ T cells, have inhibitory effects on immune and inflammatory reactions, especially on the response to an individual’s own antigens [25,26]. They act as anti-inflammatory cells to counter the proinflammatory immune cells responsible for obesity-induced inflammation [27]. A decrease in Tregs will cause metabolic disorders in adipose tissue and insulin resistance [28]. The imbalance of regulatory T (Treg) cells contributes to tissue-specific and systemic inflammation and immunity in T2D, and restoration of the Treg balance is a potential therapeutic approach for prevention and treatment [29]. This is consistent with observations in patients with T2D who are obese and have insulin resistance [30]. Based on these previous findings, the interaction between
Conclusions
In conclusion, co-culture with PAD-JAZF1 affected cell populations and secretion status.
Figures
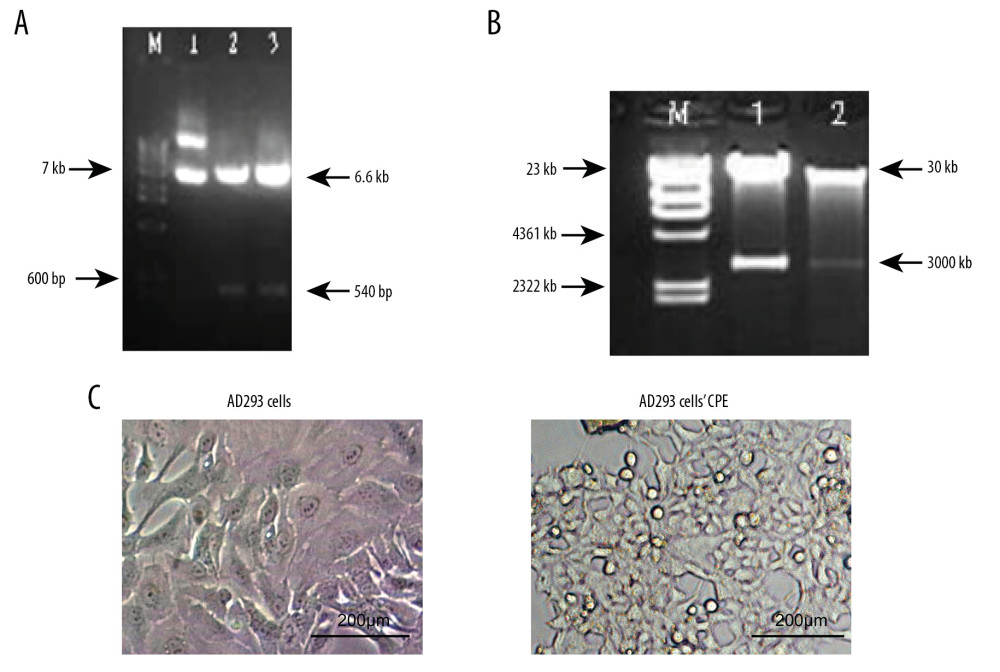 Figure 1. Restriction mapping and cytopathic effect (CPE) on AD293 cells transfected with the adenoviral plasmid. (A) Shuttle plasmid pShuttle-JAZF1 (lane M): wide-range DNA marker; (lane 1): Recombinant plasmid pShuttle (6.6 kb); (lane 2, 3): pShuttle-JAZF1 digested by XhoI and EcoRI (540 bp). (B) Recombinant plasmid pAD-JAZF1 (lane M): λhindIII digestion, up to 23 kb; (lane 1, 2): pAd-JAZF1 digested by PacI. (C) AD293 cells and the CPE.
Figure 1. Restriction mapping and cytopathic effect (CPE) on AD293 cells transfected with the adenoviral plasmid. (A) Shuttle plasmid pShuttle-JAZF1 (lane M): wide-range DNA marker; (lane 1): Recombinant plasmid pShuttle (6.6 kb); (lane 2, 3): pShuttle-JAZF1 digested by XhoI and EcoRI (540 bp). (B) Recombinant plasmid pAD-JAZF1 (lane M): λhindIII digestion, up to 23 kb; (lane 1, 2): pAd-JAZF1 digested by PacI. (C) AD293 cells and the CPE. 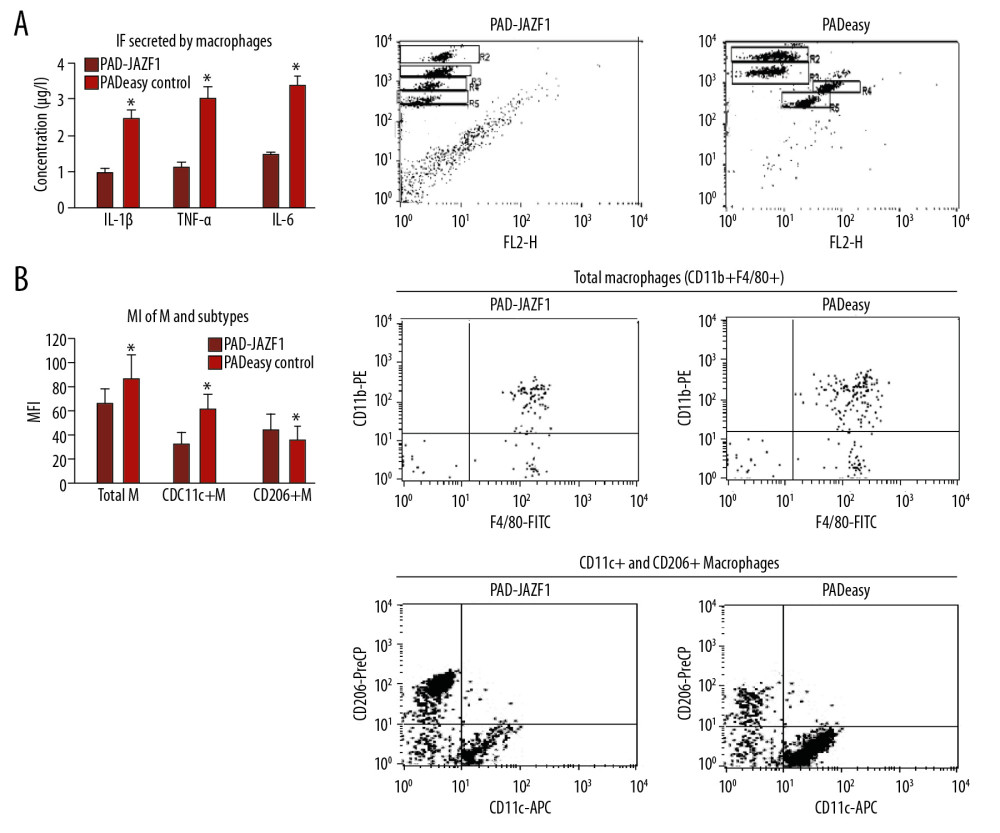 Figure 2. Cell populations and secretion levels in mouse peritoneal macrophages and subtypes in extracts obtained from co-culture conditions. (A) Inflammation-related factors secreted by mouse peritoneal macrophages. Flow cytometry results of IL-1β (gate R3), IL-6 (gate R4), and TNF-α (gate R5) in each group. (B) Mouse peritoneal macrophages and subtype populations. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group.
Figure 2. Cell populations and secretion levels in mouse peritoneal macrophages and subtypes in extracts obtained from co-culture conditions. (A) Inflammation-related factors secreted by mouse peritoneal macrophages. Flow cytometry results of IL-1β (gate R3), IL-6 (gate R4), and TNF-α (gate R5) in each group. (B) Mouse peritoneal macrophages and subtype populations. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group. 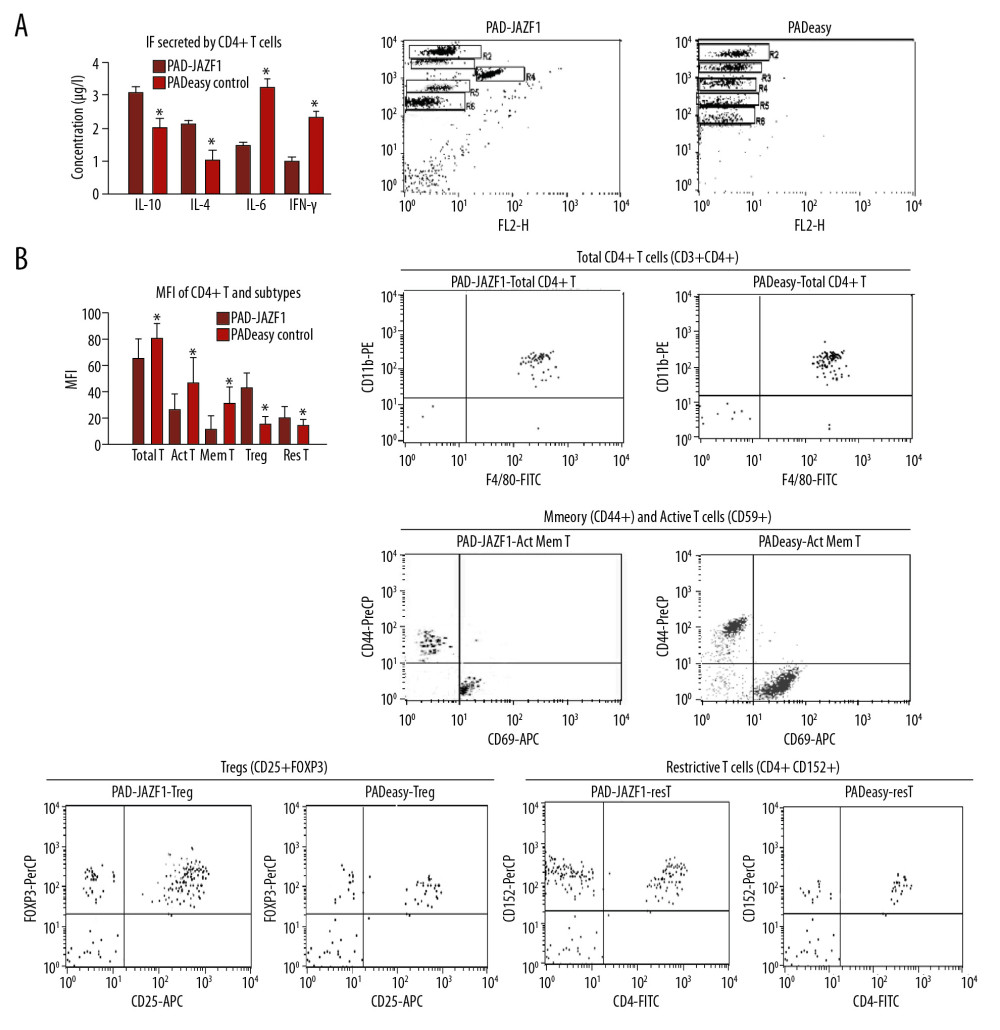 Figure 3. Cell populations and secretion of inflammatory factors by mouse CD4+ T cells and subtypes in co-culture conditions. (A) Inflammatory factors secreted by CD4+ T cells. Flow cytometry results for IL-6 (gate R3), IL-10 (gate R4), IFN-γ (gate R5), and IL-4 (gate R6) in each group. (B) Mouse CD4+ T cells and their subtypes. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group.
Figure 3. Cell populations and secretion of inflammatory factors by mouse CD4+ T cells and subtypes in co-culture conditions. (A) Inflammatory factors secreted by CD4+ T cells. Flow cytometry results for IL-6 (gate R3), IL-10 (gate R4), IFN-γ (gate R5), and IL-4 (gate R6) in each group. (B) Mouse CD4+ T cells and their subtypes. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group. 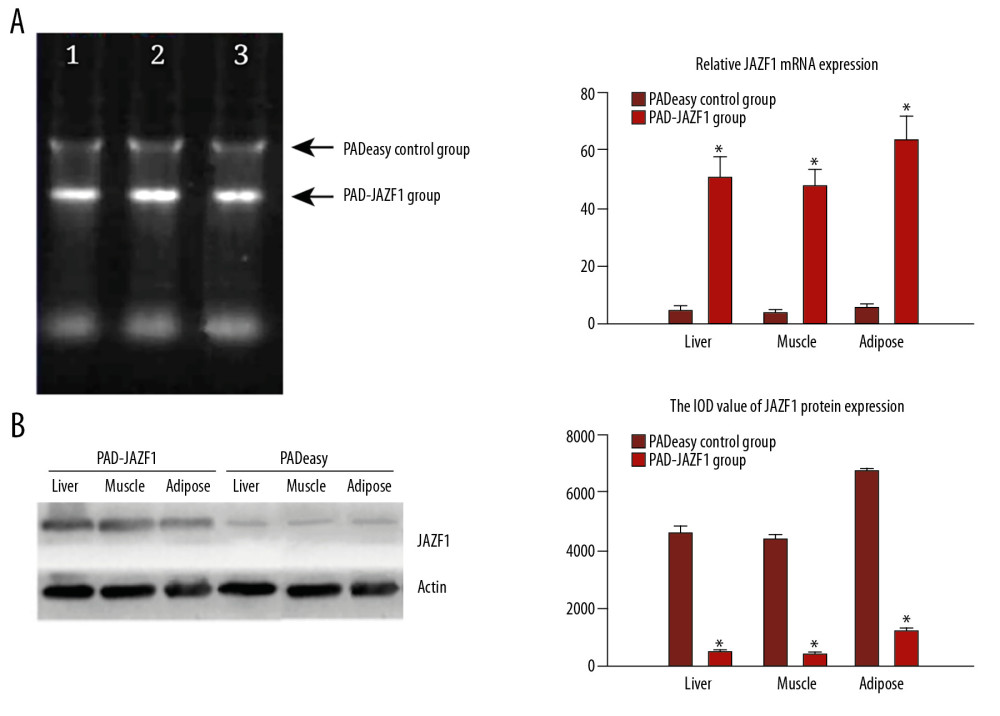 Figure 4. mRNA and protein expression levels of JAZF1 in mouse liver, muscle, and adipose tissues in the PAD-JAZF1 and control groups. (A) JAZF1 mRNA expression levels in the 2 groups. Lane 1, liver; lane 2, muscle; lane 3, adipose tissue 4. (B) JAZF1 protein expression levels in the PAD-JAZF1 and control groups.
Figure 4. mRNA and protein expression levels of JAZF1 in mouse liver, muscle, and adipose tissues in the PAD-JAZF1 and control groups. (A) JAZF1 mRNA expression levels in the 2 groups. Lane 1, liver; lane 2, muscle; lane 3, adipose tissue 4. (B) JAZF1 protein expression levels in the PAD-JAZF1 and control groups. 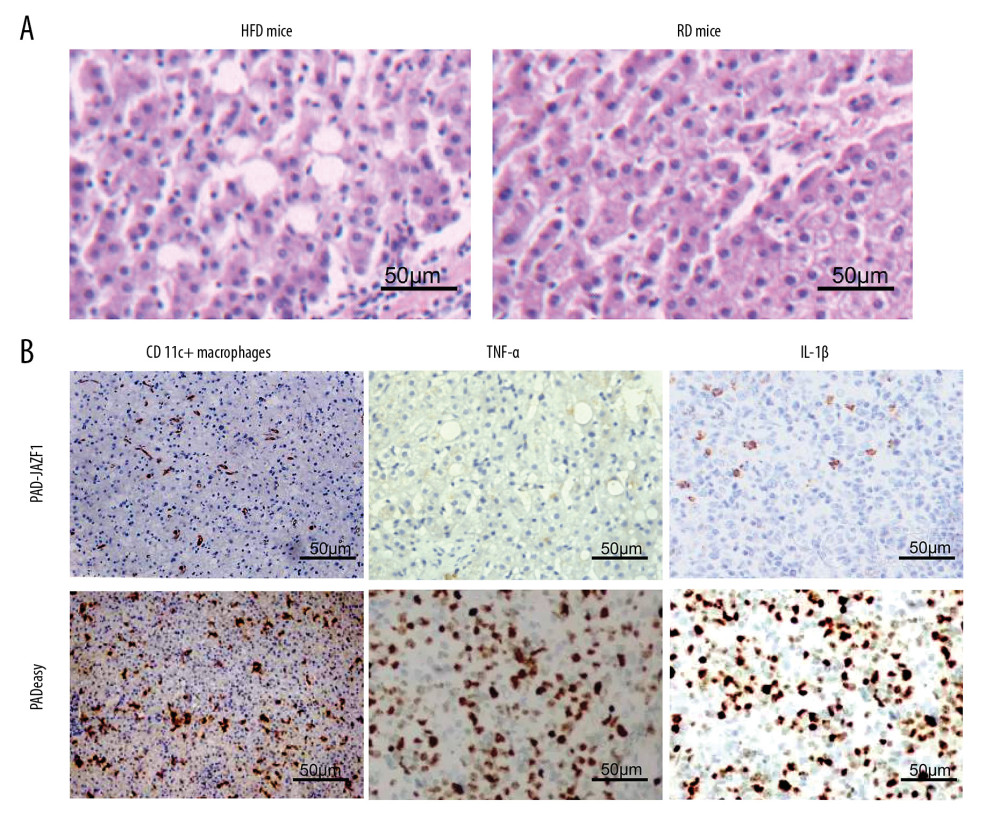 Figure 5. CD11c+ macrophages and TNF-α and IL-1β expression levels in the liver. (A) HE staining results for the liver of high-fat diet (HFD)-fed mice and regular diet (RD)-fed mice. (B) IHC staining results for CD11c+ macrophages and TNF-α and IL-1β levels in the livers of the PAD-JAZF1 group and pAdEasy control group.
Figure 5. CD11c+ macrophages and TNF-α and IL-1β expression levels in the liver. (A) HE staining results for the liver of high-fat diet (HFD)-fed mice and regular diet (RD)-fed mice. (B) IHC staining results for CD11c+ macrophages and TNF-α and IL-1β levels in the livers of the PAD-JAZF1 group and pAdEasy control group. 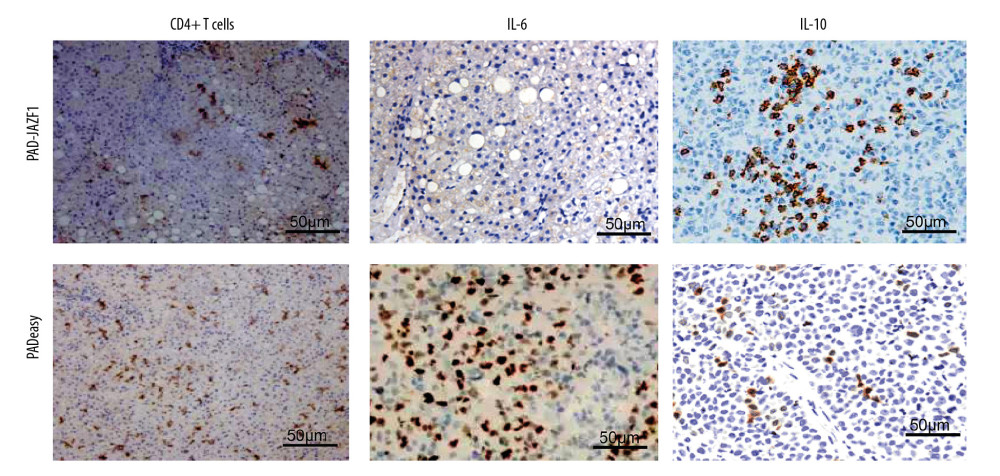 Figure 6. IHC analysis of CD4+ T cells and IL-6 and IL-10 expression levels in the livers of the PAD-JAZF1 and pAdEasy control groups.
Figure 6. IHC analysis of CD4+ T cells and IL-6 and IL-10 expression levels in the livers of the PAD-JAZF1 and pAdEasy control groups. References
1. Chatterjee S, Khunti K, Davies MJ, Type 2 diabetes: Lancet, 2017; 389; 2239-51
2. Jafarirad S, Ayoobi N, Karandish M, Dark chocolate effect on serum adiponectin, biochemical and inflammatory parameters in diabetic patients: A randomized clinical trial: Int J Prev Med, 2018; 9; 86
3. Redwine LS, Pung MA, Wilson K, Differential peripheral inflammatory factors associated with cognitive function in patients with heart failure: Neuroimmunomodulation, 2018; 25; 146-52
4. Li C, Xu MM, Wang K, Macrophage polarization and meta-inflammation: Transl Res, 2018; 191; 29-44
5. Becker M, Levings MK, Daniel C, Critical players in adipose-immune crosstalk: Eur J Immunol, 2017; 47; 1867-74
6. Meng F, Lin Y, Yang M, JAZF1 inhibits adipose tissue macrophages and adipose tissue inflammation in diet-induced diabetic mice: Biomed Res Int, 2018; 2018 4507659
7. Gong X, Zhang C, Yiliyasi-Aisa Y-A, A comparative analysis of genetic diversity of candidate genes associated with type 2 diabetes in worldwide populations: Yi Chuan, 2016; 38; 543-59
8. Wei Q, Zhou B, Yang G, JAZF1 ameliorates age and diet-associated hepatic steatosis through SREBP-1c -dependent mechanism: Cell Death Dis, 2018; 9; 859
9. Rasheed MA, Kantoush N, Abd El-Ghaffar N, Expression of JAZF1, ABCC8, KCNJ11and Notch2 genes and vitamin D receptor polymorphisms in type 2 diabetes, and their association with microvascular complications: Ther Adv Endocrinol Metab, 2017; 8; 97-108
10. Liao ZZ, Wang YD, Qi XY, Xiao XH, JAZF1, a relevant metabolic regulator in type 2 diabetes: Diabetes Metab Res Rev, 2019; 35; e3148
11. Hebbar P, Abubaker JA, Abu-Farha M, A perception on genome-wide genetic analysis of metabolic traits in Arab populations: Front Endocrinol (Lausanne), 2019; 10; 8
12. Yuan L, Luo X, Zeng M, Transcription factor TIP27 regulates glucose homeostasis and insulin sensitivity in a PI3-kinase/Akt-dependent manner in mice: Int J Obes (Lond), 2015; 39; 949-58
13. Yang M, Dai J, Jia Y, Overexpression of juxtaposed with another zinc finger gene 1 reduces proinflammatory cytokine release via inhibition of stress-activated protein kinases and nuclear factor-kappaB: FEBS J, 2014; 281; 3193-205
14. Nguyen KD, Qiu Y, Cui X, Alternatively activated macrophages produce catecholamines to sustain adaptive thermogenesis: Nature, 2011; 480; 104-8
15. Lumeng CN, Adipose tissue macrophages: A piece of the PAI of metabolic syndrome: Sci Transl Med, 2010; 2; 20ps7
16. Lumeng CN, Bodzin JL, Saltiel AR, Obesity induces a phenotypic switch in adipose tissue macrophage polarization: J Clin Invest, 2007; 117; 175-84
17. Lumeng CN, Deyoung SM, Bodzin JL, Saltiel AR, Increased inflammatory properties of adipose tissue macrophages recruited during diet-induced obesity: Diabetes, 2007; 56; 16-23
18. Lumeng CN, Saltiel AR, Inflammatory links between obesity and metabolic disease: J Clin Invest, 2011; 121; 2111-17
19. Fujisaka S, Usui I, Ikutani M, Adipose tissue hypoxia induces inflammatory M1 polarity of macrophages in an HIF-1alpha-dependent and HIF-1alpha-independent manner in obese mice: Diabetologia, 2013; 56; 1403-12
20. Kambara K, Ohashi W, Tomita K: Am J Pathol, 2015; 185; 162-71
21. Pejnovic NN, Pantic JM, Jovanovic IP, Galectin-3 deficiency accelerates high-fat diet-induced obesity and amplifies inflammation in adipose tissue and pancreatic islets: Diabetes, 2013; 62; 1932-44
22. Jagannathan-Bogdan M, McDonnell ME, Shin H, Elevated proinflammatory cytokine production by a skewed T cell compartment requires monocytes and promotes inflammation in type 2 diabetes: J Immunol, 2011; 186; 1162-72
23. Chalubinski M, Luczak E, Wojdan K, Innate lymphoid cells type 2 – emerging immune regulators of obesity and atherosclerosis: Immunol Lett, 2016; 179; 43-46
24. Ming GF, Xiao D, Gong WJ, JAZF1 can regulate the expression of lipid metabolic genes and inhibit lipid accumulation in adipocytes: Biochem Biophys Res Commun, 2014; 445; 673-80
25. Sun L, Fu J, Zhou Y, Metabolism controls the balance of Th17/T-regulatory cells: Front Immunol, 2017; 8; 1632
26. Han JM, Patterson SJ, Speck M, Insulin inhibits IL-10-mediated regulatory T cell function: Implications for obesity: J Immunol, 2014; 192; 623-29
27. Lee BC, Lee J, Cellular and molecular players in adipose tissue inflammation in the development of obesity-induced insulin resistance: Biochim Biophys Acta, 2014; 1842; 446-62
28. Feuerer M, Herrero L, Cipolletta D, Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters: Nat Med, 2009; 15; 930-39
29. Wang M, Chen F, Wang J, Th17 and Treg lymphocytes in obesity and Type 2 diabetic patients: Clin Immunol, 2018; 197; 77-85
30. Reilly SM, Saltiel AR, Adapting to obesity with adipose tissue inflammation: Nat Rev Endocrinol, 2017; 13; 633-43
Figures
 Figure 1. Restriction mapping and cytopathic effect (CPE) on AD293 cells transfected with the adenoviral plasmid. (A) Shuttle plasmid pShuttle-JAZF1 (lane M): wide-range DNA marker; (lane 1): Recombinant plasmid pShuttle (6.6 kb); (lane 2, 3): pShuttle-JAZF1 digested by XhoI and EcoRI (540 bp). (B) Recombinant plasmid pAD-JAZF1 (lane M): λhindIII digestion, up to 23 kb; (lane 1, 2): pAd-JAZF1 digested by PacI. (C) AD293 cells and the CPE.
Figure 1. Restriction mapping and cytopathic effect (CPE) on AD293 cells transfected with the adenoviral plasmid. (A) Shuttle plasmid pShuttle-JAZF1 (lane M): wide-range DNA marker; (lane 1): Recombinant plasmid pShuttle (6.6 kb); (lane 2, 3): pShuttle-JAZF1 digested by XhoI and EcoRI (540 bp). (B) Recombinant plasmid pAD-JAZF1 (lane M): λhindIII digestion, up to 23 kb; (lane 1, 2): pAd-JAZF1 digested by PacI. (C) AD293 cells and the CPE. Figure 2. Cell populations and secretion levels in mouse peritoneal macrophages and subtypes in extracts obtained from co-culture conditions. (A) Inflammation-related factors secreted by mouse peritoneal macrophages. Flow cytometry results of IL-1β (gate R3), IL-6 (gate R4), and TNF-α (gate R5) in each group. (B) Mouse peritoneal macrophages and subtype populations. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group.
Figure 2. Cell populations and secretion levels in mouse peritoneal macrophages and subtypes in extracts obtained from co-culture conditions. (A) Inflammation-related factors secreted by mouse peritoneal macrophages. Flow cytometry results of IL-1β (gate R3), IL-6 (gate R4), and TNF-α (gate R5) in each group. (B) Mouse peritoneal macrophages and subtype populations. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group. Figure 3. Cell populations and secretion of inflammatory factors by mouse CD4+ T cells and subtypes in co-culture conditions. (A) Inflammatory factors secreted by CD4+ T cells. Flow cytometry results for IL-6 (gate R3), IL-10 (gate R4), IFN-γ (gate R5), and IL-4 (gate R6) in each group. (B) Mouse CD4+ T cells and their subtypes. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group.
Figure 3. Cell populations and secretion of inflammatory factors by mouse CD4+ T cells and subtypes in co-culture conditions. (A) Inflammatory factors secreted by CD4+ T cells. Flow cytometry results for IL-6 (gate R3), IL-10 (gate R4), IFN-γ (gate R5), and IL-4 (gate R6) in each group. (B) Mouse CD4+ T cells and their subtypes. Values are presented as means±SEM (n=6/group). * P<0.05 vs. the pAdEasy control group. Figure 4. mRNA and protein expression levels of JAZF1 in mouse liver, muscle, and adipose tissues in the PAD-JAZF1 and control groups. (A) JAZF1 mRNA expression levels in the 2 groups. Lane 1, liver; lane 2, muscle; lane 3, adipose tissue 4. (B) JAZF1 protein expression levels in the PAD-JAZF1 and control groups.
Figure 4. mRNA and protein expression levels of JAZF1 in mouse liver, muscle, and adipose tissues in the PAD-JAZF1 and control groups. (A) JAZF1 mRNA expression levels in the 2 groups. Lane 1, liver; lane 2, muscle; lane 3, adipose tissue 4. (B) JAZF1 protein expression levels in the PAD-JAZF1 and control groups. Figure 5. CD11c+ macrophages and TNF-α and IL-1β expression levels in the liver. (A) HE staining results for the liver of high-fat diet (HFD)-fed mice and regular diet (RD)-fed mice. (B) IHC staining results for CD11c+ macrophages and TNF-α and IL-1β levels in the livers of the PAD-JAZF1 group and pAdEasy control group.
Figure 5. CD11c+ macrophages and TNF-α and IL-1β expression levels in the liver. (A) HE staining results for the liver of high-fat diet (HFD)-fed mice and regular diet (RD)-fed mice. (B) IHC staining results for CD11c+ macrophages and TNF-α and IL-1β levels in the livers of the PAD-JAZF1 group and pAdEasy control group. Figure 6. IHC analysis of CD4+ T cells and IL-6 and IL-10 expression levels in the livers of the PAD-JAZF1 and pAdEasy control groups.
Figure 6. IHC analysis of CD4+ T cells and IL-6 and IL-10 expression levels in the livers of the PAD-JAZF1 and pAdEasy control groups. Most Viewed Current Articles
13 Apr 2020 : Original article 19,586
Outcome of 24 Weeks of Combined Schroth and Pilates Exercises on Cobb Angle, Angle of Trunk Rotation, Chest...DOI :10.12659/MSMBR.920449
Med Sci Monit Basic Res 2020; 26:e920449
20 Apr 2018 : Original article 17,466
Brain Training Games Enhance Cognitive Function in Healthy SubjectsDOI :10.12659/MSMBR.909022
Med Sci Monit Basic Res 2018; 24:63-69
23 Jul 2016 : Review article 10,353
Cardiac Hypertrophy: An Introduction to Molecular and Cellular BasisDOI :10.12659/MSMBR.900437
Med Sci Monit Basic Res 2016; 22:75-79
10 Aug 2020 : Clinical Research 9,788
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Rando...DOI :10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264








