10 August 2020: Clinical Research
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Randomized Controlled Study
Su-yeon Hong1BCD, Young Moon2BCD, Jong-duk Choi3AEF*DOI: 10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264
Abstract
BACKGROUND: The decreased postural control ability of stroke patients affects their ability to balance in various postures such as sitting and standing. This study aimed to determine whether cognitive task training for stroke patients is effective in improving walking and balancing abilities.
MATERIAL AND METHODS: Seventeen stroke patients (10 males, 7 females) were randomized by ballot to be assigned to the cognitive task group (CBT) or the general task group (GBT). For the cognitive task training, a dual task of balance and cognition using traffic signals, a familiar form to the subjects, was applied as a program. In both groups the interventions were performed for 30 min a day, 3 times a week, for 4 weeks. The timed up and go test (TUG), the Berg balance scale (BBS), and gait ability evaluation were performed to compare the therapeutic effects.
RESULTS: After the intervention, the BBS showed significant differences in both groups (p<0.05). The cognitive task training group had significant improvement in all outcome scores after the intervention (p<0.05). The TUG score of the CBT group significantly decreased to 6.17 s (p<0.05), but that of the GBT showed no statistically significant change.
CONCLUSIONS: Cognitive task training could be used in clinical rehabilitation as a more effective intervention method to improve balance and gait ability of stroke patients.
Keywords: Cognitive Therapy, postural balance, Stroke, Cognition, Gait, Task Performance and Analysis
Background
Stroke is a cerebrovascular disease that causes motor, cognitive, perceptual, and visual disorders [1]. Stroke patients are restricted from participating in various activities such as activities of daily living, social activities, and work [2,3]. The reduction in post-stroke mobility decreases the ability to maintain a center of balance by having both sides imbalanced, causes asymmetric postures, and affects the orientation response, resulting in serious problems with postural control ability [4]. The decreased postural control ability of stroke patients affects their ability to balance in various postures, such as sitting and standing, which are behavioral skills essential for daily living [5]. The reduced balance in stroke patients is a barrier to performing daily living activities, reducing individual independence and limiting social activities [6–8].
In addition, post-stroke gait disorders have a negative effect on the level of functional independence and prognosis; therefore, regaining gait ability is an important factor directly related to patient independence and is one of the goals of rehabilitation [9]. Cho and Lee [10] reported that the gait ability of stroke patients is associated with bipedal balance and that walking that necessitates postural balancing works together when it requires performing complex exercise tasks in everyday life. The gait of hemiplegia patients with stroke has a short weight-support time on the affected side and a long swing phase, producing a difference in step length between the affected and the less-affected sides. These patients also have a slow gait cycle and velocity [11] and a reduced coordination ability to appropriately respond to a variety of environments and tasks [12].
Postural control can be categorized into conscious and unconscious control in response to vision, the vestibular system, and proprioceptors [13]. Posture control is also associated with adjusting postural stability and orientation in space [14,15].
Automatic stabilization is needed to maintain balance [16,17]. It is defined as a postural response that occurs unconsciously and automatically while consciously maintaining posture or performing movements [18]. However, in stroke patients, the center of the body is moved to the less-affected side, symmetrical weight-loading does not occur, and proper muscle control is difficult because of abnormal muscle recruitment [19,20]. Pathologic neuromuscular control conditions in such stroke patients make it difficult to unconsciously and automatically adjust posture, which must normally be achieved.
Recently, methods for restoring the motor function of stroke patients have been proposed. Among them, Carr and Shepherd’s [21] dual-task training based on motor learning has been assessed in various ways. Dual-task training is a therapeutic intervention in which subjects simultaneously perform a motor task or a movement task and a cognition task, and it is used as an intervention to restore the movement of neurologically impaired patients such as those with stroke [22]. Several previous studies have reported that dual-task training is effective in improving stroke symptoms, endurance, and walking by applying dual-task training to stroke patients [23,24]. Shumway-Cook and Woollacott [25] and Andersson et al. [26] reported a reduction in perturbation due to increased arousal levels to improve performance during secondary tasks in a dual-task environment. Interestingly, Huxhold et al. [27] also reported that this effect is typically present in young adults and in older people or patients with balance impairment when performing simple cognitive tasks.
During dual-task training, the subject can focus on the cognitive task, and by focusing posture control on the unconscious and automatic control pattern, the subject can produce the results of postural ability and functional enhancement [25].
Although there have been many studies on improvement of postural control of stroke patients, most have focused on conscious-level postural control, and the exact mechanisms underlying the effects of interventions that promote unconscious-level postural control to make automatic stabilization are still unknown [28]. Moreover, there have been few studies that promote the automatic stabilization mechanism through cognitive tasks based on the concept of dual-task training, and studies applying it to stroke patients are still lacking. Thus, the present study aimed to determine whether cognitive balancing dual-task training that promotes unconscious-level motor control for stroke patients is more effective in improving walking and balancing abilities compared with general balancing training.
Material and Methods
PARTICIPANTS:
Among stroke patients who had been diagnosed at least 6 months before, those who were able to walk 10 m, who were able to stand for 3 min without aid, and who had at least 24 points in the Mini-Mental State Examination were selected for this study. The exclusion criteria were: (1) accompanying diseases or disorders other than stroke that can interfere with balance training; and (2) uncontrolled health conditions in which exercise is contraindicated. This study was reviewed and approved by the Institutional Review Board of Daejeon University (approval number: 1040647-201806-HR-026-01), and was conducted in accordance with the criteria established by the Helsinki Declaration.
STUDY DESIGN:
Thirty-one subjects were recruited for this study, and 7 of them were excluded because they refused to participate. The remaining 24 were randomized by ballot to be assigned to the cognitive task training (CBT) group or the general task training (GBT) group. As 4 subjects in the CBT group and 3 in the GBT group were excluded because they transferred to other hospitals or were discharged, 17 subjects remained for this experiment. TUG, BBS, and gait ability evaluation were performed to compare the therapeutic effects of the intervention. A pre-intervention evaluation was performed before the intervention, and a post-evaluation was performed after the 4-week intervention to compare and analyze the results.
INTERVENTION:
For the cognitive task training, a dual task of balance and cognition using traffic signals, a familiar form for the subjects, was applied as a program. For the intervention, the task was performed with scaffolds on which the starting and the target points were marked, monitors for showing visual cues of the cognitive task, and elastic bands for controlling the amounts and difficulties of resistance.
The traffic signal cognitive task for the CBT group was a task in which the subject, in a standing posture, moved the lower extremity of the less-affected side toward the 3 flexion directions of the hip joint and then moved it back in place according to the visual cues on the monitor. These visual cues on the monitor were red and green, the colors of traffic lights. The subject was asked to stop moving when the cue turned red and to perform the task when the cue turned green. There were 3 traffic signals, with each representing 1 of the 3 directions. The colors and locations of the lights were set to change randomly on the monitor. The subject was expected to move the lower extremity of the less-affected side toward the indicated direction within 5 s after a signal turned green and then move back the extremity in place within 5 s. The subject started the task in a standing posture with both feet on the designated positions of the scaffold marked with the starting and target points (Figure 1A). One set consisted of 30 random movements. The monitor was placed where the subject could see it straight ahead, adjusted for subject height.
Given the fatigue of the subjects, a 1-min rest was allowed between each of the 5 sets. The steps of the cognitive balance training were organized in order of increasing difficulty. Step 1 was performed without elastic bands (Figure 1B). Step 2 made a subject perform the task with an elastic band. The subject was asked to move the less-affected lower extremity toward the targeted point against the resistance of the elastic band and then move the extremity back in place (Figure 1C). In step 3, the subject performed the same movements with an elastic band of a different color to increase resistance. The elastic bands were located around the ankle of the less-affected side. The exercise loading was reset every week according to the adaptability of the subject. The task performance of each subject was video-recorded, and the subject was required to move on to the next step when the success rate was 80% or over.
The GBT group performed the task of moving the lower extremity of the affected side in the same way as the CBT group but without a cognitive task. Given the muscular fatigue of the subjects, a 1-min rest was allowed between each of the 5 sets. The difficulty was controlled in the same way as in the CBT group. In both groups, the interventions were performed for 30 min a day, 3 times a week, for 4 weeks.
TIMED UP AND GO TEST (TUG): The TUG test is a simple method to assess quickness, velocity, agility, and dynamic balance. It measures the time that a subject sitting on an armchair takes to rise from the chair with a starting signal, walk 3 m, turn around a cone, walk back to the chair, and completely sit down as fast as possible. The intra-rater reliability was set to r=0.99, and the inter-rater reliability was set to r=0.98 [29].
BERG BALANCE SCALE (BBS): The BBS is a tool for assessing the dynamic balance ability of hemiplegic patients with brain diseases. It consists of 14 items related to activities of daily life. The total score is 56 points; the higher the score is, the better the balance of a subject. The test-retest reliability was 0.98, and the inter-rater reliability was set to r=0.98 [30].
GAIT: The GAITRite (CIR Systems, Inc., USA) is used to assess the gait ability of patients. The validity and reliability of this evaluation tool are verified by measuring the temporal and spatial factors of gait in several studies to collect quantitative data on the types of patients’ gait. The rater reliability was set to r=0.90 [31]. The intraclass correlation coefficients of all gait measurements of comfortable gait velocity were above 0.96 [32].
STATISTICAL ANALYSIS:
The collected data were statistically analyzed using SPSS for Windows (ver. 22.0). The general characteristics of the subjects are provided by means and standard deviation values with descriptive statistics. Normality of data distribution was assessed with the Shapiro-Wilk test. The homogeneity test of the general characteristics between the groups and the results before the experiment was conducted by a chi-square test and a paired
Results
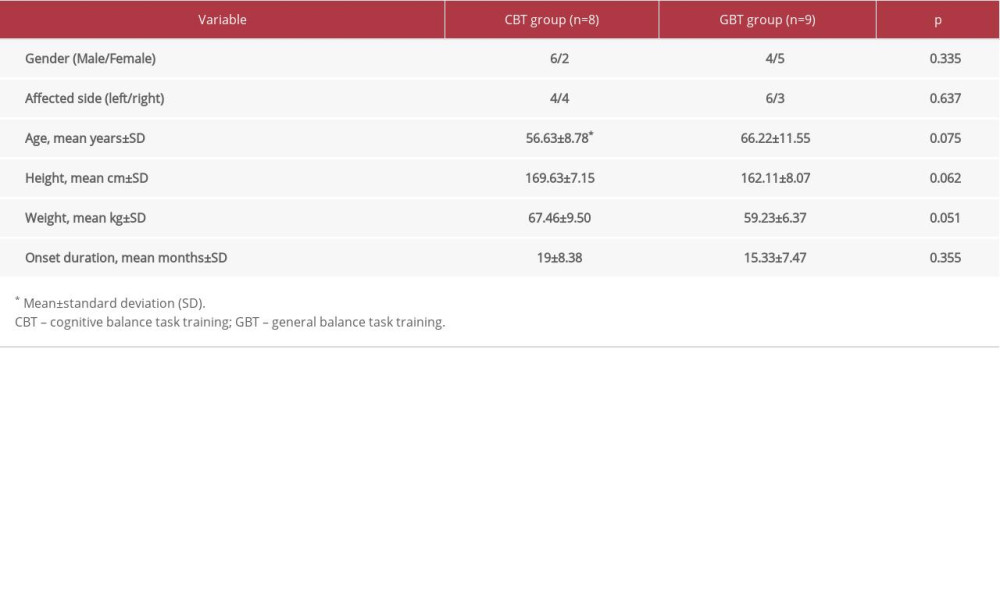
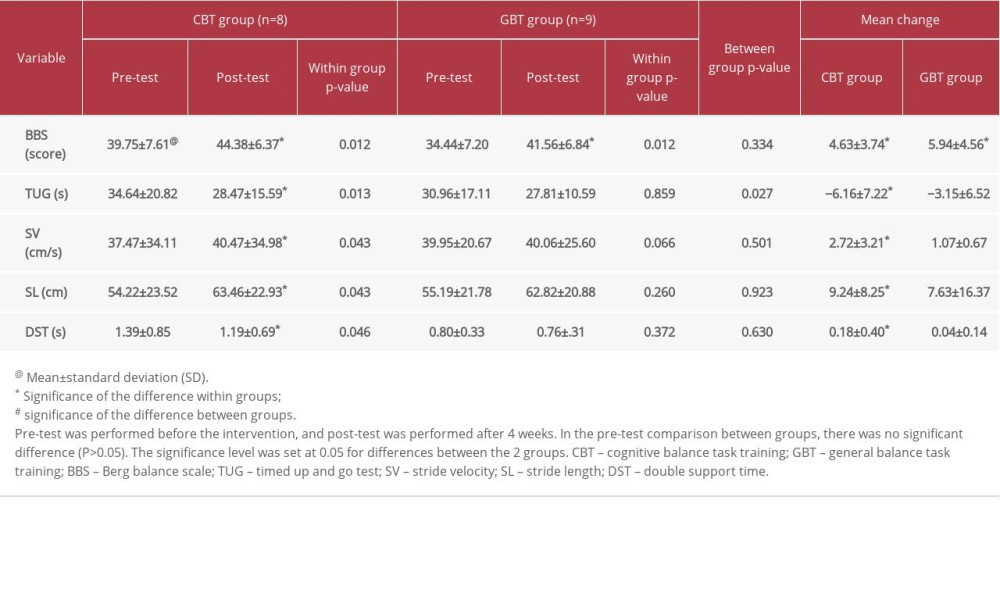
A total of 17 subjects participated in this study: 8 in the CBT group and 9 in the GBT group. Table 1 shows the general characteristics of the subjects. After the intervention, the BBS of the CBT group significantly increased by 4.63 (p<0.05), and that of the GBT group also significantly increased to 7.12 (p<0.05). The TUG of the CBT group significantly decreased to 6.17 s (p<0.05), but that of the GBT showed no statistically significant difference (Table 2).
The stride velocity of the CBT group significantly increased from 37.47±34.11 cm/s to 40.57±34.98 cm/s (p<0.05), and that of the GBT group showed improvement but had no statistically significant difference. The stride length of the affected side in the CBT group significantly increased from 61.22±23.52 cm to 63.46±22.93 cm (p<0.05), and that of the GBT group was longer than the value before intervention but had no statistically significant difference. The double support time of the CBT group significantly decreased from 1.39±0.85 s to 1.19±0.69 s (p<0.05). No significant difference was found in the GBT group (Table 2).
Discussion
Some of the most distinctive features of stroke patients are asymmetrical postures and movement patterns between the affected and the less-affected sides and difficulty in normal weight loading and movement in sitting, standing, and sit-and-stand postures and during gait, which affect their overall body movements in their daily lives [33]. In this study, we assessed the effects of clinical application of cognitive dual-task balance training on the automatic stabilization mechanism for automatic postural control, which is essential for balance and gait in stroke patients.
Posture control is influenced by various factors, such as posture difficulty, cognitive task complexity, and balance ability of subjects. Most patients with neurological problems have impaired balance control [15]. Jeon and Chung [34] reported that it is necessary to compare the effects of the task types used in the dual task on postural control and to select cognitive tasks that can effectively improve postural control.
Repetitive learning of daily life-related tasks in stroke patients promotes neuroplasticity of the brain [35], thereby changing the postural control sequence from conscious to automatic processing [36]. Therefore, in this study, we applied the traffic signal cognition task with a low level of difficulty to stroke patients with impaired balance control. The traffic signal cognitive task, which was selected as an intervention program for cognitive balance training, was considered to cause the subject to focus on cognitive tasks, thereby promoting postural control, which is the primary task, and triggering automatic stabilization [37,38].
Mobility, one of the therapeutic goals of rehabilitation, is defined as the ability to move from one place to another. Therefore, restoring as much independent mobility as possible to the subject is the primary goal of rehabilitation [15]. Pai and Patton [39] suggested that moving the body in a stable state, in which the center of gravity of the body is above the basal plane, creates a new basal plane, changing from static balance control to dynamic balance control. These dynamic challenges can improve balance [40]. As a result of the cognitive balance training conducted in this study, the subjects showed functional improvement in the TUG and BBS. This improvement is considered to be influenced by the forward motion of the healthy foot used as a training method and the use of repeated static and dynamic balances [41,42].
The cognitive task of this study was a repetitive task with a target point. It is effective in inducing movement of the center of gravity of the body and in repeatedly replacing concentric and eccentric control. It appears to have contributed to improvement in the static and dynamic balance of subjects.
Proprioceptive neuromuscular facilitation is an indirect treatment method that creates muscle contraction of the affected side using the radiation effect by giving resistance to the healthy side [43]. Cordova et al. [44] assessed it as the basis for the automatic stabilization mechanism through the electromyography results of the support leg, which is the opposite side during the lower-extremity resistance exercise. Moreover, the right shoulder spreading movement was reported to have activated the opposite left erector spinae to create automatic stability of the spine [45]. The first determinants of walking speed are stride length and repeated speed during walking [46]. These elements of time and distance, combined with stator and stance, form the characteristics of an individual’s stride and appear as the basic walking ability. Hemiplegic patients are characterized by slow gait cycles and speeds, differences between the lesion and step lengths, and short statures and relatively long stilts on the affected side [47]. The cause of the slower walking speed of stroke patients is related to shorter coverage [11]. Although many stroke patients recover their independent gait, gait disturbances occur. In particular, the reduction of the ability to walk over long distances is a major limitation to community participation [48].
Clinically, walking speed is used as a measure of independent walking ability to perform daily activities and the level of functional recovery [49]. According to Kreb et al. [50], increasing muscular strength in the lower limbs through training increases the ground reaction force, and an improved balance ability shortens the double support time, increasing gait velocity. When gait velocity increases, the stance phase gradually shortens compared with the swing phase [51]. The results of the present study show that the support time of cognitive balance was significantly reduced from 1.39±0.85 s to 1.19±0.69 s. Stride length increased significantly from 61.22±23.52 cm to 63.46±22.939 cm, and the affected stride velocity increased from 37.47±34.11 m/s to 40.47±34.98 m/s. This suggests that the cognitive task training used in this study promoted the automatic posture control of the subjects, thereby improving the ability and unconscious walking ability.
This was a pilot study conducted to identify the effects of cognitive task training. The intervention duration was 4 weeks, which might be too short to effectively improve the function of patients with chronic stroke and could be a limitation in determining the long-term effects after the intervention. In addition, there was no difference in the GBT group after intervention compared with before the intervention. For comparison between groups and within groups, only the
Conclusions
This study investigated the effects of cognitive task training focused on unconscious-level motor control for chronic stroke patients and compared the effects of general balance training. In the evaluation of the balance and gait ability of stroke patients, cognitive balance training showed a statistically significant improvement over general balance training. Therefore, cognitive dual-task balance training could be used in clinical rehabilitation as a more effective intervention to improve balance and gait ability of stroke patients.
References
1. Laver KE, George S, Thomas S, Virtual reality for stroke rehabilitation: Cochrane Database Syst Rev, 2015; 12; CD008349
2. Go AS, Mozaffarian D, Roger VL, A report from the American Heart Association: Circulation, 2013; 127; e6-245
3. Sansonetti D, Hoffmann T, Cognitive assessment across the continuum of care: The importance of occupational performance-based assessment for individuals post-stroke and traumatic brain injury: Aust Occup Ther J, 2013; 60; 334-42
4. Ikai T, Kamikubo T, Takehara I, Dynamic postural control in patients with hemiparesis: Am J Phys Med Rehabil, 2003; 82; 463-69
5. Benaim C, Pérennou DA, Villy J, Validation of a standardized assessment of postural control in stroke patients: The postural assessment scale for stroke patients (PASS): Stroke, 1999; 30; 1862-68
6. Perry J, Burnfield J: Gait Analysis: Normal and pathological function, 2010; 485-86, Slack
7. Persson CU, Hansson PO, Danielsson A, A validation study using a modified version of Postural Assessment Scale for Stroke Patients: Postural Stroke Study in Gothenburg (POSTGOT): J Neuroeng Rehabil, 2011; 8; 57
8. Wang CH, Hsueh IP, Sheu CF, Discriminative, predictive, and evaluative properties of a trunk control measure in patients with stroke: Phys Ther, 2005; 85; 887-94
9. Taylor-Piliae RE, Latt LD, Hepworth JT, Predictors of gait velocity among community-dwelling stroke survivors: Gait Posture, 2012; 35; 395-99
10. Cho MS, Lee DY, The effects of standing balance and ambulation, activities of daily living using bio-feedback training with weight bearing in the less than 3 months and more than 6 months groups hemiparesis patients: Journal of Special Education & Rehabilitation Science, 2007; 46; 123-42
11. Wagenaar RC, Beek WJ, Hemiplegic gait: A kinematic analysis using walking speed as a basis: J Biomech, 1992; 25; 1007-15
12. Roerdink M, Lamoth CJ, Kwakkel G, Gait coordination after stroke: Benefits of acoustically paced treadmill walking: Phys Ther, 2007; 87; 1009-22
13. Horak FB, Macpherson JM, Postural orientation and equilibrium: Handbook of Physiology: Section 12, Exercise Regulation and Integration of Multiple Systems, 1996; 255-92, New York, Oxford University Press
14. Brauer SG, Mediolateral postural stability: Changes with age and prediction of fallers: Doctoral dissertation, 1998, University of Queensland
15. Shummway-Cook A, Woollacott M: Motor Control: Translating research into clinical practice, 2012; 161-93, St West Camden, Lipponcott, Williams & Sildins
16. Cordo PJ, Nashner LM, Properties of postural adjustments associated with rapid arm movements: J Neurophysiol, 1982; 47; 287-302
17. Horak F, Nashner L, Central programming of postural movements: Adaptation to altered support surface configurations: J Neurophysiol, 1986; 55; 1369-81
18. Page P, Frank CC, Lardner R: Assessment and treatment of muscle imbalance: The Janda approach, 2010; 16-22, Illinois, Human Kinetics
19. Dickstin R, Nissan M, Pillar T, Foot-ground pressure pattern of standing hemiplegic patients. Major characteristics and patterns of improvement: Physical Therapy, 2014; 64; 19-23
20. Tyson SF, Hanley M, Chillala J, Balance disability after stroke: Phys Ther, 2006; 86; 30-38
21. Carr JH, Shepherd RB, Stroke rehabilitation: Guideline for exercise and training to optimize motor skill: Butterworth Heiemann, 2000; 1; 3-31
22. Plummer-D’Amato P, Altmann LJ, Saracino D, Interactions between cognitive tasks and gait after stroke: A dual task study: Gait Posture, 2008; 27; 683-88
23. Duncan P, Studenski S, Richards L, Randomized clinical trial of therapeutic exercise in subacute stroke: Stroke, 2003; 34; 2173-80
24. Salbach NM, Mayo NE, Wood-Dauphinee S, A task-orientated intervention enhances walking distance and speed in the first year post stroke: A randomized controlled trial: Clin Rehabil, 2004; 18; 509-19
25. Shummway-Cook A, Woollacott M: Motor Control: Translating research into clinical practice, 2012; 208-9, St West Camden, Lipponcott Wiliams & Sildins
26. Andersson G, Hagman J, Talianzadeh R, Effect of cognitive load on postural control: Brain Res Bull, 2002; 58; 135-39
27. Huxhold O, Li SC, Schmiedek F, Dual-tasking postural control: Aging and the effects of cognitive demand in conjunction with focus of attention: Brain Res Bull, 2006; 69; 294-305
28. Jo SJ, Choi JD, Effects of sensorimotor training based of automatic stabilization on postural control in lower extremity: Master thesis, 2013, Graduate School of Daejeon University
29. Podsiadlo D, Richardson S, The timed “Up & Go”: A test of basic functional mobility for frail elderly persons: J Am Geriatr Soc, 1991; 39; 142-48
30. Lee JJ, Lee HJ, Park J, The Korean version of Berg Balance Scale as an index of activity related to ambulation in subjects with stroke: J Korean Acad Rehabil Med, 2007; 31; 400-3
31. Portney L, Watkins MP: Foundations of clinical research: applications to practice, 2000, Upper Saddle River, NJ, Prentice-Hall, Inc
32. Van Uden CJ, Besser MP, Test-retest reliability of temporal and spatial fait characteristics measured with an instrumented walkway system (GAITRite): BMC Musculoskelet Disord, 2004; 17; 13
33. Brière A, Lauzière S, Gravel D, Perception of Weight-bearing distribution during sit-to-stand tasks in hemiparetic and healthy individuals: Stroke, 2010; 14; 1704-8
34. Jeon HW, Chung YJ, The effect of dual-task on standing postural control in persons with chronic stroke: PTK, 2010; 17; 20-30
35. French B, Thomas L, Leathley M, Does repetitive task training improve functional activity after stroke? A Cochrane systematic review and meta-analysis: J Rehabil Med, 2010; 42; 9-14
36. McCulloch K, Attention and dual-task conditions: Physical therapy implications for individuals with acquired brain injury: J Neurol Phys Ther, 2007; 31; 104-18
37. Subramaniam S, Wan-Ying Hui-Chan C, Bhatt T, A cognitive-balance control training paradigm using Wii Fit to reduce fall risk in chronic stroke survivors: J Neurol Phys Ther, 2014; 38; 216-25
38. Lee IW, Kim YN, Lee DK, Effect of a virtual reality exercise program accompanied by cognitive tasks on the balance and gait of stroke patients: J Phys Ther Sci, 2015; 27; 2175-77
39. Pai YC, Patton J, Center of mass velocity-position predictions for balance control: J Biomech, 1997; 30; 347-54
40. Flynn S, Palama P, Bender A, Feasibility of using Sony PlayStation 2 gaming platform for an individual poststroke: A case report: J Neurol Phys Ther, 2007; 31; 180-89
41. Dusane S, Wang E, Bhatt T, Transfer of reactive balance adaptation from stance-slip perturbation to stance-trip perturbation in chronic stroke survivors: Restor Neurol Neurosci, 2019; 37; 469-82
42. Gerards MHG, McCrum C, Mansfield A, Perturbation-based balance training for falls reduction among older adults: Current evidence and implications for clinical practice: Geriatr Gerontol Int, 2017; 17; 2294-303
43. Alder S, Beckers D, Buck M: PNF in practice: An illustrated guide, 2000, Berlin Heidelberg, Springer
44. Cordova ML, Juttte LS, Hopkins JT, EMG comparison of selected ankle rehabilitation exercises: J Sports Rehabil, 1999; 8; 209-18
45. Davey NJ, Lisle RM, Loxton-Edwards B, Activation of back muscles during voluntary abduction of the contralateral arm in humans: Spine, 2002; 27; 30-34
46. Cichy B, Wilk M, Gait analysis in osteoarthritis of the hip: Med Sci Monit, 2006; 12; CR507-13
47. Mauritz EW, Quinn ME, Seddon PG, Body weight support treadmill and overground ambulation training for two patients with chronic disability secondary to stroke: Phys Ther, 2002; 82; 53-61
48. Michaelsen SM, Ovando AC, Romaguera F, Effect of backward walking treadmill training on walking capacity after stroke: A randomized clinical trial: Int J Stroke, 2014; 9; 529-32
49. Kim CM, Eng JJ, The relationship of lower-extremity muscle torque to locomotor performance in people with stroke: Physical Therapy, 2003; 83; 49-57
50. Krebs DE, Scarborough DM, McGibbon CA: Am J Phys Med Rehabil, 2007; 86; 93-103
51. Drużbicki M, Guzik A, Przysada G, Changes in gait symmetry after training on a treadmill with biofeedback in chronic stroke patients: A 6-month follow-up from a randomized controlled trial: Med Sci Monit, 2016; 22; 4859-68
Most Viewed Current Articles
13 Apr 2020 : Original article 19,586
Outcome of 24 Weeks of Combined Schroth and Pilates Exercises on Cobb Angle, Angle of Trunk Rotation, Chest...DOI :10.12659/MSMBR.920449
Med Sci Monit Basic Res 2020; 26:e920449
20 Apr 2018 : Original article 17,466
Brain Training Games Enhance Cognitive Function in Healthy SubjectsDOI :10.12659/MSMBR.909022
Med Sci Monit Basic Res 2018; 24:63-69
23 Jul 2016 : Review article 10,353
Cardiac Hypertrophy: An Introduction to Molecular and Cellular BasisDOI :10.12659/MSMBR.900437
Med Sci Monit Basic Res 2016; 22:75-79
10 Aug 2020 : Clinical Research 9,788
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Rando...DOI :10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264