01 November 2021: Human Study
Prevalence of Hepatitis B and C Virus Infections: Influence of National Health Care Policies and Local Clinical Practices
Veeravan Lekskulchai1ACDEF*DOI: 10.12659/MSMBR.933692
Med Sci Monit Basic Res 2021; 27:e933692
Abstract
BACKGROUND: BACKGROUND Because reliable epidemiological data are necessary to eliminate hepatitis B and C virus (HBV and HCV) infections, factors influencing their prevalence should be determined. This study aimed to disclose practical issues that affect the prevalence of these viral infections. MATERIAL AND METHODS All medical records with laboratory findings during 2016 to 2018 were reviewed, and all relevant data were extracted. All HBV and HCV infections were followed within these 3 years and investigated in detail. RESULTS The total number of records was 103 197, with a male to female ratio of 1: 1.4. Hepatitis B surface antigen (HBsAg) was tested in 12 934 cases, with a male to female ratio of 1: 2.6. Anti-HCV antibody (anti-HCV Ab) testing was done in 475 cases (53% male). The seroprevalence of HBV and HCV was 5.2% and 4.4%, respectively. Chronic HBV and HCV infections and their life-threatening complication, liver cancer, were highly detected in men aged 41-60 years. CONCLUSIONS HBsAg was highly screened in women owing to the nationwide implementation of the universal HBsAg screening in pregnant women to prevent vertical transmission. Screening for anti-HCV Ab was neglected, probably due to lack of vaccine and high costs of anti-HCV drugs, which most people in low- to middle-income countries generally cannot afford. Local practices under national health care policies and limited budget and resources can cause underestimation of the prevalence of the HBV and HCV infections and persistent transmission of these viruses owing to unidentified cases.
MATERIAL AND METHODS:
RESULTS:
CONCLUSIONS:
Keywords: Hepatitis B, Hepatitis C, Prevalence, Seroepidemiologic Studies, Delivery of Health Care, Female, Health Policy, Hepacivirus, Hepatitis B Surface Antigens, Hepatitis C Antibodies, Humans, Male, Pregnancy
Background
Hepatitis B virus (HBV) and hepatitis C virus (HCV) infections are global public health concerns since they can cause chronic infection and place infected people at high risk of death from cirrhosis and hepatocellular carcinoma (HCC). The World Health Organization (WHO) estimated that 257 million people worldwide were living with chronic hepatitis B infection (CHB), defined as hepatitis B surface antigen (HBsAg) positive, and 71 million people worldwide had chronic HCV infection (CHC), identified by anti-HCV antibody (anti-HCV Ab) positivity, in 2015 [1]. The estimated CHB prevalence was increased to 292 million globally in 2016 [2].
In 2016, the 69th World Health Assembly adopted the Global Health Sector Strategy on Viral Hepatitis 2016–2021. The strategy outlines 5 synergistic prevention and treatment service coverage targets, including the implementation of universal hepatitis B immunization and antiviral treatment of highly viremic mothers infected with HBV during the third trimester to prevent vertical transmission; blood donor screening for HBV and HCV; safe injection practices; stringent infection-control programs to reduce the burden of HBV and HCV infections; and antiviral treatments for patients with HBV and HCV infection to achieve the elimination of viral hepatitis as a public health threat by 2030 (defined as 90% reduction in incidence and 65% reduction in mortality) [3]. However, achieving these goals requires health care policies to address prevention of new infections and dedicated hepatitis programs and budgets, principally for the costs of diagnosis and treatment [3,4]. In resource-limited countries, achieving these goals may be less possible owing to the limited availability of reliable epidemiological data, insufficient public awareness of risk factors and modes of transmission, and financial barriers to treatment and care [5].
Thailand is a Southeast Asian country and one of the HBV and HCV endemic regions [4,6,7]. To reduce the transmission of HBV and HCV, the Thai government has implemented universal hepatitis B immunization for newborns to reduce vertical transmission from infected mothers [8]. The use of healthy volunteer blood donors and the application of nucleic acid amplification technology for testing for HBV and HCV in all blood-donating units have been in place in Thailand for decades [9,10]. However, as a resource-limited country, Thailand may be unable to provide reliable epidemiological data and to eliminate infections in the country by 2030.
This study aimed to determine the prevalence of HBV and HCV infections in the context of a hospital service during 2016 to 2018. The influence of local guidelines and national viral hepatitis prevention programs on epidemiological data of HBV and HCV infections in real clinical practices was evaluated.
Material and Methods
The study was conducted at the HRH Maha Chakri Sirindhorn Medical Centre, Nakhon Na Yok province, Thailand, under the approval of the Srinakharinwirot University Ethics Committee for Human Research. All medical records containing laboratory testing for patients admitted from 2016 to 2018 were collected anonymously. The records were sorted by the patients’ hospital number. Data in the records, including the patients’ sex and age, diagnosis, and laboratory findings, were reviewed, and all relevant data were extracted. All relevant cases were reviewed continuously during these 3 years.
The HBsAg, anti-HBs antibody (anti-HBs Ab), hepatitis B envelope antigen (HBeAg), anti-HCV, and alpha-fetoprotein (AFP) were analyzed by an Architect ci8200 instrument (Abbott Laboratories, Abbott Park, IL, USA). AFP was used as an indicator of HCC. Possible causes of HCC were tracked from previous diagnoses. The HBV viral load was performed by a Cobas TaqMan 48 analyzer in combination with the Cobas AmpliPrep Instrument (Roche Diagnostics, Mannheim, Germany). All tests were conducted according to the manufacturers’ instructions. Positivity for HBsAg, anti-HBs Ab, HBeAg, and anti-HCV Ab were defined at the signal-to-cutoff ratio of >10, >10, >1.0, and >1.0, respectively. The cutoff value for the HBV viral load was set at HBV DNA >10 000 copies/mL and that of AFP was defined at >400 IU/mL.
All statistical analyses were performed by using SPSS Statistics version 19.0 (IBM Corp., Armonk, NY). The categorical variables were compared by chi-square (χ2) test. Statistical significance was defined as
Results
Laboratory investigations were found in 103 197 records; they belonged to patients aged between 1 and 106 years, with a male to female ratio of 1: 1.4 (Table 1). The population in Thailand and in the central part of Thailand where this study was conducted are also presented in Table 1, which shows almost equal numbers of males and females [11]. In contrast, as shown in Table 2, HBsAg testing was significantly higher in females than in males, with a male to female ratio of ~1: 3 (χ2=956;
In practice, HBsAg and anti-HBs Ab were tested simultaneously and the HBV vaccine was only given to individuals with negative test results. As shown in Table 3, the number of positive results for anti-HBs Ab was significantly higher than for HBsAg (χ2=8.365;
Anti-HCV Ab was newly detected in 186 cases, with 72% male prevalence (Figure 2A). CHC was diagnosed in 163 cases (Figure 2B). The seroprevalence of HCV infection, defined as anti-HCV Ab positivity, was 4.4%. HCV infection was predominantly in males, particularly in those aged 41 to 70 years (Figure 2). Overall, there were 349 individuals with HCV infection, and 225 were males. Among people with HCV infection, 19 males and 4 females were co-infected with HIV.
HCC was detected in 142 cases with an average age of 55 years, and 85% of cases were in men (Table 4). The most common cause of HCC in males was CHB, but in females, other causes were more common (Figure 3). HCC was solely diagnosed in 35 cases; neither HBsAg nor anti-HCV Ab was tested in these cases.
Discussion
In 2016 the WHO set the goal to substantially eliminate HBV and HCV infections and their associated mortality by 2030 [3,12], and most countries became a part of the challenge. However, the goals may not be achieved in some countries because there is little funding available for these viruses at an international level, in addition to a lack of dedicated hepatitis budgets or programs in many countries, especially those with limited resources [3,4]. In addition, a lack of reliable epidemiological data can impede achieving the goal [3,5]. The current study shows some clinical practice issues that skew epidemiological data on HBV and HCV infections in Thailand.
Based on the knowledge that mother-to-child transmission is the most important route of HBV infection [13], many countries, including Thailand, have tried to end this route of transmission. The Thai government has deployed universal screening of HBsAg during pregnancy and universal vaccination for newborns of infected mothers for over 20 years. These programs are still running as national health care policies. Consequently, all women receiving care for pregnancy or childbirth undergo HBsAg screening [14]. Implementing universal screening for HBsAg during pregnancy has skewed epidemiological data on HBV infection. As shown in Tables 1 and 2, the Thai population has almost equal numbers of males and females, but HBsAg screening was significantly highly in females; consequently, early HBV infection was highly detected in young women (Figure 1A). As previously reported, the test is infrequently used in the Emergency Department where there are a high number of new patients from various groups, including those at high risk of HBV infection [15]. These practices have led to a missed chance for detecting asymptomatic HBV infection and to underestimation of the prevalence of HBV infection. The HBV seroprevalence found in the current study was 5.2% (Table 3), which is consistent with the estimated prevalence of 5% to 7% for people living in Southeast Asian countries [16]. Since all countries in Southeast Asia face similar HBV problems to Thailand, the prevalence in these countries might be influenced by their health care policies and local practices as well.
Anti-HBs Ab was positive mostly in adults from natural infection (Table 3), indicating that HBV transmission from routes other than vertical transmission was high in the community. According to a previous report, most adults exposed to HBV spontaneously clear the infection [17], which might lead to less individual and public awareness of HBV infection. However, 2–6% of people who get hepatitis B as adults can become chronically infected [18]. Since men in the community were less likely to be screened for HBsAg, it was not surprising to find that they had a higher prevalence of CHB (Figure 1A). Most HBV-infected people are asymptomatic and consequently are unaware that they should get diagnosis and treatment. Without screening, people with unidentified infection can transmit the virus to others for several years before the disease progresses to the symptomatic stage [17,19]. These findings emphasize the need for national screening programs to find individuals who are already infected, both for accurate epidemiological data and to provide proper treatment.
Because HBV shares the same routes of transmission as HCV and HIV, such as sex with an infected partner or sharing blood-contaminated items such as needles for tattooing and body piercings [18], co-infections are possible. In the study community, co-infection with HBV and HIV was detected in 2.7% of cases, which was lower than previously reported rates of 5% to 10% [16,20]. Unlike HBV infection, people are highly aware of HIV infection. Moreover, a number of governmental HIV prevention policies have been implemented for several decades, which may have helped to reduce the incidence of HIV infection in several high-risk groups [21]. Co-infection with HBV and HCV was detected in 1.0% of cases. This rate was also lower than the previous report of 2% to 8% [22]; however, this rate might be inaccurate owing to underutilization of the anti-HCV Ab test.
The screening for anti-HCV Ab was neglected in local clinical practice partly because no prophylactic vaccine and no national health care support were available for HCV prevention. Moreover, because Thailand is a low- to middle-income country, where most test kits and drugs are imported, access to viral hepatitis testing and treatment is costly [3,5]. Even among patients with a diagnosis, only a minority receive treatment [23]. In practice, testing may be used for supporting the diagnosis in symptomatic patients rather than for screening individuals in risk groups, which might consequently lead to unreliable seroprevalence estimates of HCV in this community.
As previously stated, the majority of HCV infections are asymptomatic for years; thus, many individuals are unaware that they are infected and do not seek screening and treatment [19]. However, up to 80% of people infected with HCV are unable to spontaneously eliminate the virus and, without an effective treatment, the infection will progress to CHC [24,25]. Accordingly, most HCV-infected people in this study had CHC and sought diagnosis and treatment at older ages (Figure 2B). HCV co-infection with HIV was detected in 7% of cases, which was lower than previously reported at 42–87% [26]. This result might also be caused by the low anti-HCV Ab screening rate.
HCC was highly detected in males at an average age of 55 years (Table 4), which accorded with previous reports [27–29]. The ratio of male to female HCC cases was 5.8 to 1, which was higher than that found in a previous report of 2.4 to 1 [30]. Additionally, CHB was the most common cause of HCC in males (Figure 3A). Thus, early detection and treatment of HBV in men are necessary to reduce the prevalence of CHB and HCC. CHC caused around 14% of HCC (Figure 3B), which was close to that seen in other regions [27,30]. Therefore, screening for early HCV infection and increasing access to effective anti-HCV drugs are necessary for eliminating HCV infection and liver cancer. In some HCC cases, neither HBsAg nor anti-HCV Ab was tested. According to previous reports, a high number of patients with HCC were diagnosed at an advanced stage, which may preclude curative treatment options [19,29,31] and limit reasons for seeking the cause.
There are some limitations in this study. First, although a high number of individuals were included in the study, the number was still small compared with the total Thai population. Second, high costs of testing and treatment are expected to cause underutilization of anti-HCV Ab tests in this community; the real cause is not known based on the available data. Likewise, due to the limited data available, causes of the high incidences of CHB, CHC, and HCC in men cannot be clearly explained.
Conclusions
Local clinical practices following the national hepatitis programs and with limited resources can skew the epidemiological data on HBV and HCV infections. Screening for HBsAg principally in pregnant women and neglecting to screen for anti-HCV Ab can lead to underestimation of HBV and HCV prevalence. Globally eliminating these 2 hepatitis viruses and their long-term life-threatening complication, HCC, may be impossible as long as unidentified infected people are left in communities.
Figures
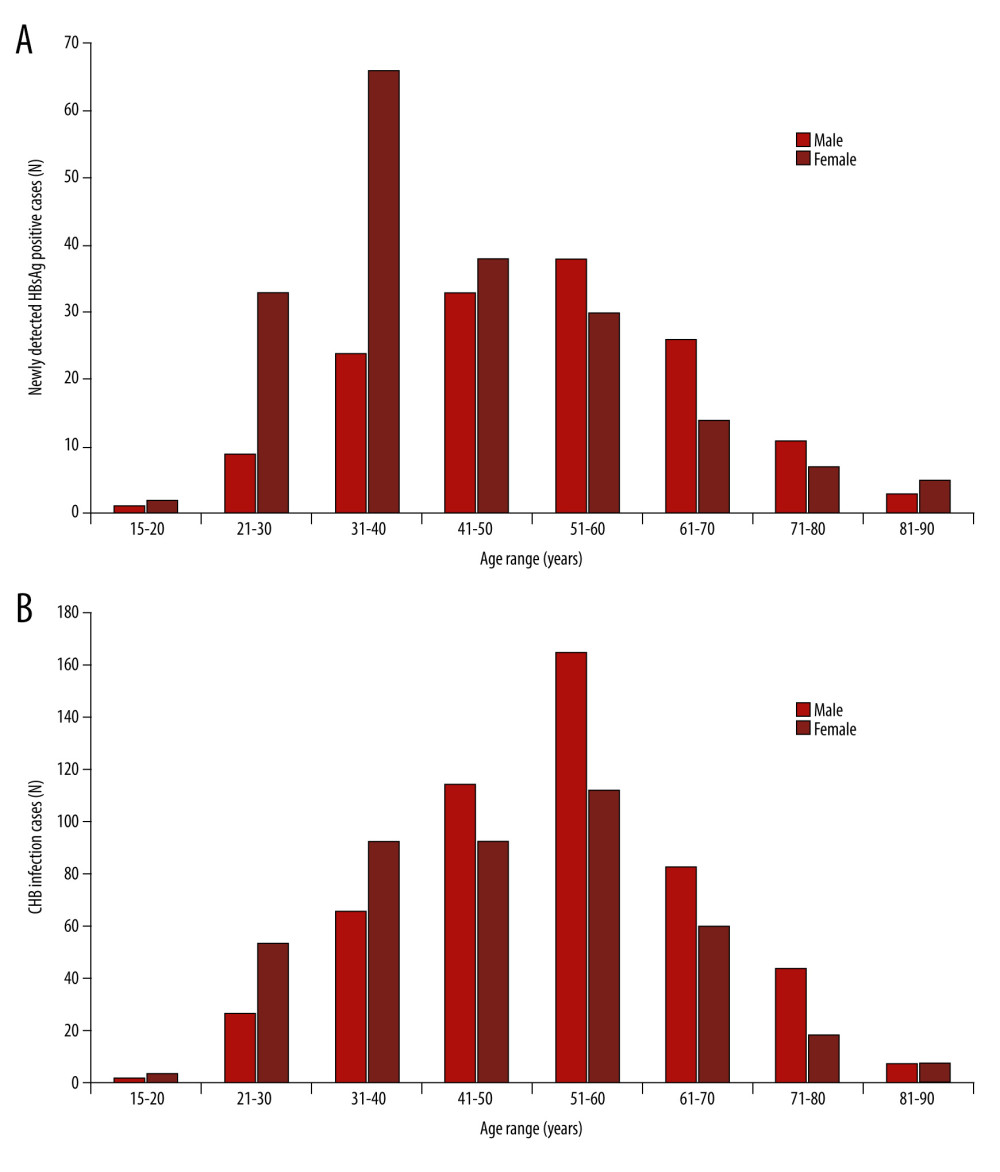 Figure 1. Numbers of viral hepatitis B infection cases based on age range and (A) the number of newly detected hepatitis B surface antigen (HBsAg)-positive cases and (B) the number of chronic hepatitis B (CHB) infection cases. The patients’ ages were at their first detected episode.
Figure 1. Numbers of viral hepatitis B infection cases based on age range and (A) the number of newly detected hepatitis B surface antigen (HBsAg)-positive cases and (B) the number of chronic hepatitis B (CHB) infection cases. The patients’ ages were at their first detected episode. 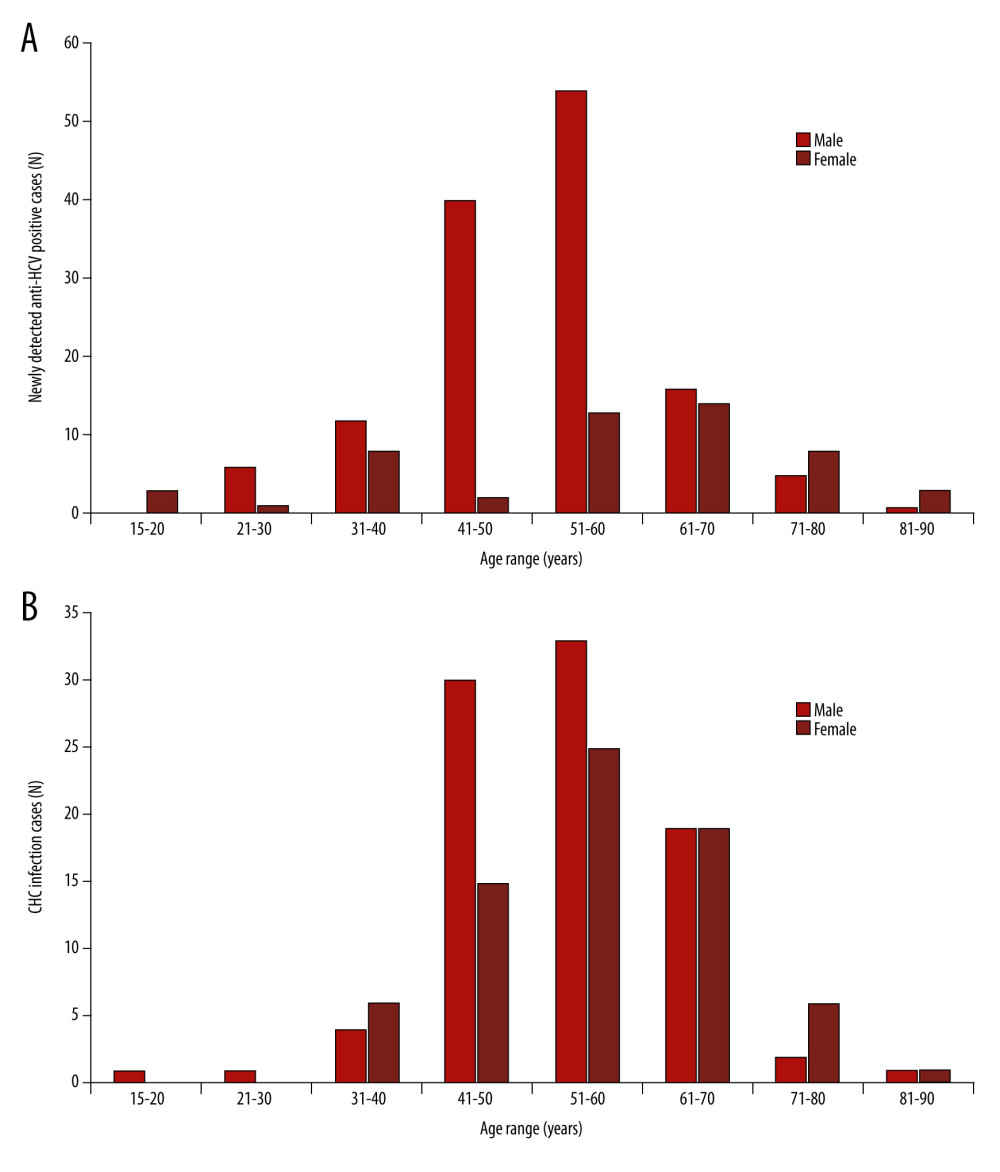 Figure 2. Numbers of viral hepatitis C infection cases based on age range and (A) the number of newly detected hepatitis C virus antibody (anti-HCV)-positive cases and (B) the number of chronic hepatitis C (CHC) infection cases. The patients’ ages were at their first detected episode.
Figure 2. Numbers of viral hepatitis C infection cases based on age range and (A) the number of newly detected hepatitis C virus antibody (anti-HCV)-positive cases and (B) the number of chronic hepatitis C (CHC) infection cases. The patients’ ages were at their first detected episode. 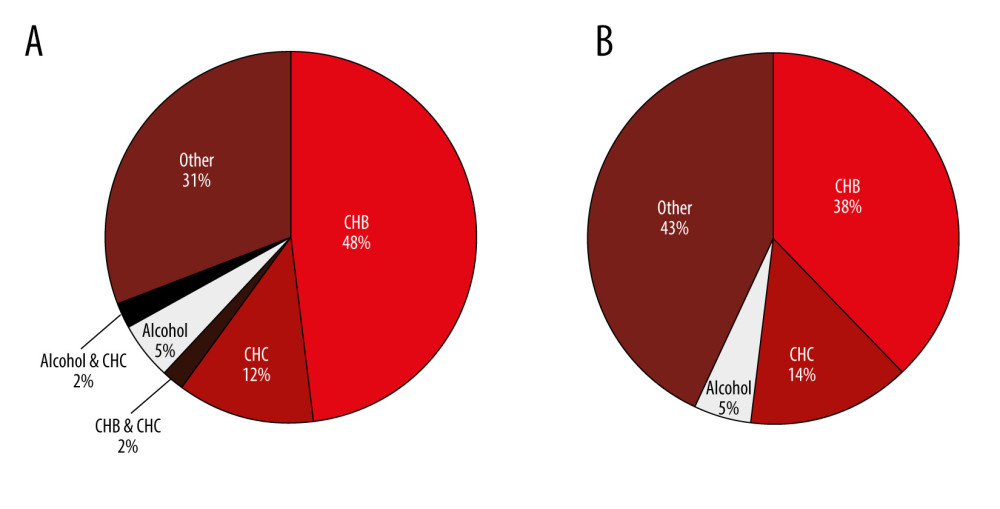 Figure 3. Causes of hepatocellular carcinoma in men (A) and in women (B). CHB – chronic hepatitis B; CHC – chronic hepatitis C.
Figure 3. Causes of hepatocellular carcinoma in men (A) and in women (B). CHB – chronic hepatitis B; CHC – chronic hepatitis C. Tables
Table 1. Sex and age of populations and study cases.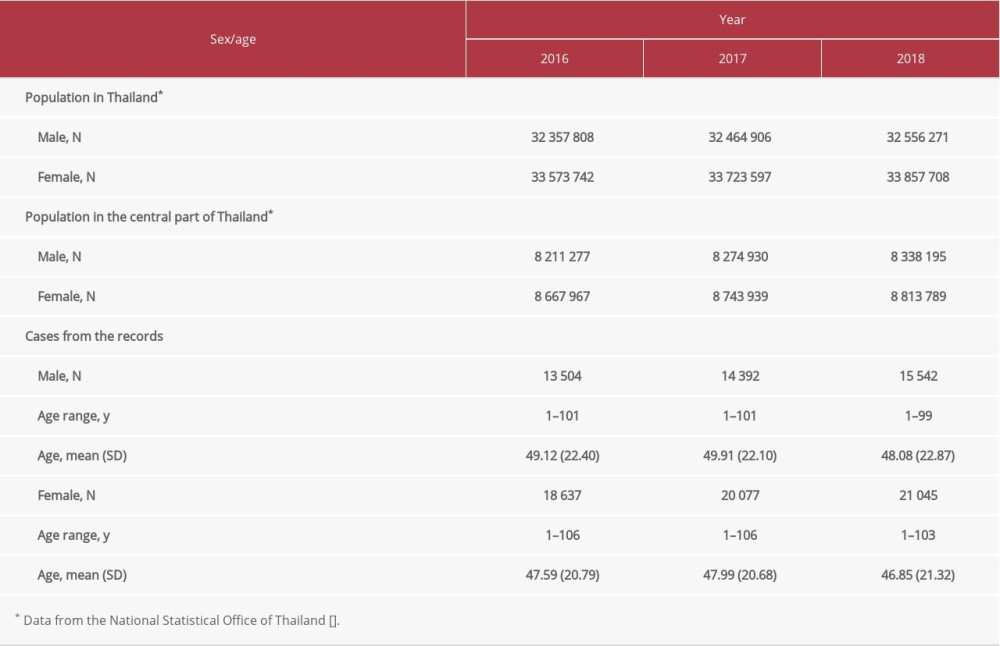 Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).*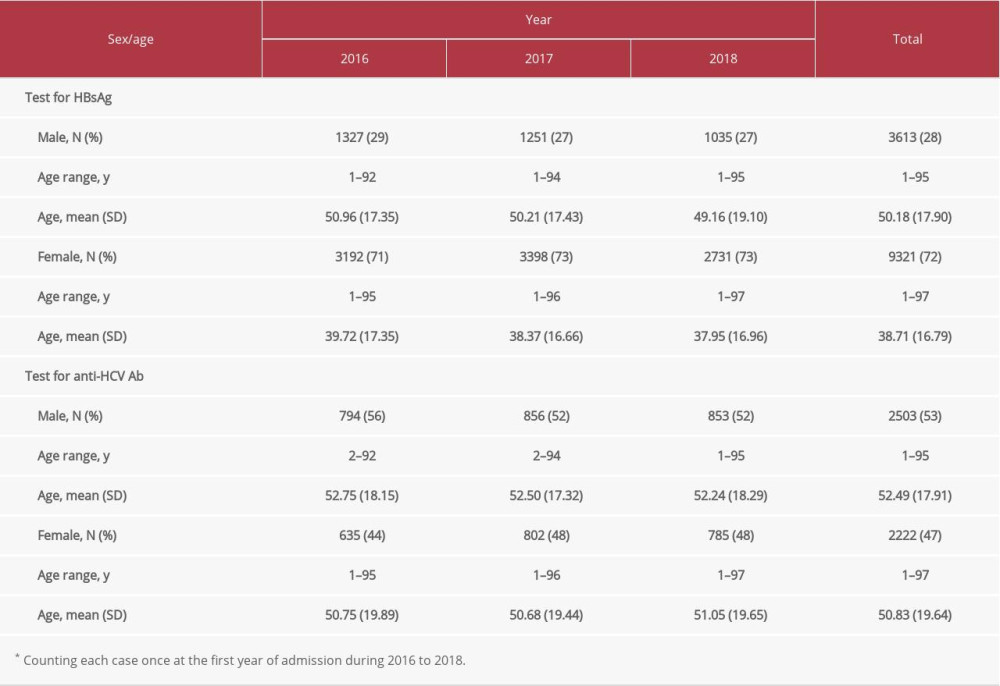 Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).*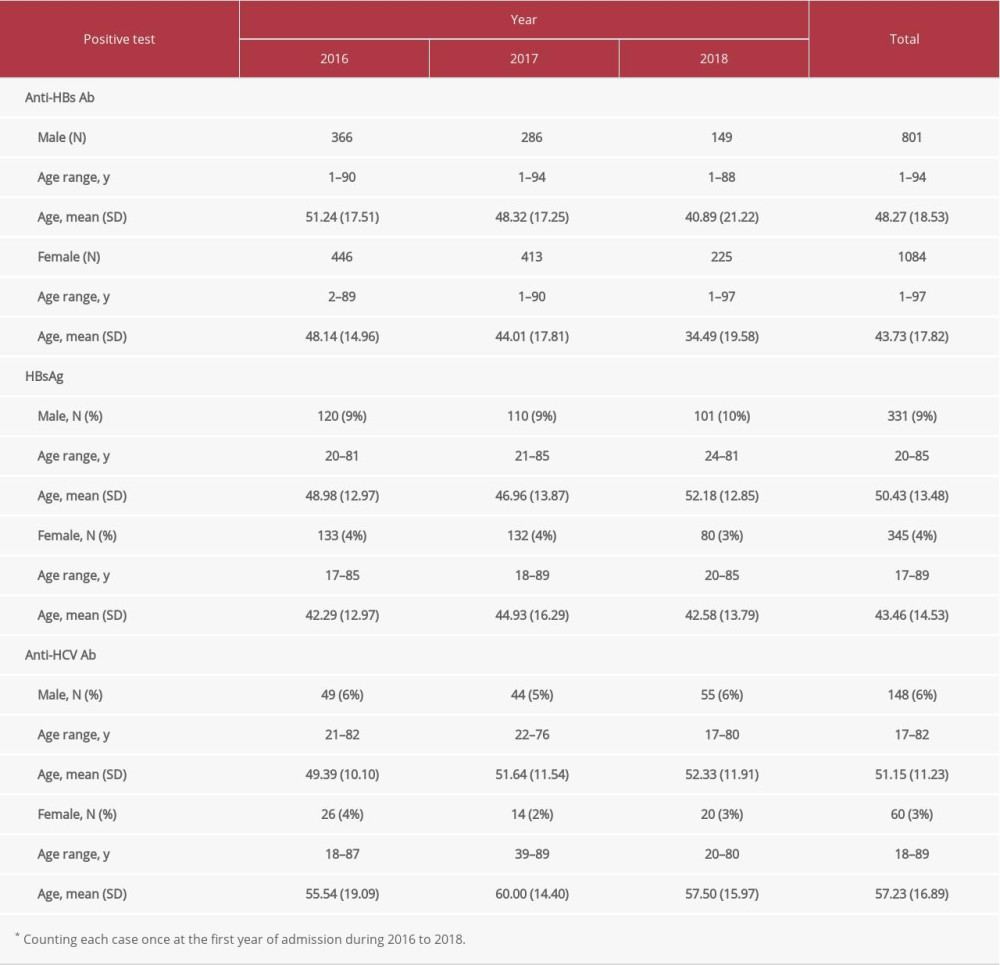 Table 4. Number of hepatocellular carcinoma cases.*
Table 4. Number of hepatocellular carcinoma cases.*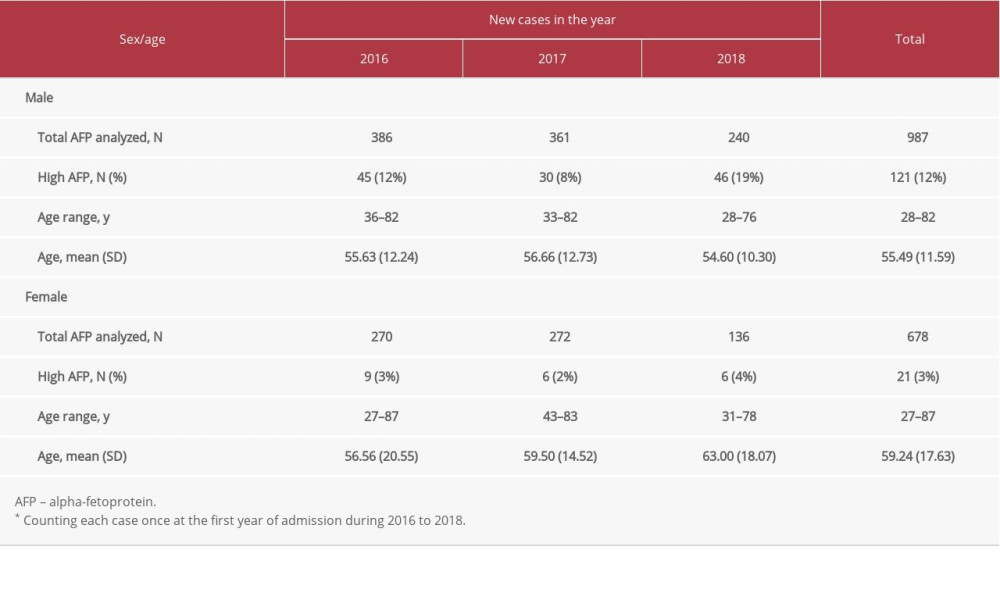
References
1. World Health Organization: Global hepatitis report, 2017 Available from: https://www.who.int/publications/i/item/global-hepatitis-report-2017
2. Polaris Observatory Collaborators, Global prevalence, treatment, and prevention of hepatitis B virus infection in 2016: a modelling study: Lancet Gastroenterol Hepatol, 2018; 3; 383-403
3. Cox AL, El-Sayed MH, Kao JH, Progress towards elimination goals for viral hepatitis: Nat Rev Gastroenterol Hepatol, 2020; 17(9); 533-42
4. Schröeder SE, Pedrana A, Scott N, Innovative strategies for the elimination of viral hepatitis at a national level: A country case series: Liver Int, 2019; 39(10); 1818-36
5. Wait S, Kell E, Hamid S, Hepatitis B and hepatitis C in Southeast and Southern Asia: Challenges for governments: Lancet Gastroenterol Hepatol, 2016; 1(3); 248-55
6. Te HS, Jensen DM, Epidemiology of hepatitis B and C viruses: A global overview: Clin Liver Dis, 2010; 14(1); 1-21
7. Wasitthankasem R, Pimsingh N, Treesun K, Prevalence of hepatitis C virus in an endemic area of Thailand: burden assessment toward HCV elimination: Am J Trop Med Hyg, 2020; 103(1); 175-82
8. Posuwan N, Wanlapakorn N, Sa-Nguanmoo P, The success of a universal hepatitis B immunization program as part of Thailand’s EPI after 22 years’ implementation: PLoS One, 2016; 11(3); e0150499
9. Prouté M, Le Coeur S, Tiv MH, Risky injection practices and HCV awareness in Chiang Mai province, Thailand: A respondent-driven sampling study of people who inject drugs: BMC Public Health, 2020; 20(1); 1450
10. Chimparlee N, Oota S, Phikulsod S, Hepatitis B and hepatitis C virus in Thai blood donors: Southeast Asian J Trop Med Public Health, 2011; 42(3); 609-15
11. National Statistical Office of Thailand: Key statistics at national, regional, and provincial levels, 2021, Bangkok. Thailand, National Statistical Office, Minister of Information and Communication Technology Available from: http://statbbi.nso.go.th/staticreport/Page/en/index.aspx
12. World Health Organization (WHO): Combating hepatitis B and C to reach elimination by 2030, 2016, Geneva, WHO Available from: https://apps.who.int/iris/handle/10665/206453
13. Gentile I, Borgia G, Vertical transmission of hepatitis B virus: Challenges and solutions: Int J Womens Health, 2014; 6; 605-11
14. Posuwan N, Wanlapakorn N, Sintusek P, Towards the elimination of viral hepatitis in Thailand by the year 2030: J Virus Erad 27, 2020; 6(3) 100003
15. Lekskulchai V, Experiences of screening for human immunodeficiency virus, viral hepatitis B, and viral hepatitis C infections at a hospital in Thailand: test utilization and outcomes: Med Sci Monit Basic Res, 2019; 25; 210-17
16. MacLachlan JH, Cowi BC, Hepatitis B virus epidemiology: Cold Spring Harb Perspect Med, 2015; 5; a021410
17. Razavi H, Global epidemiology of viral hepatitis: Gastroenterol Clin North Am, 2020; 49(2); 179-89
18. Centers for Disease Control and Prevention: Viral hepatitis surveillance – United States, 2017, 2019, Atlanta, GA, Centers for Disease Control and Prevention, US Department of Health and Human Services Available from: https://www.cdc.gov/hepatitis/statistics/2017surveillance/index.htm
19. Moore MS, Ivanina E, Bornschlegel K, Hepatocellular carcinoma and viral hepatitis in New York city: Clin Infect Dis, 2016; 63(12); 1577-83
20. Kourtis AP, Bulterys M, Hu DJ, Jamieson DJ, HIV-HBV coinfection – a global challenge: N Engl J Med, 2012; 366; 1749-52
21. Pattanasin S, van Griensven F, Mock PA, Recent declines in HIV infections at Silom Community Clinic Bangkok, Thailand corresponding to HIV prevention scale up: An open cohort assessment 2005–2018: Int J Infect Dis, 2020; 99; 131-37
22. Wu Q, Liu Q, Do hepatitis B virus and hepatitis C virus co-infections increase hepatocellular carcinoma occurrence through synergistically modulating lipogenic gene expression?: Hepatol Res, 2012; 42; 733-40
23. Papatheodoridis G, Hatzakis A, Public health issues of hepatitis C virus infection: Best Pract Res Clin Gastroenterol, 2012; 26(4); 371-80
24. Wasitthankasem R, Posuwan N, Vichaiwattana P, Decreasing hepatitis C virus infection in Thailand in the past decade: evidence from the 2014 national survey: PLoS One, 2016; 11(2); e0149362
25. Chitapanarux T, Phornphutkul K, Risk factors for the development of hepatocellular carcinoma in Thailand: J Clin Transl Hepatol, 2015; 3(3); 182-88
26. Cui Y, Jia J, Update on epidemiology of hepatitis B and C in China: J Gastroenterol Hepatol, 2013; 28(Suppl 1); 7-10
27. Kim BH, Park JW, Epidemiology of liver cancer in South Korea: Clin Mol Hepatol, 2018; 24(1); 1-9
28. Singal AG, Lampertico P, Nahon P, Epidemiology and surveillance for hepatocellular carcinoma: New trends: J Hepatol, 2020; 72(2); 250-61
29. Chonprasertsuk S, Vilaichone RK, Epidemiology and treatment of hepatocellular carcinoma in Thailand: Jpn J Clin Oncol, 2017; 47(4); 294-97
30. Rawla P, Sunkara T, Muralidharan P, Raj JP, Update in global trends and aetiology of hepatocellular carcinoma: Contemp Oncol, 2018; 22(3); 141-50
31. Zhao H, Zhu P, Han T, Clinical characteristics analysis of 1180 patients with hepatocellular carcinoma secondary to hepatitis B, hepatitis C and alcoholic liver disease: J Clin Lab Anal, 2020; 34(2); e23075
Figures
 Figure 1. Numbers of viral hepatitis B infection cases based on age range and (A) the number of newly detected hepatitis B surface antigen (HBsAg)-positive cases and (B) the number of chronic hepatitis B (CHB) infection cases. The patients’ ages were at their first detected episode.
Figure 1. Numbers of viral hepatitis B infection cases based on age range and (A) the number of newly detected hepatitis B surface antigen (HBsAg)-positive cases and (B) the number of chronic hepatitis B (CHB) infection cases. The patients’ ages were at their first detected episode. Figure 2. Numbers of viral hepatitis C infection cases based on age range and (A) the number of newly detected hepatitis C virus antibody (anti-HCV)-positive cases and (B) the number of chronic hepatitis C (CHC) infection cases. The patients’ ages were at their first detected episode.
Figure 2. Numbers of viral hepatitis C infection cases based on age range and (A) the number of newly detected hepatitis C virus antibody (anti-HCV)-positive cases and (B) the number of chronic hepatitis C (CHC) infection cases. The patients’ ages were at their first detected episode. Figure 3. Causes of hepatocellular carcinoma in men (A) and in women (B). CHB – chronic hepatitis B; CHC – chronic hepatitis C.
Figure 3. Causes of hepatocellular carcinoma in men (A) and in women (B). CHB – chronic hepatitis B; CHC – chronic hepatitis C. Tables
 Table 1. Sex and age of populations and study cases.
Table 1. Sex and age of populations and study cases. Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).* Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).* Table 4. Number of hepatocellular carcinoma cases.*
Table 4. Number of hepatocellular carcinoma cases.* Table 1. Sex and age of populations and study cases.
Table 1. Sex and age of populations and study cases. Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 2. Utilization of the tests for hepatitis B surface antigen (HBsAg) and anti-hepatitis C virus antibody (anti-HCV Ab).* Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).*
Table 3. Positivity of anti-hepatitis B surface antibody (anti-HBs Ab), hepatitis B surface antigen (HBsAg), and anti-hepatitis C virus antibody (anti-HCV Ab).* Table 4. Number of hepatocellular carcinoma cases.*
Table 4. Number of hepatocellular carcinoma cases.* Most Viewed Current Articles
13 Apr 2020 : Original article 19,586
Outcome of 24 Weeks of Combined Schroth and Pilates Exercises on Cobb Angle, Angle of Trunk Rotation, Chest...DOI :10.12659/MSMBR.920449
Med Sci Monit Basic Res 2020; 26:e920449
20 Apr 2018 : Original article 17,466
Brain Training Games Enhance Cognitive Function in Healthy SubjectsDOI :10.12659/MSMBR.909022
Med Sci Monit Basic Res 2018; 24:63-69
23 Jul 2016 : Review article 10,353
Cardiac Hypertrophy: An Introduction to Molecular and Cellular BasisDOI :10.12659/MSMBR.900437
Med Sci Monit Basic Res 2016; 22:75-79
10 Aug 2020 : Clinical Research 9,788
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Rando...DOI :10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264








