01 June 2022: Laboratory Research
Comparison of Sealing Abilities Among Zinc Oxide Eugenol Root-Canal Filling Cement, Antibacterial Bioceramic Paste, and Epoxy Resin, using as a Microbial Tracer
Tringa Kelmendi1ABCE, Ferit Koçani1D, Arsim Kurti2G, Blerim Kamberi1DF, Anila Kamberi3E*DOI: 10.12659/MSMBR.936319
Med Sci Monit Basic Res 2022; 28:e936319
Abstract
BACKGROUND: This study aimed to compare sealing abilities among 3 root canal sealers, using Enterococcus faecalis as a microbial tracer.
MATERIAL AND METHODS: Sixty-six straight single-rooted teeth were subjected to crown removal at the cementoenamel junction. Root canals were instrumented using the crown-down technique. Prepared teeth were randomly divided into 3 groups of 22 teeth each, according to the sealer used: Canason zinc oxide eugenol root-canal filling cement, Well-Root ST antibacterial bioceramic paste, and AH 26 epoxy resin. Root canals were obturated using the lateral condensation technique, then stored at 37°C and 100% humidity for 1 week to allow sealers to set completely. Subsequently, bacterial leakage was tested using a 2-chamber method. Twice weekly, fresh E. faecalis was placed into the coronal portion of the filled root canal. Specimens were monitored at 24-h intervals for 33 days. The number of leaking samples and mean interval until leakage were compared among groups.
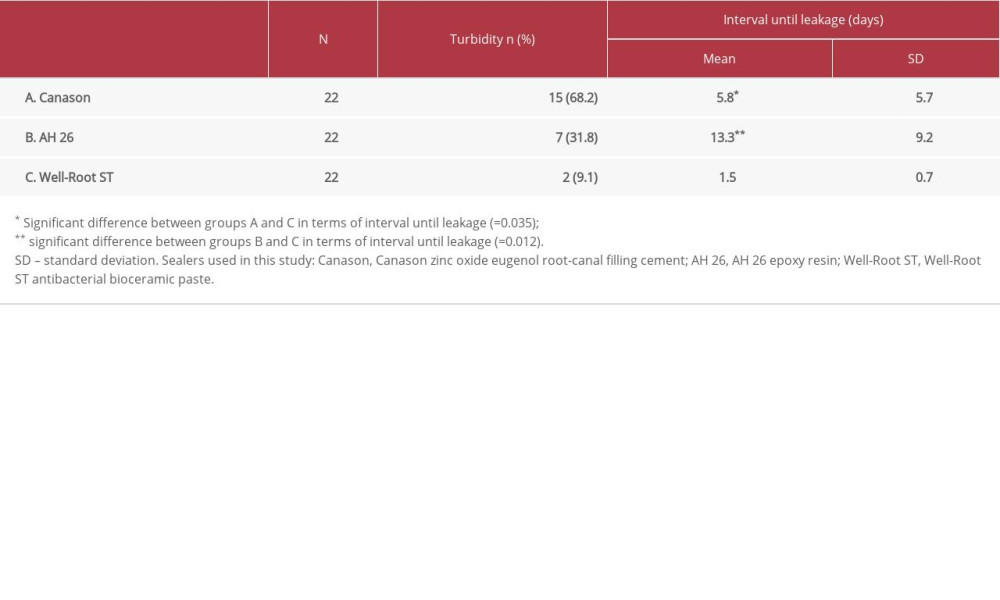
RESULTS: The mean intervals until leakage were 5.8 days in the Canason group, 1.5 days in the Well-Root ST group, and 13.3 days in the AH 26 group. These mean intervals significantly differed between the Canason and Well-Root ST groups (P=0.035) and between the AH 26 and Well-Root ST groups (P=0.012). After 33 days, survival rates were 31.8% in the Canason group, 90.9% in the Well-Root ST group, and 68.2% in the AH 26 group (P<0.001).
CONCLUSIONS: Antibacterial bioceramic paste is a significantly more effective root canal sealer than zinc oxide eugenol root-canal filling cement or epoxy resin.
Keywords: Epoxy Resin AH-26, Calcium Silicate, Zinc Oxide-Eugenol Cement, Anti-Bacterial Agents, Enterococcus faecalis, Epoxy Resins, Eugenol, Root Canal Filling Materials, Root Canal Preparation, Zinc Oxide
Background
Root canal preparation is dependent on the chemomechanical procedure, microbial control, and complete obturation of the root canal system [1]. However, complete disinfection of the root canal cannot be achieved during mechanical instrumentation and antibacterial irrigation because of the complex anatomy of the root canal system [2]. Accordingly, some bacteria remain in root dentinal tubules [3].
The goal of endodontic treatment is to achieve a bacteria-free canal [4]. Wu et al [5] reported that residual bacteria could be effectively entombed in the canal system through the use of root canal obturation materials. However, Tabassum et al [6] reported that bacterial leakage into an obturated root canal may lead to endodontic treatment failure. A well-obturated root canal is critical for long-term endodontic treatment success [7,8]. Obturation materials should be biocompatible. Although gutta-percha is not the ideal filling material, it is recommended because of its biologically inert nature [7].
In vitro approaches used to evaluate the sealing abilities of root filling materials include dye extraction methods, fluid filtration, radioisotopes, and methods that involve bacterial growth [9,10]. Bacterial microleakage assessment is considered more clinically and biologically accurate than other methods [11]. In 2014, Razavian et al [12] reported that Fillapex (bioceramic sealer) exhibited greater leakage than did AH 26 (resin-based) sealer. Similarly, Trivedi et al [13] found that epoxy resin-based sealer demonstrated better sealing ability than did bioceramic sealer. Jayalatha et al [14] reported that AH 26 had better sealing ability than did zinc oxide eugenol. Zmener et al [15] showed that AH 26 had better sealing ability than did AH Plus (resin-based sealer). Jafari et al [16] compared bacterial leakage resistances among AH 26, MTA Fillapex, and Apatite Root Canal Sealer (bioceramic sealer); AH 26 demonstrated the lowest bacterial leakage of the 3 sealers.
Canason zinc oxide eugenol root-canal filling cement (VOCO GmbH, Cuxhaven, Germany) is a zinc oxide eugenol sealer. Zinc oxide eugenol sealers have been widely used by clinicians because of their antimicrobial properties; however, they exhibit slow setting time, poor solubility, and considerable shrinkage upon setting [17,18]. Well-Root ST antibacterial bioceramic paste (Vericom, Gangwon-do, Korea) is a non-toxic hydraulic calcium silicate cement that serves as a bioceramic sealer. This hydrophilic sealer utilizes moisture within the canal to complete the setting reaction. It exhibits antimicrobial properties during the setting reaction, and it does not shrink upon setting. Thus far, there are no published data concerning the physical properties and chemical composition of Well-Root ST [19].
To the best of our knowledge, there have been few studies of coronal leakage involving Canason and Well-Root ST sealers; however, other zinc oxide eugenol and bioceramic sealers have been investigated. Additionally, AH 26 epoxy resin (Dentsply Maillefer, Ballaigues, Switzerland) is a widely used epoxy resin-based sealer that exhibits low solubility and long-term dimensional stability [20]. Among the existing root canal obturation techniques, the most common technique is lateral condensation; this technique is taught by most dental schools and frequently used by general dentists [21,22], primarily because of its ability to control the placement of gutta-percha in the root canal.
In this study, we aimed to compare sealing abilities among zinc oxide eugenol root-canal filling cement, antibacterial bioceramic paste, and epoxy resin, using
Material and Methods
ETHICS STATEMENT:
The study protocol was approved by the Ethics Commission (Nr. 3829/2017). Because the study was performed using extracted teeth, the Ethics Commission did not require patient consent.
STUDY SAMPLES:
This comparative experimental study involved 66 extracted teeth (3 groups of 22 teeth each).
SAMPLE PROCESSING:
Sixty-six freshly extracted human single-rooted mandibular premolars (with similar dimensions [18–20 mm] and straight canals) were selected and stored in sterile saline at room temperature until use. All teeth had intact coronal surfaces with closed apices. Calculus and soft tissues were removed from the root surfaces; teeth were then decoronated at the cementoenamel junction using a diamond disc under continuous water spray. The working length was determined by insertion of a size 10 K-file (Dentsply Maillefer, Ballaigues, Switzerland) into the root canal until the tip was slightly visible at the major apical foramen. The canals were prepared 1 mm shorter than this length. Only teeth with apices that permitted entry of the initial size 10 K-file were included in the experiment.
ProTaper files (Dentsply Maillefer) were used to clean and shape the root canal with a torque motor at 250 rpm. During preparation and after each file had been applied, 1 mL of 0.5% sodium hypochlorite (NaOCl; Chloraxid, Cerkamed, Stalowa Wola, Poland) was used as an irrigant. The following ProTaper files were used: S1, S2, and the finishing files F1 through F3. When instrumentation had been completed, all specimens were filled with 17% ethylenediaminetetraacetic acid (Endo-Solution, Cerkamed) for 1 min; a final rinse was then performed with 1% NaCl to eliminate any residual chemical effects of irrigating solutions, and the canals were dried with absorbent paper points (Meta Biomed Co., Ltd., Cheougja City, Chungbuk, Korea). After instrumentation, the teeth were divided into 3 groups (22 teeth per group). Canals in the Canason group were obturated with Canason zinc oxide eugenol root-canal filling cement (VOCO GmbH, Cuxhaven, Germany); canals in the AH 26 group were obturated with AH 26 epoxy resin (Dentsply Maillefer); and canals in the Well-Root ST group were obturated with Well-Root ST antibacterial bioceramic paste (Vericom, Gangwon-do, Korea).
Using the lateral condensation technique, root canals were obturated with gutta-percha cones (Dentsply Maillefer); a size 40 master cone was applied. The master gutta-percha cone was coated and inserted into the canal in a single movement. To achieve optimal lateral condensation, a size B finger spreader (Dentsply Maillefer) was used, and size 15 accessory cones were applied to a level approximately 1 mm short of the working length. Excess gutta-percha was removed from the orifice of the root canal using a heat carrier. Specimens were stored at 37°C and 100% humidity for 1 week to allow the sealers to set completely. The 1-week interval was based on our clinical experience regarding the length of time for application of final dental restorations.
All canal preparation and sealer application methods used in this study are standard clinical protocol in our department.
LEAKAGE APPARATUS PREPARATION:
A dual chamber device was created by modification of a 20-mL scintillation vial; a bacterial leakage model with an upper chamber and a lower chamber was established using the methods described by Torabinejad et al [23]. In the center of each plastic cap, a hole was created to enable a root to snugly fit into the plastic cap of the container. All procedures were performed in a biosafety cabinet. Tryptic soy broth was aseptically placed into the container to a level of nearly 2 mm above the root end. E. faecalis ATCC 51299 (Liofilchem, Roseto degli Abruzzi, Italy) was cultured at 37°C for 24 h. Aliquots of tryptic soy broth (0.5 mL) containing cultures of E. faecalis were added to upper chambers using a micropipette (Figure 1).
Fresh E. faecalis aliquots were added to upper chambers twice weekly to ensure bacterial viability throughout the experiment. The leakage apparatuses were incubated at 37°C for 33 days. The broth in each container was visually evaluated at 24-h intervals to identify turbidity, which indicated that leakage had occurred throughout the filled root canal. The presence of turbidity in the lower chamber was presumed to indicate a broken seal. Blood agar cultures were used to confirm the presence of bacteria in the lower chamber. Bacteriological assessment of the lower chamber revealed E. faecalis growth in all turbid broth (Figure 2).
STATISTICAL ANALYSIS:
Data were analyzed using SPSS software, version 22.0 (IBM Corp, Armonk, NY, USA). The chi-squared test was used to compare frequencies and percentages of turbidity among groups. The Mann-Whitney U test was used to compare intervals until leakage among groups.
Results
The findings are summarized in Table 1. The number of specimens with turbid broth was greatest in the Canason group (68.2%) and was lowest in the Well-Root ST group (9.1%). The mean intervals until leakage (± standard deviation) were 5.8±5.7 days in the Canason group, 1.5±0.7 days in the Well-Root ST group, and 13.3±9.2 days in the AH 26 group. These mean intervals significantly differed between the Canason and Well-Root ST groups (
The cumulative percentages of leaking specimens over time are shown in Figure 3. After 33 days, survival rates were 31.8% in the Canason group, 90.9% in the Well-Root ST group, and 68.2% in the AH 26 group (
Discussion
Successful endodontic treatment is based on 3-dimensional canal obturation after elimination of all bacteria from the root canal to prevent microleakage and bacterial entry. An ideal root canal sealer should have antimicrobial properties and should be able to seal the root canal system. Multiple studies have shown that calcium silicate-based materials have better sealing abilities than other root canal sealers [24,25]. However, zinc oxide eugenol-based sealers also have antimicrobial activity.
We found significant differences among the 3 groups in terms of the number of specimens with turbid broth. Bacterial entry was greatest in the Canason group, was reduced in the AH 26 group, and was lowest in the Well-Root ST group. The Canason group presumably exhibited greater leakage than the AH 26 group because the creep capacity and long setting time of AH 26 enabled it to more fully penetrate micro-irregularities; this ability enhances mechanical interlocking between sealer and root dentin. Moreover, AH 26 has low solubility and minimal expansion while setting and bonds to root dentin via adamantine [26].
The lateral condensation of gutta-percha is the most widely used obturation method because of advantages such as long-term use, predictability, controlled placement of material, and relative ease of use [27]. Various methods can be used to evaluate the coronal sealing abilities of root canal sealers. In this study, we evaluated leakage in endodontically treated single-rooted teeth using 3 endodontic sealers (Well-Root ST antibacterial bioceramic paste, Canason zinc oxide eugenol root-canal filling cement, and AH 26 epoxy resin sealer); we applied the lateral condensation technique and performed leakage analysis by means of in vitro bacterial tracing methodology. Bacterial tracing is a useful analysis method because bacterial leakage has clinical relevance in the oral cavity; specifically, leakage can occur in endodontically treated teeth [28]. Here, we used
Our results were consistent with the findings by Barnett et al [32] in that zinc oxide eugenol sealer exhibited greater coronal leakage than did other endodontic sealers. Another study [33] demonstrated that samples obturated by lateral condensation with zinc oxide eugenol sealer exhibited complete bacterial microleakage within 90 days. According to Jayalatha et al [14] and Almeida et al [34], AH 26 demonstrated less leakage than did zinc oxide eugenol sealer. Haïkel et al [35] reported a similar finding, whereby AH 26 demonstrated better sealing ability than did a zinc oxide eugenol-based root canal sealer; notably, the zinc oxide eugenol-based root canal sealer exhibited substantial leakage after 6 days, presumably because of 3-dimensional instability.
Timpawat et al [11] found that AH 26 was more adaptable and better able to prevent leakage than were Ketac-Endo (glass-ionomer) and Apexit (calcium hydroxide) sealers. The resin-based AH 26 sealer is regarded as a standard sealer in modern endodontics. In the present study, AH 26 did not completely resist bacterial leakage. These results agreed with the findings in previous studies [36,37], which showed coronal leakage of bacteria and the entry of bacterial products into root canals that had been obturated with AH 26. Furthermore, Wong et al [38] compared multiple types of sealers and found that no sealer provided 100% sealing ability, and the ability significantly differed between a zinc oxide eugenol-based sealer and a resin-based sealer. In contrast to our results, Wong et al reported that the resin-based sealer had the highest microleakage values; they also found that all sealers had greater leakage on the apical part of the root canal, rather than the coronal part.
In this study, Well-Root ST demonstrated less microleakage, compared with Canason or AH 26. Mobarak et al [39] investigated the coronal microleakage of endodontically treated teeth that had been exposed to bacterial contamination and reported that bioceramic sealer provided a better coronal seal than did other endodontic tested materials. Additionally, bioceramic sealers are biocompatible, have antimicrobial activity, and demonstrate better chemical and mechanical properties [39,40].
The results of this study agree with the findings by Pawar et al [41], Sultana et al [42], and Ballullaya et al [43]; those studies used the dye penetration method and revealed that calcium silicate-based sealers had lower leakage values. Ersahan and Aydin [24] found that a calcium silicate-based root canal sealer and an epoxy resin-based sealer demonstrated lower apical leakage values, compared with calcium hydroxide and methacrylate resin; these results were consistent with our findings. Our results differ from the findings by Razavian et al [12], who reported that AH 26 permitted less bacterial leakage than did MTA Fillapex. The results of our study also differed from findings by Trivedi et al [13] and Jafari et al [16], who found that the sealing ability of AH 26 was better than the sealing ability of MTA Fillapex. These discrepancies among studies might be related to differences in sealers selected and microleakage evaluation techniques applied.
There were some limitations in this study. First, bacterial tracing was performed using a single type of microorganism. Second, the sample size was small. Additionally, bacterial leakage assays have an important limitation with respect to clinical relevance: leakage into an obturated root canal does not always cause periapical inflammation [44]. The onset of inflammation depends on the quantity and virulence of the bacteria involved, as well as the immunologic condition of the host. Third, we did not test the use of a glass ionomer barrier to prevent leakage after root canal filling. However, a failed, defective, or inadequate coronal restoration can allow coronal leakage; thus, it is important to test the bacterial leakage effectiveness of root canal sealers, regardless of glass ionomer barrier application. Nonetheless, our results provide important evidence that root canal obturation with Canason zinc oxide eugenol root-canal filling cement exhibits lower resistance to bacterial leakage, compared with AH 26 epoxy resin or Well-Root ST antibacterial bioceramic paste.
Conclusions
Our findings indicate that antibacterial bioceramic paste is a significantly more effective root canal sealer than zinc oxide eugenol root-canal filling cement or epoxy resin.
Figures
 Figure 1. Micropipette-mediated inoculation of upper chambers with Enterococcus faecalis.
Figure 1. Micropipette-mediated inoculation of upper chambers with Enterococcus faecalis.  Figure 2. Confirmation of Enterococcus faecalis presence in lower chamber broth after growth on blood agar.
Figure 2. Confirmation of Enterococcus faecalis presence in lower chamber broth after growth on blood agar. 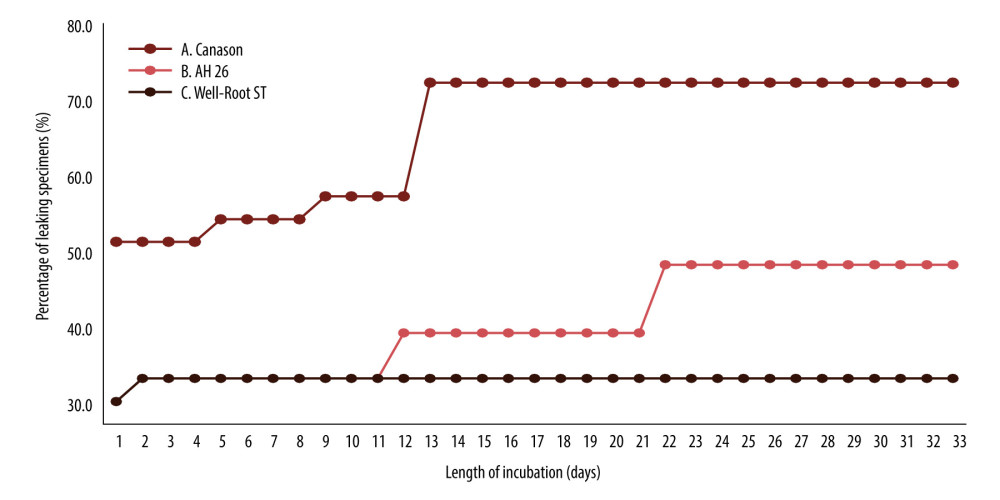 Figure 3. Cumulative percentages of leaking specimens over time. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste.
Figure 3. Cumulative percentages of leaking specimens over time. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste. 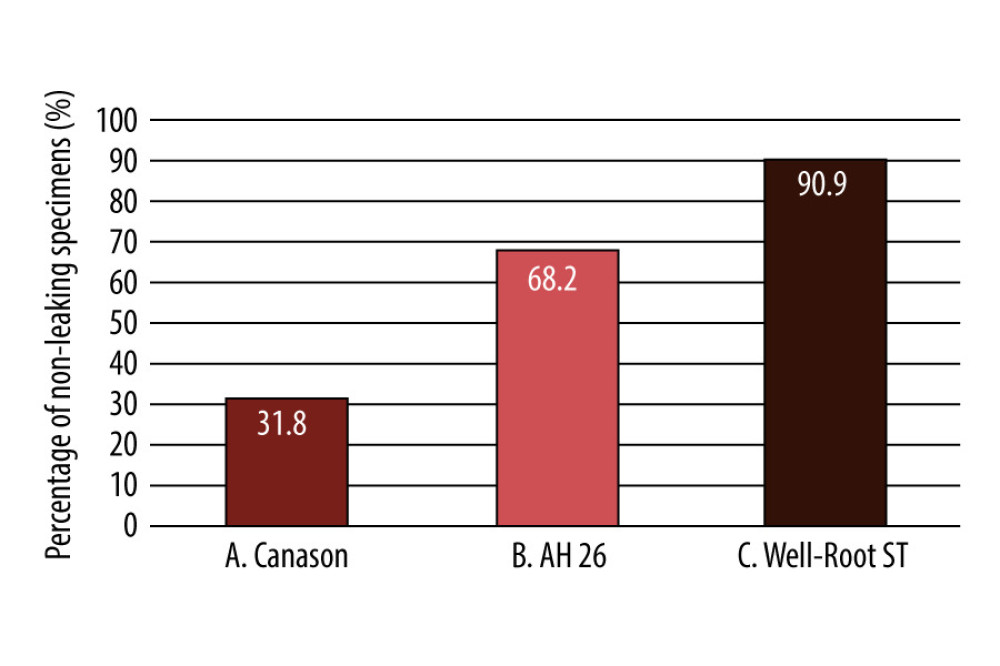 Figure 4. Percent of non-leaking samples (ie, percent survival) during the 33-day experiment. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste.
Figure 4. Percent of non-leaking samples (ie, percent survival) during the 33-day experiment. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste. References
1. Siqueira JF, Rôças IDN, Marceliano-Alves MF, Pérez AR, Ricucci D, Unprepared root canal surface areas: Causes, clinical implications, and therapeutic strategies: Braz Oral Res, 2018; 32(Suppl 1); e65
2. Kandaswamy D, Venkateshbabu N, Gogulnath D, Kindo AJ, Dentinal tubule disinfection with 2% chlorhexidine gel, propolis, morinda citrifolia juice, 2% povidone iodine, and calcium hydroxide: Int Endod J, 2010; 43(5); 419-23
3. Peters LB, Wesselink PR, Moorer WR, The fate and the role of bacteria left in root dentinal tubules: Int Endod J, 1995; 28(2); 95-99
4. Kawashima N, Wadachi R, Suda H, Root canal medicaments: Int Dent J, 2009; 59(1); 5-11
5. Wu MK, Dummer PM, Wesselink PR, Consequences of and strategies to deal with residual post-treatment root canal infection: Int Endod J, 2006; 39(5); 343-56
6. Tabassum S, Khan FR, Failure of endodontic treatment: the usual suspects: Eur J Dent, 2016; 10(1); 144-47
7. Li GH, Niu LN, Selem LC, Quality of obturation achieved by an endodontic core-carrier system with crosslinked gutta-percha carrier in single-rooted canals: J Dent, 2014; 42(9); 1124-34
8. Goldfein J, Speirs C, Finkelman M, Amato R, Rubber dam use during post placement influences the success of root canal-treated teeth: J Endod, 2013; 39(12); 1481-84
9. Pommel L, About I, Pashley D, Camps J, Apical leakage of four endodontic sealers: J Endod, 2003; 29(3); 208-10
10. Wu MK, De Gee AJ, Wesselink PR, Fluid transport and dye penetration along root canal fillings: Int Endod J, 1994; 27(5); 233-38
11. Timpawat S, Amornchat C, Trisuwan WR, Bacterial coronal leakage after obturation with three root canal sealers: J Endod, 2001; 27(1); 36-39
12. Razavian H, Barekatain B, Shadmehr E, Bacterial leakage in root canals filled with resin-based and mineral trioxide aggregate-based sealers: Dent Res J (Isfahan), 2014; 11(5); 599-603
13. Trivedi S, Chhabra S, Bansal A, Evaluation of sealing ability of three root canal sealers: An in vitro study: J Contemp Dent Pract, 2020; 21(3); 291-95
14. Jayalatha NS, Kuriakose S, Saradadevi KL, Comparison of sealing ability of three different root canal sealers – an in vitro study: J Indian Soc Pedod Prev Dent, 1998; 16(4); 122-27
15. Zmener O, Spielberg C, Lamberghini F, Rucci M, Sealing properties of a new epoxy resin-based root-canal sealer: Int Endod J, 1997; 30(5); 332-34
16. Jafari F, Sobhani E, Samadi-Kafil H, In vitro evaluation of the sealing ability of three newly developed root canal sealers: A bacterial microleakage study: J Clin Exp Dent, 2016; 8(5); e561-65
17. Allan NA, Walton RC, Schaeffer MA, Setting times for endodontic sealers under clinical usage and in vitro conditions: J Endod, 2001; 27(6); 421-23
18. Krastl G, Allgayer N, Lenherr P, Tooth discoloration induced by endodontic materials: A literature review: Dent Traumatol, 2013; 29(1); 2-7
19. Reszka P, Nowicka A, Lipski M, A comparative chemical study of calcium silicate-containing and epoxy resin-based root canal sealers: Biomed Res Int, 2016; 2016; 9808432
20. Azadi N, Fallahdoost A, Mehrvarzfar P, A four-week solubility assessment of AH-26 and four new root canal sealers: Dent Res J (Isfahan), 2012; 9(1); 31-35
21. Cailleteau JG, Mullaney TP, Prevalence of teaching apical patency and various instrumentation and obturation techniques in United States dental schools: J Endod, 1997; 23(6); 394-96
22. Levitan ME, Himel VT, Luckey JB, The effect of insertion rates on fill length and adaptation of a thermoplasticized gutta-percha technique: J Endod, 2003; 29(8); 505-8
23. Torabinejad M, Ung B, Kettering JD, In vitro bacterial penetration of coronally unsealed endodontically treated teeth: J Endod, 1990; 16(12); 566-69
24. Ersahan S, Aydin C, Solubility and apical sealing characteristics of a new calcium silicate-based root canal sealer in comparison to calcium hydroxide-, methacrylate resin- and epoxy resin-based sealers: Acta Odontol Scand, 2013; 71(3–4); 857-62
25. Asawaworarit W, Yachor P, Kijsamanmith K, Vongsavan N, Comparison of the apical sealing ability of calcium silicate-based sealer and resin-based sealer using the fluid-filtration technique: Med Princ Pract, 2016; 25(6); 561-65
26. Lee KW, Williams MC, Camps JJ, Pashley DH, Adhesion of endodontic sealers to dentin and gutta-percha: J Endod, 2002; 28(10); 684-88
27. Bakhtiar H, Heidari N, Mehrvarzfar P, In vitro comparative study of the microbial leakage of one-step, thermafil and lateral condensation techniques: J Contemp Dent Pract, 2012; 13(1); 27-30
28. Mathur R, Sharma M, Sharma D, Evaluation of coronal leakage following different obturation techniques and in-vitro evaluation using methylene blue dye preparation: J Clin Diagn Res, 2015; 9(12); ZC13-17
29. Rôças IN, Siqueira JF, Santos KR: J Endod, 2004; 30(5); 315-20
30. Kelmendi T, Kocani F, Krasniqi B, Coronal leakage of two different root canal sealers: J Int Dent Med Res, 2020; 13(1); 128-33
31. Murad CF, Sassone LM, Souza MC: RSBO, 2012; 9(2); 143-50
32. Barnett F, Trope M, Rooney J, Tronstad L, In vivo sealing ability of calcium hydroxide-containing root canal sealers: Endod Dent Traumatol, 1989; 5(1); 23-26
33. Chailertvanitkul P, Saunders WP, Mackenzie D, An assessment of microbial coronal leakage in teeth root filled with gutta-percha and three different sealers: Int Endod J, 1996; 29(6); 387-92
34. Almeida JF, Gomes BP, Ferraz CC, Filling of artificial lateral canals and microleakage and flow of five endodontic sealers: Int Endod J, 2007; 40(9); 692-99
35. Haïkel Y, Wittenmeyer W, Bateman G, A new method for the quantitative analysis of endodontic microleakage: J Endod, 1999; 25(3); 172-77
36. Chailertvanitkul P, Saunders WP, MacKenzie D, Weetman DA, An in vitro study of the coronal leakage of two root canal sealers using an obligate anaerobe microbial marker: Int Endod J, 1996; 29(4); 249-55
37. Yücel AC, Güler E, Güler AU, Ertaş E, Bacterial penetration after obturation with four different root canal sealers: J Endod, 2006; 32(9); 890-93
38. Wong JG, Caputo AA, Li P, White SN, Microleakage of adhesive resinous materials in root canals: J Conserv Dent, 2013; 16(3); 213-18
39. Mobarak A, Moussa S, Zaazou A, Abdelfattah H, Comparison of bacterial coronal leakage between different obturation materials (an in vitro study): Alex Dent J, 2015; 40(1); 1-7
40. Hwang JH, Chung J, Na HS, Comparison of bacterial leakage resistance of various root canal filling materials and methods: Confocal laser-scanning microscope study: Scanning, 2015; 37(6); 422-28
41. Pawar SS, Pujar MA, Makandar SD, Evaluation of the apical sealing ability of bioceramic sealer, AH Plus & Epiphany: An in vitro study: J Conserv Dent, 2014; 17(6); 579-82
42. Sultana M, Musani MA, Ahmed IM, An in-vitro comparative study for assessment of apical sealing ability of Epiphany/AH Plus sealer with Resilon/gutta-percha root canal filling materials: J Int Soc Prev Community Dent, 2016; 6(4); 321-26
43. Ballullaya SV, Vinay V, Thumu J, Stereomicroscopic dye leakage measurement of six different root canal sealers: J Clin Diagn Res, 2017; 11(6); ZC65-68
44. Ricucci D, Gröndahl K, Bergenholtz G, Periapical status of root-filled teeth exposed to the oral environment by loss of restoration or caries: Oral Surg Oral Med Oral Pathol Oral Radiol Endod, 2000; 90(3); 354-59
Figures
 Figure 1. Micropipette-mediated inoculation of upper chambers with Enterococcus faecalis.
Figure 1. Micropipette-mediated inoculation of upper chambers with Enterococcus faecalis. Figure 2. Confirmation of Enterococcus faecalis presence in lower chamber broth after growth on blood agar.
Figure 2. Confirmation of Enterococcus faecalis presence in lower chamber broth after growth on blood agar. Figure 3. Cumulative percentages of leaking specimens over time. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste.
Figure 3. Cumulative percentages of leaking specimens over time. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste. Figure 4. Percent of non-leaking samples (ie, percent survival) during the 33-day experiment. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste.
Figure 4. Percent of non-leaking samples (ie, percent survival) during the 33-day experiment. Sealers used in this study: Canason, Canason zinc oxide eugenol root-canal filling cement; AH 26, AH 26 epoxy resin; Well-Root ST, Well-Root ST antibacterial bioceramic paste. Most Viewed Current Articles
13 Apr 2020 : Original article 19,586
Outcome of 24 Weeks of Combined Schroth and Pilates Exercises on Cobb Angle, Angle of Trunk Rotation, Chest...DOI :10.12659/MSMBR.920449
Med Sci Monit Basic Res 2020; 26:e920449
20 Apr 2018 : Original article 17,466
Brain Training Games Enhance Cognitive Function in Healthy SubjectsDOI :10.12659/MSMBR.909022
Med Sci Monit Basic Res 2018; 24:63-69
23 Jul 2016 : Review article 10,353
Cardiac Hypertrophy: An Introduction to Molecular and Cellular BasisDOI :10.12659/MSMBR.900437
Med Sci Monit Basic Res 2016; 22:75-79
10 Aug 2020 : Clinical Research 9,788
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Rando...DOI :10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264