27 December 2021: Human Study
A National Cross-Sectional Study from the Republic of Kosovo on Lot Quality Assurance Sampling (LQAS) to Evaluate the Vaccination Status of Children Between 12 and 24 Months of Age During 2018 to 2020
Bashkim Bejtë GllarevaDOI: 10.12659/MSMBR.934194
Med Sci Monit Basic Res 2021; 27:e934194
Abstract
BACKGROUND: In the Republic of Kosovo, full vaccination status in children under age 2 years includes: 1 dose of Bacillus Calmette-Guérin (BCG) hepatitis B virus (HBV) vaccine; 3 doses of diphtheria, tetanus, pertussis, hepatitis B, polio, and Haemophilus influenzae type b (DTaP-HB-IPV-Hib) vaccine; 3 doses of inactivated polio vaccine (IPV); and 1 dose of measles, mumps, and rubella (MMR) vaccine. Lot quality assurance sampling (LQAS) is a method used to assess the performance of health quality indicators.
MATERIAL AND METHODS: A national cross-sectional study with children aged between 12 and 24 months from Kosovo was performed between 2018 and 2020. The vaccination status of children was assessed with lot quality assurance sampling (LQAS) using randomized samples.
RESULTS: Among 430 children, more than 90% had completed the full immunization schedule. Delays in children’s immunizations were observed. Most vaccinations showed short delays of less than 1 month, followed by delays of up to 3 months. The main reason for vaccination delay was the COVID-19 pandemic, following by child’s illness at the scheduled time of vaccination or the parents were too busy to take the child to the vaccination site. Meanwhile, child age was the only parameter that showed difference among non-vaccinated and fully vaccinated (P<0.001).
CONCLUSIONS: LQAS analysis showed that between 2018 and 2020 lack of full immunization was due to delay caused by the parent not taking the child to the vaccination site, which may be prevented by improving information given to parents and the use of vaccination reminders.
Keywords: Kosovo, Lot Quality Assurance Sampling, Vaccination coverage, COVID-19, Child, Preschool, Cross-Sectional Studies, Disease Outbreaks, Female, Humans, Immunization, Immunization Schedule, Infant, Male, Pandemics, SARS-CoV-2, Vaccination, Vaccination Hesitancy, Vaccines
Background
The Coronavirus disease 2019 (COVID-19) pandemic has triggered much discussion on the topic of vaccination and immunization coverage and has delayed national programs to vaccinate children [1]. Through immunizations, once-common infectious diseases such as measles, mumps, and whooping cough have been eliminated as threatening diseases for children. The aim of immunization is to prevent disease. Immunization not only protects against serious diseases but also prevents the spread of those diseases to others or at least reduces their severity [2]. Immunization decreases disease spread, benefiting both vaccinated and unvaccinated people. For example, diseases such as polio and smallpox have been eradicated due to mass immunization [3].
Although children receive most of the vaccinations, adults also need to be sure they are already immune to certain infections and stay up to date on certain vaccinations because childhood illnesses can have significant impact on adults and can cause complications; therefore, it is necessary to vaccinate people at the proper age. The benefit of doing so is that children are protected from diseases that could cause them serious health problems in adulthood. The recommendation for children aged 0–6 years are diphtheria, tetanus, pertussis (DTaP), Haemophilus influenzae type B (Hib), hepatitis B (HepB), rotavirus, pneumonococcal disease (PCV13), poliovirus (IPV), influenza, measles, mumps, rubella, varicella (chickenpox), hepatitis A (HepA), and meningococcus [1]. The current Center of Disease Control (CDC) recommendations for vaccinating children ages 12–24 months are: chickenpox (varicella) (1st dose), DTaP (4th dose), Hib (4th dose), measles, mumps, and rubella (MMR) (1st dose), IPV (3rd dose), PCV13 (4th dose), HepA (1st dose), HepB (3rd dose between 6 months and 18 months), and influenza (Flu) (every year) [4]. In the past, these diseases were life-threatening for children, but now, due to established vaccination programs, they are not [5].
The immunization coverage of some preventable diseases, such as measles, has declined in the last decade. Vaccine hesitancy (VH), defined as the reluctance or refusal to vaccinate, might be one of the problems causing this phenomenon [6]. Reasons given for refusing vaccination include lack of confidence and trust in the safety and efficacy of vaccines, lack of confidence in administration of the vaccine, distrust of healthcare professionals, and lack of trust in the pharmaceutical industry [7]. Moreover, social networks and media have promoted these doubts among parents [8,9]. Surprisingly, in the European Union, one-fifth of parents have doubts about vaccinating their children [10], involving vaccine safety, false associations between vaccines and other diseases, and even the mistaken belief that vaccination has more risks than benefits [11]. Due to this phenomenon, it is interesting to assess the immunization coverage of children with vaccines that are part of the mandatory national strategy of vaccinations. Therefore, this national cross-sectional study from the Republic of Kosovo aimed to use lot quality assurance sampling (LQAS) to evaluate the vaccination status of children 12–24 months of age from 2018 to 2020, to determine if the immunization of children is done at the appropriate age, and to determine the most important reasons for vaccination refusal and vaccination delay.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
The study was designed as a national cross-sectional study with a clearly defined population of children. The study population included all children in Kosovo ages 12–24 months, which provided a pool of about 29 000 children.
The screening of children has been performed with lot quality technique monitoring, which ensures the assessment of immunization coverage through aggregation of data from all units in the area being monitored. LQAS is a statistical methodology that first obtains random samples from a specific area or district within a selected region, where lots are then classified and labeled. The second step is estimation of prevalence [12]. In the present study, the research on the quality of vaccination services using lots consisted in assessing the degree of vaccination coverage. Degree of accuracy 5% and degree of reliability 95% were selected, and based on these a minimum sample size of 384 children ages 12–24 months was estimated. The number of children required was then determined within each area. At this stage, the areas were listed according to lot number. The lot was equivalent to the municipalities in Kosovo. The Republic of Kosovo consists of 26 municipally unit areas; therefore, these regions have been used as lots. Each lot had a sample size of 15 children, forming a total pool of 390 children.
The choice of a child for the involvement in the research was random. In our study the vaccine coverage with Bacillus Calmette-Guérin (BCG), hepatitis B virus (HBV), diphtheria, tetanus, pertussis, hepatitis B, polio, and Haemophilus influenzae type b (DTaP-HB-IPV-Hib), inactivated polio vaccine (IPV), and measles, mumps, and rubella (MMR) were evaluated.
Assessment criteria were used to assess the immunization coverage of children as “fully immunized”, “partially immunized”, or “not fully immunized”. The criteria were set by the national schedule strategy of childhood immunization as follows:
The quality data were also gathered, namely, the data on vaccination time and causes of immunization failure or delay were collected:
SCREENING AND DATA COLLECTION:
Selection of samples was random. Interviewers were trained how to do interviews in the field. After training on how to select the case/household, data on immunization coverage of children were collected by 2 questionnaires; (i) immunization coverage and (ii) a form to determine the reasons for non-vaccination or partial vaccination, focusing on parental education, anti-vaccination movements, or problems pertaining to health services. The immunization status was double-checked with the immunization cards.
Screening of households was implemented by selecting the samples according to the following information:
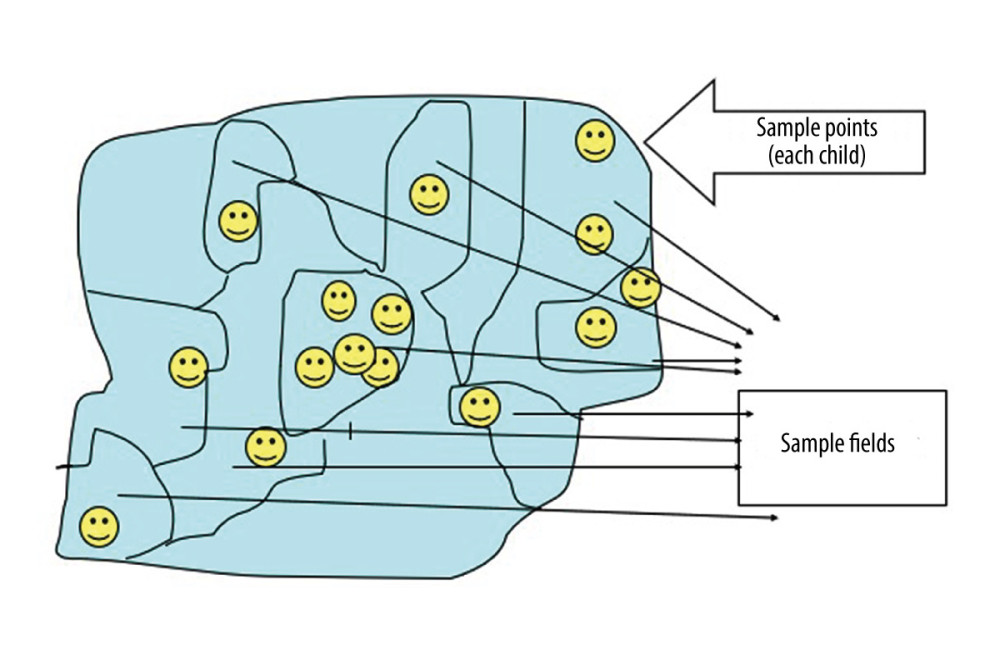
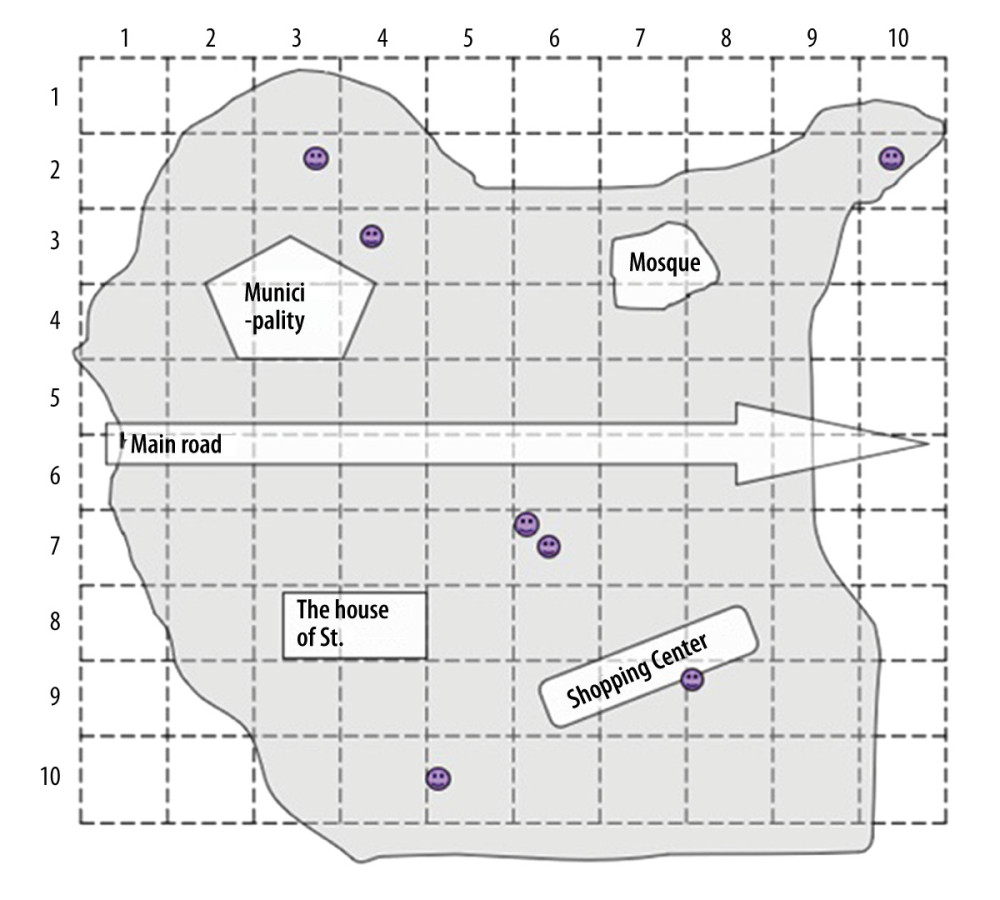
When we could not obtain any available list of residential houses, but the area could be divided into parts in which the number of residential houses or population figures are known, we used subdivisions/sample fields (Figure 1). When the list of residential houses was not available and the area had no subdivision, we used a grid placed on a map to identify sample point areas (Figure 2).
In the first case, subdivisions were presented as residential households. Interviewers went to the approximate center of the sampling field and randomly chose the direction of the first house. Then, the total number of houses along the randomly selected direction were counted and the interviewer randomly chose a number between 1 and the number of counted houses (Figure 1). This number represented the sample point at which we were looking for children aged 12–24 months. If we needed to find more than 1 sample point (child), the same steps were repeated.
In the second case, when the area was large and had several subdivisions (like the city center) and we did not know the approximate population, we used a map and/or sketches. The map was divided into grids with 10 rows and columns. We randomly selected the grids to get enough samples for our sample points. If the grid square contained no households, then we chose another to replace it. We went to each randomly selected grid and proceeded with the screening. The step was repeated to randomly select our sample points/children (Figure 2).
When we got to the house we chose, we first verified whether they had children eligible for the interview (a child who is 12–24 months of age on the day of the interview). If there were no children of this age group, we then went to the nearest house in the same direction we chose randomly. In this way we continued until we found a child of the appropriate age. We only interviewed 1 child at each home and never more. If there were more than 1 child in the house for the interview, we randomly chose one of them. For example, if there were 2 children, we marked them as #1 and #2 and then randomly selected one of them. If there were 3, then we marked them as #1, #2, and #3 and randomly selected one of them. The questionnaires were given in paper form to the parents or legal guardians. Children were allowed to participate when consent from their parents was obtained. Immunization status was obtained from the vaccination cards. Interviewers asked whether the child had an immunization card, and if so, asked the respondent to allow the card to be photographed. The photographs were assessed by 2 reviewers, who classified children according to whether they had missed a vaccine dose that was scheduled.
STATISTICAL ANALYSIS:
Data analysis was performed using SPSS software package (IBM, New York, USA). Descriptive analysis of the data was carried out. Pearson’s chi-square or Fisher’s exact test was performed for analyzing factors for non-vaccination. Statistical significance for all tests was set at α=0.05.
Results
Overall, 430 children were assessed in this research, with a mean age of 13.3±6.3 months. Distribution among child gender was equal (Table 1). Of the total number of parents, the majority had completed secondary education.
Information on immunizations from 2018 to 2020 among 430 children showed that more than 90% had completed the full immunization schedule. Only a minority of children were not immunized or partially immunized. However, despite the high levels of full immunization coverage, delays in children’s immunizations were observed (Table 2). With the majority of vaccinations, short delays of less than 1 month were observed, followed by delays of up to 3 months.
The reasons for not vaccinating their children or reasons for delay were gathered in 70 cases (Table 3). As the part of the survey was performed during 2020, the main reason for delay in vaccinating children was the COVID-19 pandemic (n=27 cases), and the second most common reason was that the child was sick at the time of scheduled vaccination or the parents were too busy to take the child for vaccination. Other reasons are presented in Table 3.
Younger children were more likely to have complete immunization coverage and older children were more likely to have only partial immunization coverage (Table 4). Other factors such as age of parents or parent’s level of education were not significantly correlated with the immunization coverage, but proportionally more mothers with high or secondary level of education had children who had complete immunization coverage.
Discussion
The development of vaccines is one of the greatest achievements of modern medicine. Preventive vaccines and immunization strategies have reduced infant mortality rates. Although the overall coverage was found to be high in the present study (over 90% of sampled children were fully vaccinated), among the same children delays in immunizations were observed. The reality of immunizations in the current study population showed many children had fallen behind the recommended timelines of immunizations (Table 2). Most of these delays were short (1–3 months), but milestone analysis of immunization timelines showed late immunizations between birth and age 24 months compared to recommended immunization timelines, and some children had delays of more than 1 year. Similar results were also reported in the USA by Robison et al [13]; however, they found out that most children (not a minority, as in our study) were not catching up at each milestone and were not up to date with immunizations. This was mainly due to not having encounters with immunization providers or missed opportunities at the visit. Additional findings which also correlate with our delays were provider reluctance to administer vaccinations during sick or other non-routine visits, namely in emergency care, with limited time and pressure of other priorities making it difficult to include review of records and administration of vaccines, as well as parental reluctance or parental belief that their child has received all needed care, including immunizations. For instance, in Kosovo, due to the high rate of vaccinations against measles and diphtheria, we do not have registered cases of these 2 diseases. Mumps cases are presently affecting certain groups of the population that have not received 2 doses of mumps vaccine. The situation is quite different with the BCG vaccine. Vaccination is provided in maternity hospitals and the coverage rate is up to 98% in our region. Despite this, Kosovo has a high incidence of tuberculosis. However, in our country, a national strategy and program for immunizing children below 2 years of age is implemented and shows good coverage. Nevertheless, there has been a recent increase in vaccine hesitancy and reluctance, especially now regarding COVID-19 vaccines.
In addition to the factor presented above, the COVID-19 pandemic has significantly disrupted health services throughout the world due to social distancing, healthcare staff shortages, and increase of COVID-19 cases [14]. Specially, childhood immunizations were interrupted and concerns about bringing children to health facilities have contributed to reduced vaccine coverage [15,16]. However, a study from Africa suggested that the number of child deaths due to vaccine-preventable diseases would exceed the number of deaths due to COVID-19 acquired during visits to health facilities when obtaining regular vaccination [17]. Studies showed that due to COVID-19, vaccine coverage was reduced and reported cases of previously controlled vaccine-preventable disease have increased [18–21]. Our research showed a delay in 27 out of 70 cases due to the COVID-19 pandemic. Thus, the results confirm the negative impact of the COVID-19 pandemic on immunization coverage. The results also show other more worrying causes for non-vaccination or delays in vaccinations. Based on the responses to the questionnaire, the findings suggest that parents were too busy to bring their child for vaccination, and secondly, they were afraid of adverse effects. Moreover, there was also a perception that vaccine-preventable diseases are no longer a risk for children. Lastly, in our population, cases of VH were also reported, which in other studies was confirmed to contribute to declines in vaccine coverage in recent years [6–9,22]. For instance, we registered 2 cases of total parental refusal to vaccinate children. There were also recorded cases of parents being unaware or misinformed about vaccinations. To have an opportunity for vaccination, a child must be brought to a health care facility several times during the first year of life. The complexity of the vaccination schedule, requiring multiple visits to health care facilities, might be why some parents are unaware or are too busy.
In Kosovo, the overall vaccination coverage is not worrying, but it was observed that parents are delaying the vaccination of their children. Accessing immunization services and possible VH have been triggers that resulted in delay and non-vaccination during the pandemic. Therefore, it is critical to build parental the trust and confidence regarding children’s immunization and to reduce limitations where possible. Silveira et al [23] who analyzed the Brazilian population, showed that the coverage typically increased with family wealth, although VH was higher among the better-off and wealthiest families. The potential characteristics or factors, which were limited in our survey and might affect the non-vaccinations, are age of parents or level of education, which were not significantly correlated with the immunization coverage according to our results, but proportionally more mothers with high or secondary level of education had children with full immunization coverage. This appears to contradict the findings of Silveira et al [23], but our study did not evaluate the social-economic status of interviewed families. On the other hand, a study by Gebre Eyesus et al [24] indicated that parents who received immunization service more times were more likely to be knowledgeable about vaccines than parents who received the service only once. This might be attributed to the health education provided by healthcare professionals during the visits. A similar correlation between the mother’s educational level and vaccination status was observed in the same study. The maternal educational status enhanced parental health-seeking behavior and had a positive effect on immunization coverage.
An interesting finding of our study is that fully immunized children were on average younger than 1 year of age. This confirms that most parents are following the national strategy of immunizations and that in Kosovo we have a problem of delayed vaccinations. The mean age of non-immunized children was 16 months, while that of partially immunized was 19 months. In this 3-month difference, you are able to partially cover the immunization schedule, and the status of a child might convert from non-immunized to partially immunized. Therefore, it is extremely important to educate parents to continue with the vaccination schedule despite the delay, as the schedule might be prolonged to up to 3 years of age instead doing it only in the first 2 years after birth. It is also important to increase parents’ knowledge of the benefits of vaccines. This will be possible only when the whole community of healthcare professionals are themselves vaccinated and then disseminate clear health messages on benefits, improve overall accessibility in facilities despite pandemic conditions, implement national strategies in countries that are lacking, and develop an effective strategy to overcome VH [1]. Increasing the responsibilities of family medicine practitioners in the vaccination process necessary because they are the first contact with the public. Therefore, general practitioners as primary healthcare providers must become more concerned about immunization rates of their young patients. One way to help minimize delays and vaccinate children at an appropriate age is to track their progress through immunization milestones between birth and age 2 years [25]. Healthcare providers, especially in communities with low immunization rates, need to provide catch-up immunizations and have effective recall programs in place that provide timely reminders to parents when vaccinations are scheduled.
As was observed in our analysis, parental forgetfulness and busy schedules have a substantial impact on whether a child becomes fully immunized with all recommended vaccines. We are living in a digital era, so to overcome the barrier of parental forgetfulness, one solution is immunization appointment reminders and calls [26]. SMS or e-mail reminders can notify parents about upcoming or missed appointments, while recalls inform parents when their child is overdue for an immunization. Previous studies showed that such an approach is effective and increases appointment attendance rates [27–29]. In addition to reminders, it is important to also explore other strategies for increasing demand for vaccines and attendance at appointments. Although not as effective as expected, interventions to combat VH and fears of adverse effects are needed. The COVID-19 pandemic may influence public views on vaccinations and thus affect children’s immunizations, but we cannot yet tell in which direction [26].
Our study has some limitations. Screening with the lot technique has disadvantages; for example, coverage in individual units can be acceptable or not acceptable as specific levels of coverage can be calculated only for all the units in the study. Selecting lot sample size involves the assessment of risks. Moreover, participation in our study was voluntary, which could have led to a selection bias, so our results to some extent cannot be generalized to the whole country. Resistance of child caregivers who did not participate might be due to VH; thus, we identified only a small proportion of VH parents, and we underestimated the problem of VH. Another limitation is that data were collected between 2018 and 2020, so our results may not reflect behavior and thinking of parents before or after the COVID-19 pandemic. Therefore, as mentioned, the results might not necessarily apply to Kosovo today. A major strength of the study is that this is the first such survey in Kosovo and it covered the whole country, making it a valuable contribution to understanding the status of childhood vaccination and immunization coverage in Kosovo.
Conclusions
This is the first such study conducted in our region and showed that overall coverage in Kosovo is above 90%. LQAS analysis showed that from 2018 to 2020, lack of full immunization was due to delays in vaccination scheduling, which may be prevented by improving information given to parents and by the use of vaccination reminders.
The findings also confirmed that the major reason for non-vaccinations and delay was the COVID-19 pandemic, as proved in many other studies. Moreover, only a small proportion of parent showed VH. They had lower levels of education, so educational status and knowledge status of parents was an important improvement point correlated with immunization. Public health education must be intensified through the provision of information on vaccines, education on adverse effects and materials, and distributed by family medicine practitioners to enhance the knowledge of parents about immunization and vaccine-preventable diseases.
References
1. Nuzhath T, Ajayi KV, Fan Q, Childhood immunization during the COVID-19 pandemic in Texas: Vaccine, 2021; 39(25); 3333-37
2. Bedford H, Elliman D, Concerns about immunisation: BMJ, 2000; 320(7229); 240-43
3. Greenwood B, The contribution of vaccination to global health: Past, present and future: Philos Trans R Soc Lond B Biol Sci, 2014; 369(1645); 20130433
4. Centers for Disease Control and Prevention (CDC): Vaccines for your Children. 12 to 23 months. 2021 Available from: https://www.cdc.gov/vaccines/parents/by-age/months-12-23.html
5. Lee Ventola C, Immunization in the United States: Recommendations, barriers, and measures to improve compliance. Part 1: Childhood vaccinations: PT, 2016; 41(7); 426-36
6. MacDonald NE, Eskola J, Liang X, Vaccine hesitancy: Definition, scope and determinants: Vaccine, 2015; 33(34); 4161-64
7. Larson HJ, Cooper LZ, Eskola J, Addressing the vaccine confidence gap: Lancet, 2011; 378(9790); 526-35
8. Salmon DA, Moulton LH, Omer SB, Knowledge, attitudes, and beliefs of school nurses and personnel and associations with nonmedical immunization exemptions: Pediatrics, 2004; 113(6); e552-59
9. Karafillakis ELHA consortium, Vaccine hesitancy among healthcare workers in Europe: A qualitative study: Vaccine, 2016; 34(41); 5013-20
10. Stefanoff P, Mamelund SE, Robinson M, Tracking parental attitudes on vaccination across European countries: The Vaccine Safety, Attitudes, Training and Communication Project (VACSATC): Vaccine, 2010; 28(35); 5731-37
11. Elizondo-Alzola U, Carrasco MG, Pinós L, Vaccine hesitancy among paediatric nurses: Prevalence and associated factors: PLoS One, 2021; 16(5); e0251735
12. Pagano M, Valadez JJ, Commentary: Understanding practical lot quality assurance sampling: Int J Epidemiol, 2010; 39(1); 69-71
13. Robison SG, Kurosky SK, Young CM, Immunization milestones: A more comprehensive picture of age-appropriate vaccination: J Biomed Biotechnol, 2010; 2010; 916525
14. Nelson R, COVID-19 disrupts vaccine delivery: Lancet Infect Dis, 2020; 20(5); 546
15. Saso A, Skirrow H, Kampmann B, Impact of COVID-19 on immunization services for maternal and infant vaccines: Results of a survey conducted by imprint-the immunising pregnant women and infants network: Vaccines (Basel), 2020; 8(3); 556
16. Olorunsaiye CZ, Yusuf KK, Reinhart K, Salihu HM, COVID-19 and child vaccination: A systematic approach to closing the immunization gap: Int JMCH AIDS, 2020; 9(3); 381-85
17. Abbas K, Procter SR, van Zandvoort K, Routine childhood immunisation during the COVID-19 pandemic in Africa: A benefitrisk analysis of health benefits versus excess risk of SARS-CoV-2 infection: Lancet Glob Health, 2020; 8(10); e1264-72
18. Chandir S, Siddiqi DA, Mehmood M, Impact of COVID-19 pandemic response on uptake of routine immunizations in Sindh, Pakistan: An analysis of provincial electronic immunization registrydata: Vaccine, 2020; 38(45); 7146-55
19. Ahmadi A, Essar MY, Lin X, Polio in Afghanistan: The current situation amid COVID-19: Am J Trop Med Hyg, 2020; 103(4); 1367-69
20. Alrabiaah AA, Alshaer AH, Estrella SMC, Effects of the Coronavirus disease 2019 pandemic on routine pediatric immunization coverage rates at the main University Hospital inSaudi Arabia: Saudi Med J, 2020; 41(11); 1197-203
21. Buonsenso D, Cinicola B, Kallon MN, Iodice F, Child healthcare and immunizations in Sub-Saharan Africa during the COVID-19 pandemic: Front Pediatr, 2020; 8; 517
22. Domingues C, Maranhão AGK, Teixeira AM, The Brazilian National Immunization Program: 46 years of achievements andchallenges: Cad Saude Publica, 2020; 36(Suppl 2); e00222919
23. Silveira MF, Tonial CT, Maranhão AGK, Missed childhood immunizations during the COVID-19 pandemic in Brazil: Analyses of routine statistics and of a national household survey: Vaccine, 2021; 39(25); 3404-9
24. GebreEyesus FA, Tarekegn TT, Amlak BT, Knowledge, attitude, and practices of parents about immunization of infants and its associated factors in Wadla Woreda, North East Ethiopia, 2019: Pediatric Health Med Ther, 2021; 12; 223-38
25. Luman ET, Chu SY, When and why children fall behind with vaccinations. Missed visits and missed opportunities at milestone ages: Am J Prevent Med, 2009; 36(2); 105-11
26. Jong KM, Sikora CA, MacDonald SE, Childhood immunization appointment reminders and recalls: Strengths, weaknesses and opportunities to increase vaccine coverage: Public Health, 2021; 194; 170-75
27. Briss PA, Rodewald LE, Hinman AR, Reviews of evidence regarding interventions to improve vaccination coverage in children, adolescents, and adults: Am J Prev Med, 2000; 18; 97-140
28. Jacobson Vann JC, Szilagyi P, Patient reminder and recall systems to improveimmunization rates: Cochrane Libr, 2005; 3; 1-71
29. Williams N, Woodward H, Majeed A, Saxena S, Primary care strategies to improve childhood immunisation uptake in developed countries: Systematicreview: JRSM Short Rep, 2011; 2; 1-21
Figures
Tables
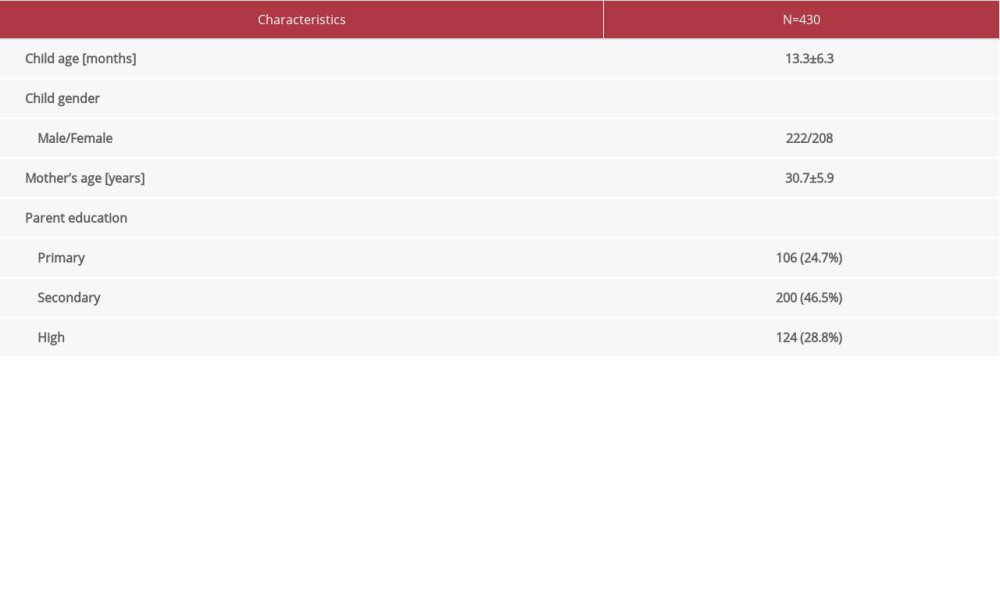 Table 1. Basic characteristics of participants.
Table 1. Basic characteristics of participants.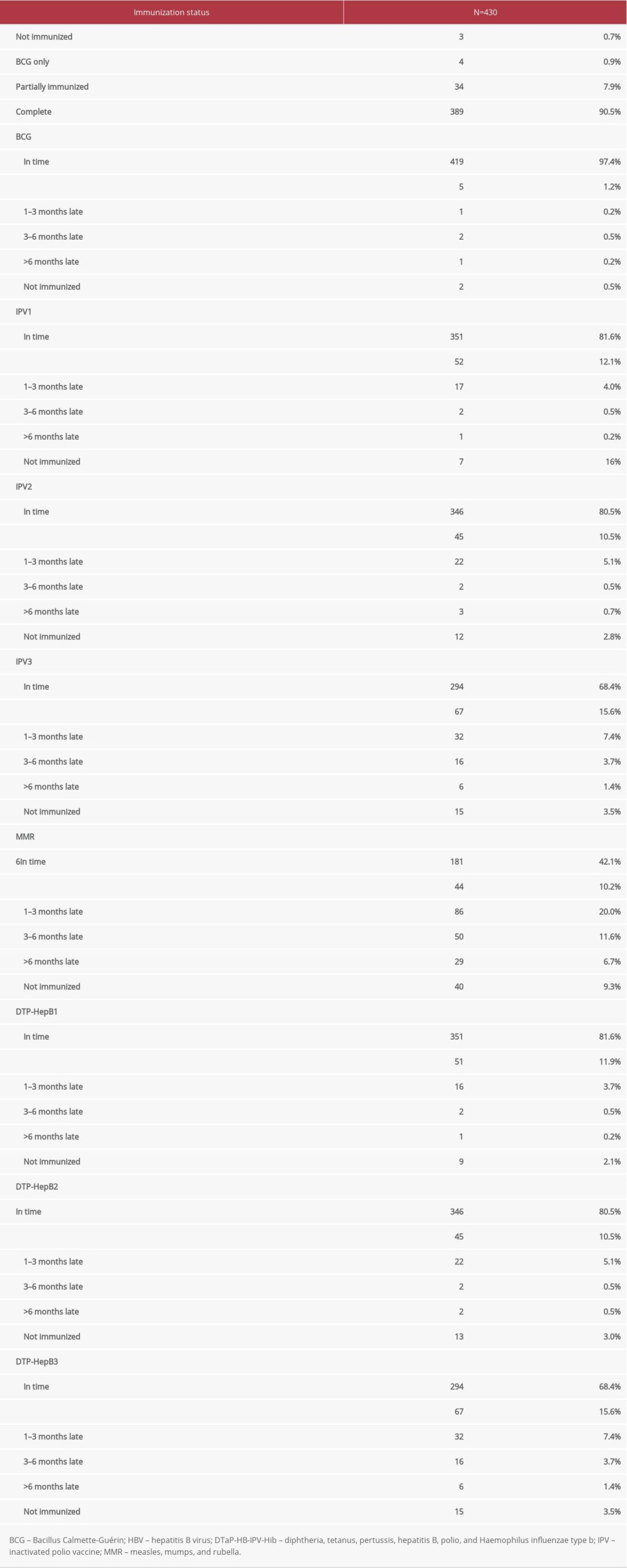 Table 2. Immunization status of children on the day of screening.
Table 2. Immunization status of children on the day of screening.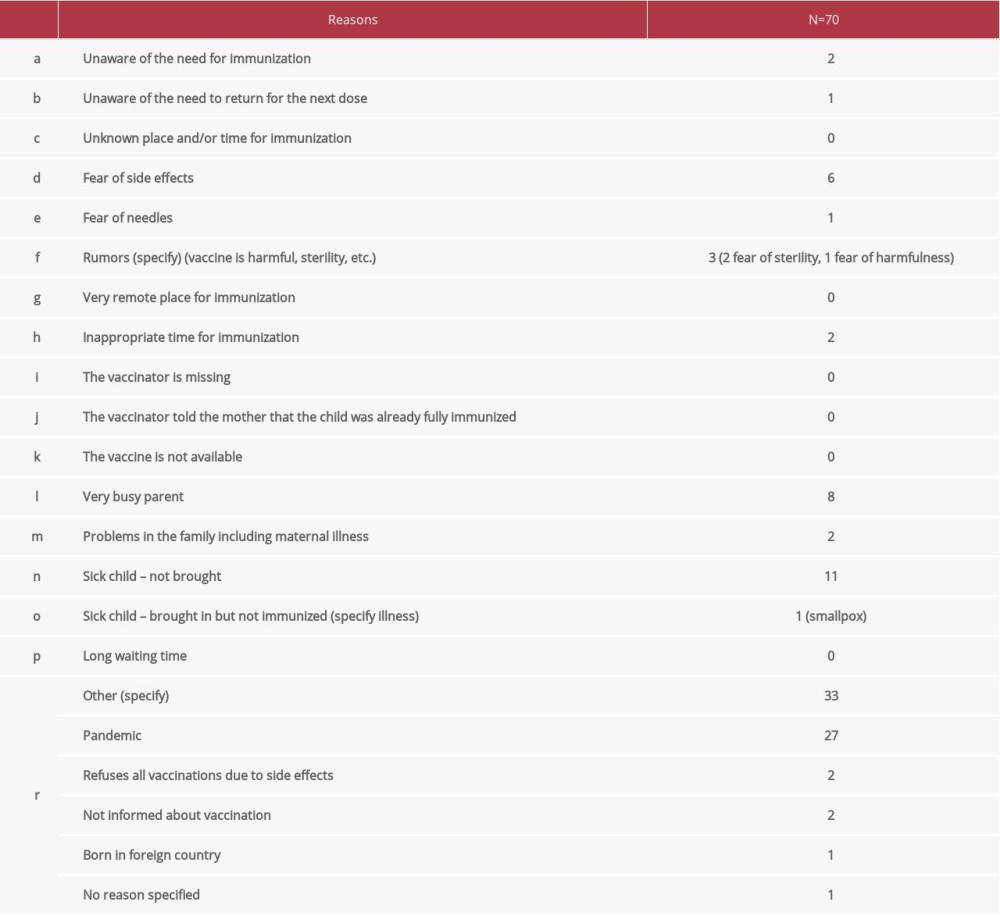 Table 3. Reasons for not vaccinating or delay in vaccinations.
Table 3. Reasons for not vaccinating or delay in vaccinations.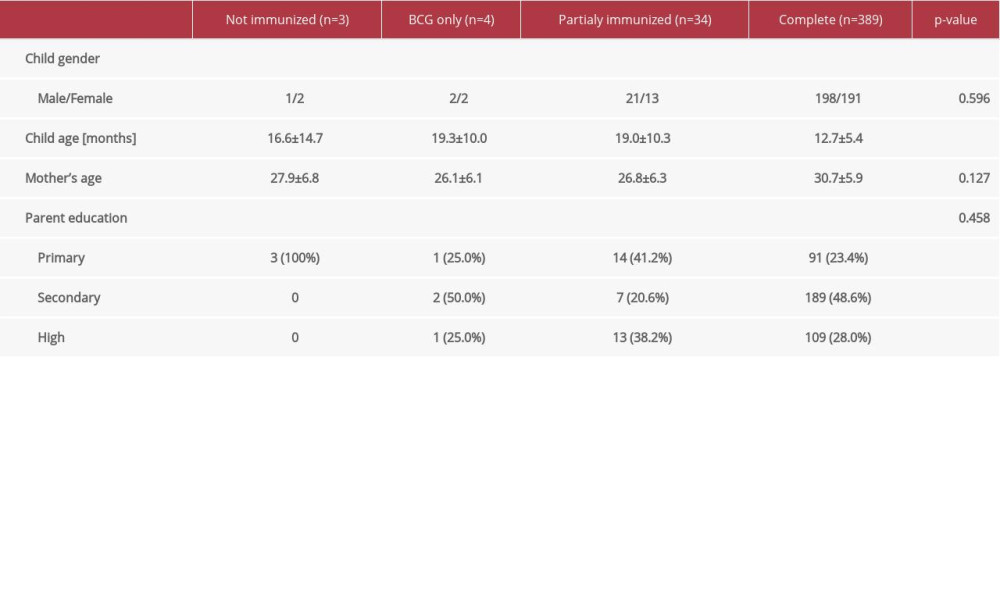 Table 4. Differences in basic parameters among children with different immunization status
Table 4. Differences in basic parameters among children with different immunization status Table 1. Basic characteristics of participants.
Table 1. Basic characteristics of participants. Table 2. Immunization status of children on the day of screening.
Table 2. Immunization status of children on the day of screening. Table 3. Reasons for not vaccinating or delay in vaccinations.
Table 3. Reasons for not vaccinating or delay in vaccinations. Table 4. Differences in basic parameters among children with different immunization status
Table 4. Differences in basic parameters among children with different immunization status Most Viewed Current Articles
13 Apr 2020 : Original article 19,586
Outcome of 24 Weeks of Combined Schroth and Pilates Exercises on Cobb Angle, Angle of Trunk Rotation, Chest...DOI :10.12659/MSMBR.920449
Med Sci Monit Basic Res 2020; 26:e920449
20 Apr 2018 : Original article 17,466
Brain Training Games Enhance Cognitive Function in Healthy SubjectsDOI :10.12659/MSMBR.909022
Med Sci Monit Basic Res 2018; 24:63-69
23 Jul 2016 : Review article 10,353
Cardiac Hypertrophy: An Introduction to Molecular and Cellular BasisDOI :10.12659/MSMBR.900437
Med Sci Monit Basic Res 2016; 22:75-79
10 Aug 2020 : Clinical Research 9,788
Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Rando...DOI :10.12659/MSMBR.925264
Med Sci Monit Basic Res 2020; 26:e925264